Abstract
Background:
The clinical presentations in patients with SARS-CoV-2 (COVID-19) infections range from asymptomatic upper respiratory infections to acute respiratory failure with bilateral pulmonary infiltrates requiring mechanical ventilation. Clinicians often measured inflammatory markers in hospitalized patients to characterize the severity of the infection. Multiple studies published in 2020 reported information on the frequency of elevated inflammatory markers in hospitalized patients in various categories of disease severity.
Methods:
The PubMed database was searched using the terms “Inflammatory markers in COVID-19 patients” and “Clinical features of patients infected with COVID-19.” Thirty-three publications were analyzed in detail to determine which inflammatory markers were increased and the frequency of these increases in various clinical classifications, including patients requiring hospitalization, patients with pneumonia, patients with severe infection, patients requiring intensive care unit admission, and patients who died.
Results:
C-reactive protein (CRP) was the most frequently elevated inflammatory marker in all categories. Ferritin, D-dimer, and erythrocyte sedimentation rate levels were also frequently elevated. In general, frequencies were higher in patients with more severe infections. For example, 24 out of 24 patients who died had an elevated CRP level.
Conclusion:
This review provides concrete information about the frequency of various inflammatory markers in patients with COVID-19 infection who required hospitalization. It also provides us some insight into the approach clinicians took in the early phase of this pandemic in their efforts to characterize these patients and perhaps understand the disease process better. However, these results might suggest that clinicians and laboratory directors should develop protocols to optimize laboratory testing.
Introduction
The COVID-19 pandemic began in late December 2019 in China and subsequently spread worldwide throughout 2020. These infections caused significant morbidity and mortality and overwhelmed healthcare systems. Most clinicians were confronted with situations that they had never seen before and were uncertain about the best methods to decrease the spread of infection and to manage infected patients. Initial patient evaluation often included routine laboratory tests, chest radiographs, chest computed tomography, and cultures to rule out bacterial pathogens. Many clinicians measured inflammatory markers in these patients. Relevant questions associated with these tests included: First, are these markers increased in COVID-19 infections, and, if so, which markers are increased? Second, do the levels of inflammatory markers predict the clinical severity in these cases? This question is particularly relevant when levels on admission are analyzed. For example, does a very high level of a marker predict respiratory failure and the need for mechanical ventilation. Third, do particular inflammatory markers have pathogenetic significance? Additional considerations include the trend or trajectory of individual markers and potential significance of a combination of elevated markers. For example, the level of CRP might reflect the total inflammatory response and ongoing tissue injury. The level of D-dimer might reflect the potential for significant clot formation and provide an indication for therapeutic anticoagulation.
The widespread clinical activity during this pandemic led to multiple publications reporting the epidemiology of these infections, clinical presentations, laboratory test results, treatment trials, and morbidity and mortality. Clinicians measured inflammatory marker levels in an effort to gauge the severity of infection and possibly predict outcomes. Multiple publications have reported the results with inflammatory markers in these patients, and this information forms the basis for this review.
Methods
Literature Search Strategy
A literature search in PubMed was conducted to identify studies published in 2020 reporting inflammatory markers in COVID-19 patients. The specific search terms include “Inflammatory markers in COVID-19 patients” and “Clinical features of patients infected with COVID-19.” Based on the intensive literature search, 783 articles that reported the inflammatory marker results in COVID-19 patients were retrieved. Thirty-three reports on the inflammatory markers in COVID-19 patients were selected for complete review. The study screening strategy and exclusion reasons are outlined in Figure 1.

Article search and selection strategy.
Eligibility Criteria and Data Extraction Procedure
All articles that reported inflammatory markers in COVID-19 patients were reviewed. Records were checked for duplicates. Quality criteria were developed before starting the review of full papers. Inclusion/exclusion criteria were defined regarding the relevance of the articles to the research questions of interest. The inclusion criteria included studies that were published in 2020 that reported the inflammatory markers measured during the initial evaluation of hospitalized COVID-19 patients and clearly recorded the number and percentage of patients who had an inflammatory marker elevated during the hospital admission. Articles that met the above criteria were considered for the final review. Titles were checked twice in both excluded and included lists before the start of the data extraction process to avoid missing publications. Those papers considered relevant were retained, and their results were extracted into a word document. Data extracted from valuable papers included author’s name, year of publication, PMID, study country and total number of patients. See the Supplemental Files for article summaries and reference list.
