Abstract
Introduction/Objectives:
Annually, HPV infections result in $775 million in direct medical costs and approximately 46 000 new cases of HPV-associated cancers. Safe and highly effective vaccines have been available to prevent HPV for children/adolescents since 2006. Vaccination rates remain low, especially in rural areas. Parental attitudes and beliefs affect HPV vaccination rates.
Methods:
We developed, tested, and administered a survey that asked how parents and healthcare providers interacted about the HPV vaccine following a healthcare visit with an age-eligible child, as part of a multicomponent randomized controlled trial designed to improve HPV vaccination rates in rural Oregon. The 21-item survey assessed parents’ information-seeking behavior, knowledge about HPV cancer risk reduction, the HPV vaccine series, and their vaccine confidence.
Results:
Forty-three participants (59.7%) were in the intervention group; 29 (40.3%) were controls. Over 90% of healthcare visits were illness, injury, sports physical, or well-child visits (n = 67 or 93.1%), and 6.9% of visits were vaccine-specific. No statistically significant differences were found between study groups for healthcare visits. Over half the parents reported having discussions about HPV and the HPV vaccine (54.5%) with their care providers, 31.3% had recently learned about HPV, HPV risks, and the HPV vaccine prior to the visit, 83.1% were knowledgeable about cancers associated with HPV, and 79.2% were considering vaccinating their child(ren), which did not differ between study groups.
Conclusions:
Knowledge about HPV-related cancers and consideration for vaccinating children was higher than expected, but not associated with the intervention tested.
Introduction
The human papilloma virus (HPV), which is transmitted through genital and skin-to-skin contact, 1 is the most common sexually transmitted infection in the United States (US), with nearly 14 million infections occurring each year. 2 Annually in the US, HPV infections result in $775 million in direct medical costs and approximately 46 000 new cases of HPV-associated cancers. 3 These cancers cause 6100 deaths in the US each year. 4
Since 2006, safe and highly effective vaccines have been available to prevent HPV. 5 These vaccines work best when given to adolescents aged 9 to 12 years, due to a more robust immune response at a younger age; however, they can be given to people of any gender, up to age 45 years.6-8 Approximately 92% of HPV-attributable cancers can be prevented by HPV vaccination each year. Despite being available for over 15 years, the uptake of the vaccine remains low. The Healthy People 2030 goal for HPV vaccination is 80%, yet the 2020 National Immunization Survey findings reveal that only 61.4% of females and 56.0% of males in the US are fully vaccinated for HPV.7,9 Further, HPV vaccination rates for rural adolescents remain significantly lower than for their urban peers. 9 In Oregon, 2020 vaccine data show that only 55% of adolescents aged 13 to 17 years were fully vaccinated for HPV. 10
Parents’ attitudes and beliefs affect HPV vaccination rates.11-13 Parents commonly cite safety