Results
From all screened studies, 33 articles were included in the analysis. A summary of these studies can be found in the Supplemental File. Of these 33 articles, 20 articles did not develop a clinical classification of the patients and included all cases admitted to the hospital, 5 articles classified the patients as ICU and non-ICU cases, 5 articles classified the patients as severe and non-severe cases, 2 articles classified the patients as survivors and non-survivors, and 1 article classified the patients as with pneumonia and without pneumonia.
In the studies that included all hospitalized patients, CRP was the most frequently elevated inflammatory marker, followed by ESR (Table 1).
No Specific Patient Classification.

The numerator represents the number of patients with an elevated inflammatory marker based on the criteria used by the individual laboratory in the studies. Then denominator is the total number of patients in the studies.
In the studies that classified the patients as either with or without pneumonia, CRP was the most frequently elevated inflammatory marker in both the categories (Table 2). Ferritin was the second most frequently elevated inflammatory marker in both patient groups. Pneumonia was defined as radiologically or clinically confirmed if findings were present within 48 h after admission.
Patients Classified With Pneumonia Versus No Pneumonia.
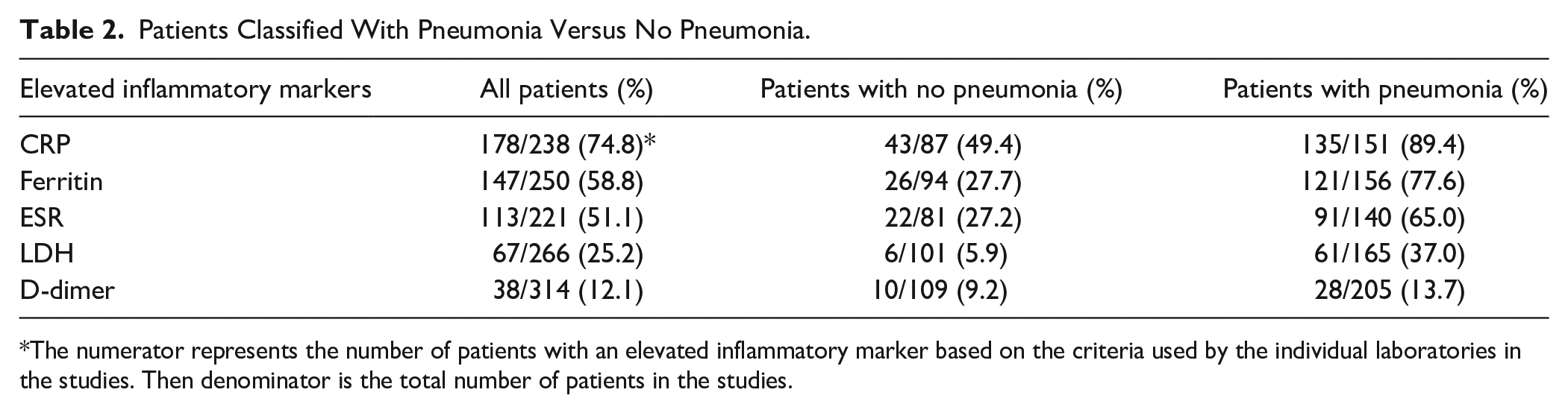
The numerator represents the number of patients with an elevated inflammatory marker based on the criteria used by the individual laboratories in the studies. Then denominator is the total number of patients in the studies.
In the studies that classified patients as either severe or non-severe cases, ESR was the most frequently elevated inflammatory marker in both categories followed by CRP in both the categories (Table 3). Zhang et al 1 defined severe COVID cases as a case with one of the following criteria: respiratory frequency ≥30 per minute, oxygen saturation ≤93% at rest, and oxygenation index PaO2/FiO2 ≤ 300 mmHg. In one study, 2 the classification was based on the Seventh Edition of the Chinese National Health Commission and patients should meet any of the following criteria: (1) shortness of breath, respiratory rate ≥30 breaths per minute; (2) oxygen saturation ≤93% at rest; (3) PaO2/FiO2 ≤ 300 mmHg; and (4) lung images showing obvious progression of the opacities size >50% within 24 to 48 h. Three studies3-5 used criteria based on the Fifth Version of the Chinese National Health Commission which required severe/critical patients to meet any of the following conditions: (1) respiratory rate of 30 breaths per minute or greater; (2) peripheral oxygen saturation of 93% or less at rest; (3) PaO2/ FiO2 ≤ 300 mmHg; (4) respiratory failure with mechanical ventilation; (5) shock; and (6) patients with other organ failure who needed intensive care unit and admission.
Patients Classified as Severe Infection Versus Non-Severe Infection.

The numerator represents the number of patients with an elevated inflammatory marker based on the criteria used by the individual laboratories in the studies. Then denominator is the total number of patients in the studies.
In the studies that classified patients as either ICU or non-ICU patients, CRP was the most frequently elevated inflammatory marker in both categories (Table 4). LDH was the second most frequently elevated marker in the ICU patients and ferritin was the second most frequently elevated marker in the non-ICU patients. This suggests that CRP levels correlated with the severity of the disease and inflammation when compared to the other inflammatory markers.
Patients Classified as ICU Admission Versus No ICU Admission.

The numerator represents the number of patients with an elevated inflammatory marker based on the criteria used by the individual laboratories in the studies. Then denominator is the total number of patients in the studies.
In the studies that classified patients as either survivors or non-survivors, CRP was the most frequently elevated inflammatory marker in the non-survivors, and ferritin was the most frequently elevated inflammatory marker in the survivors (Table 5). The next highest markers were ferritin in non-survivors and D-dimer in survivors.
Patients Classified as Survivors Versus Non- Survivors.

The numerator represents the number of patients with an elevated inflammatory marker based on the criteria used by the individual laboratories in the study. Then denominator is the total number of patients in the studies.
The most frequently increased inflammatory markers in these 5 categories are summarized in Table 6. CPR was most frequently elevated inflammatory markers in most categories. Ferritin, D-dimer, and ESR were also frequently elevated, and LDH was elevated in ICU patients. In each Tables 1 to 5, the frequency of elevated inflammatory markers was higher in the cohort in the more severe infection category than the comparison category.
Summary of 5 Tables.

The numerator represents the number of patients with an elevated inflammatory marker based on the criteria used by the individual laboratories in the 5 tables. Then denominator is the total number of patients in the studies in individual tables.
Discussion
Coronaviruses are enveloped, positive-sense, single-stranded RNA viruses which can infect several host species and can be divided into 4 genera. SARS-CoV-2 is a β coronavirus that can infect only mammals. The life cycle of virus in the host cells consists of 5 steps in sequence-attachment, penetration, biosynthesis, maturation, and release. 6 SARS-CoV-2 binds to angiotensin converting enzyme 2 (ACE2) using surface spike (S) protein in conjunction with transmembrane serine protease 2 (TMPRSS2) and enters alveolar epithelial cells. The release of chemokines and cytokines then recruits macrophages and dendritic cells into the lung during the early phase of infection. Direct viral infection of pulmonary macrophages and dendritic cells causes expression of several proinflammatory cytokines and chemokines. Then, dendritic cells phagocytose virus in the lungs, migrate to secondary lymphoid organs, and activate antigen-specific T cells which move into the lung and destroy infected alveolar cells. 7
The symptoms in patients infected with SARS-CoV-2 range from minimal/no symptoms to severe respiratory failure with multiple organ failure which can be divided into 2 phases: a physiological host response and pathogenic host response. Several cohort studies have reported markedly elevated levels of circulating proinflammatory cytokines and chemokines, which correlate with disease severity and mortality. A recent meta-analysis suggested that serum IL-6 cut-offs > 55 pg/ml and >80 pg/ml could identify patients at high risk for severe COVID-19 infection and mortality, respectively. 8 However, elevation in tradition biochemical markers of acute infection also occur. The measurement of inflammatory markers can provide important information about the presence of ongoing inflammation. Important considerations include the frequency in which the marker is increased, the magnitude of this increase, and the trajectory during the clinical course. Also, does a particular marker predict specific organ involvement, or the outcome, or response to treatment?
C-reactive protein is a major acute-phase protein with rapid, often within hours, and highly sensitive and specific increases in response to infection or inflammation. This protein recognizes pathogens and facilitates clearance by activating the complement system and phagocytic cells; therefore, CRP is an important component in the first line of innate host defenses, and it helps clear apoptotic and necrotic host cells. 9 In a small study with 27 patients, CRP correlated with CT findings and was significantly increased in the early stage of severe COVID-19 infection, even before changes in the CT score. 10 Moreover, it is it is not influenced by other factors, like gender, age, or pregnancy. In our review, elevated CRP levels occurred in most patients with COVID-19 and were associated with severity of the illness, complications, such as respiratory failure, that required ICU monitoring, and mortality.
The ESR is a marker of long-term inflammation, reflecting the changes in inflammation over the course of several weeks and may be useful for monitoring disease progression in COVID-19 patients over a longer time frame than other markers. However, ESR increases are not specific for inflammation, and, and it is affected by other physiological and pathophysiological conditions, 11 such as anemia since a decreased RBC count can lead to increased ESR. COVID-19 infection can cause anemia in some patients, which might lead to increased ESR levels. Monitoring ESR levels in these patients may help clinicians better understand the course of the disease and possibly provides a laboratory index of the patient’s overall health. In this review, elevated ESR levels occurred in patients with COVID-19 and were more frequently elevated in patients in the pneumonia and mortality categories but not in severity categories. However, Lapić et al. 11 reported a significant difference in ESR levels between severe and non-severe cases based on a pooled analysis of 3 observational studies.
Lactate dehydrogenase (LDH) is an intracellular enzyme found in the cells in almost all organs and has been used as a marker of tissue injury and cell death. Severe infections cause cytokine-mediated tissue damage and LDH release. In addition, LDH-3 (one of the 5 isozymes in humans) is found in pneumocyte and is expected to be elevated in patients with severe COVID-19 lung infections, such as a severe form of interstitial pneumonia or acute respiratory distress syndrome. A recent pooled analysis of 9 studies revealed an association between elevated LDH levels and disease severity and mortality 12 which agree with our findings. In addition, elevated LDH values frequently occur in patients with pneumonia in an ICU. Kojima et al analyzed the clinical factors associated with the progression of COVID-19 pneumonia on computed tomography of the chest. 13 This study reported that LDH was independently associated with the development of severe pneumonia. Thus, monitoring LDH levels may help identify patients at risk for lung involvement and guide management decisions, such as intubation and mechanical ventilation.
Ferritin is an acute phase reactant which can reflect both acute and chronic inflammatory reactions. It is synthesized in monocytes and macrophages in response to alteration in cytokine status and is a key protein in iron metabolism. Its level can indicate changes in iron metabolism that occur during inflammation or infection. Hyperferritinemia develops in patients with COVID-19 infection since many important SARS-CoV-2 regulatory and functional proteins use iron. One hypothesis proposes that SARS-CoV-2 infection interacts with hemoglobin and facilitates iron chelation; however, this is controversial. Two larges systematic review and meta-analysis studies14,15 were done and showed significant associations between elevated ferritin levels and severity of infection, the requirement for intensive care, and mortality.
D-dimer is the soluble plasmin-mediated degradation product of fibrin that reflects activation of coagulation and fibrinolysis. D-dimer levels help diagnose and monitor blood clotting disorders, such as deep vein thrombosis (DVT) and pulmonary embolism (PE). Elevated D-dimer levels may indicate the presence of blood clots or an increased risk of clotting and have been associated with an increased risk of thrombosis in COVID-19 patients. Also, elevated D-dimer levels may indicate increased fibrinolysis, which can occur in bleeding disorders or disseminated intravascular coagulation (DIC). Monitoring D-dimer levels may help identify patients at increased risk of thrombotic events and guide the use of anticoagulant therapy. Elevated D-dimer levels have also been associated with mortality, severity of disease, an increased risk of respiratory failure, especially ARDS, in COVID-19 patients. 16 These studies support the information recovered in our review. Therefore, monitoring D-dimer levels may help identify patients at increased risk of respiratory failure and guide management decisions, such as early initiation of mechanical ventilation and should increase concern about the possibility of DVT and the need for anticoagulation.
Fibrinogen is an acute phase protein synthesized by liver in response to IL-1 and IL-6 stimulation; it is also involved in fibrin formation during coagulation. Dynamic changes in fibrinogen occur in patients with COVID-19, and its level is higher in COVID-19 patients, especially in critically ill patients. However, it might not have a predictive value for mortality rate. 16 Fibrinogen levels should be measured with D-dimer to diagnose DIC.
Procalcitonin is a glycoprotein calcitonin pro-hormone released by thyroid parafollicular cells. Procalcitonin levels are significantly increased in patients with microbial infection since it is released by all parenchymal tissue under influence of endotoxins and pro-inflammatory cytokines. However, cytokines released in COVID-19, particularly interferon (INF)-γ, have a negative effect on PCT levels. 17 COVID-19 patients are at increased risk of developing bacterial superinfections, which can worsen their clinical outcomes, and PCT levels can help clinicians identify bacterial superinfections in COVID-19 patients and guide appropriate antibiotic therapy. Our study demonstrated a significant association between elevated procalcitonin and disease severity, intensive care admission, and mortality which could reflect superimposed bacterial infection. PCT levels are not associated with uncomplicated COVID-19 infections.
The COVID-19 pandemic significantly increased demands on all aspects of healthcare systems. These demands included more ICU beds, more mechanical ventilators, more medications, more laboratory testing, and more health care workers. This review of literature published in 2020 demonstrates the clinicians used several inflammatory markers to evaluate and monitor hospitalized patients with COVID-19 infection. Durant et al analyzed the effect of COVID-19 testing on laboratory utilization between February 2, 2020 and April 11, 2020. 18 In their hospital there was a significant increase in workload which included send out laboratory testing, COVID-19 diagnostic testing, and patient management-related testing. There was also an increase in point-of-care testing. These tests significantly increased costs; the Office of Inspector General in United States reported that laboratory cost paid for by Medicare Part B increased from $8 billion in 2020 to $9.3 billion in 2021. 19 The costs in 2021 included $2.0 billion for COVID-19 tests, $1.9 billion for genetic tests, and $5.5 million for the top 5 laboratory tests, including CBC panels and chemistry panels. The overuse of laboratory testing could place unusual demands on laboratories, including testing reagents and personal time.
Emanuel et al discussed the fair allocation of medical resources during the COVID-19 pandemic in 2020 based on ethical principles. 20 These authors made 6 recommendations which addressed the multiple aspects of managing patients during this pandemic but did not make specific recommendations regarding laboratory testing. Abdul-Moheeth et al and coauthors reported the outcome of a program designed to reduce laboratory testing in noncritically ill patients with COVID-19 infection. 21 They analyzed the mean daily testing of chemistry panels, CBCs, D-dimer levels, and troponin levels and then developed a customized order set in the EMR. This effort resulted in a significant reduction in daily laboratory testing. Shin et al analyzed the order patterns for C-reactive protein, ferritin, lactate dehydrogenase, and procalcitonin and developed an electronic health record intervention which included both information and a restriction which prevented multiple day orders. 22 They reported a significant decrease in the order frequency of these laboratory tests following the intervention. These 2 studies demonstrated that an organized effort which includes education and order controls in electronic medical record ordering sets can reduce the use of laboratory tests in these patients with no effect on outcomes. In retrospect, laboratory managers and clinicians should have frequent meetings to create reasonable test guidelines.
The study design used in this project has definite limitations. This study analyzed the frequency of elevated inflammatory markers in patients with COVID 19 infection classified into broad categories. It did not analyze trends in inflammatory markers, the use of a particular marker to diagnose specific complications, or changes in levels associated with treatment. These considerations will require prospective studies of well-defined cohorts of patients.
Conclusion
Early in this pandemic it became apparent the patients had a range of presentations, including severe infection requiring ICU care and mechanical ventilation. Clinicians used routine laboratory tests for inflammatory markers to evaluate the severity of the infection and possibly predict outcomes. This review of articles published in 2020 during the first year of this pandemic indicates that CRP levels were frequently elevated, especially in critically ill patients. The measurement of this inflammatory marker alone likely provides reasonable information about the severity of infection and the potential for significant complications and/or mortality. LDH levels potentially identify patients with significant pulmonary involvement. D-dimer levels are sensitive but not specific markers for the presence of deep venous thrombosis and/or pulmonary emboli but are potentially useful in managing outpatients with established thromboembolic disease. Future studies should be designed to ask specific questions regarding the relationship between inflammatory marker levels and COVID-19 infections. Can inflammatory marker trends predict responses to treatment, predict complications, or predict clinical deterioration. These studies will require the development of protocols involving homogeneous cohorts if possible with the selection of specific markers and standard collection timeframes.
Supplemental Material
sj-docx-1-jpc-10.1177_21501319231206911 – Supplemental material for COVID-19 Infections and Inflammatory Markers in Patients Hospitalized During the First Year of the Pandemic
Supplemental material, sj-docx-1-jpc-10.1177_21501319231206911 for COVID-19 Infections and Inflammatory Markers in Patients Hospitalized During the First Year of the Pandemic by Parkavi Kalaiselvan, Pitchaporn Yingchoncharoen, Jerapas Thongpiya, Arunee Motes and Kenneth Nugent in Journal of Primary Care & Community Health
Footnotes
Acknowledgements
None
Author Contributions
PK—conceptualization, literature review, original draft preparation, draft revision, and approval of the final version. PY—literature review, original draft preparation, manuscript revision, and approval of final version. JT—literature review, original draft preparation, approval of final version. AM—literature review, manuscript editing and review, approval of final version. KN—conceptualization, literature review, manuscript drafting, manuscript revision, approval of final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability
All information used in this manuscript is available in the references.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
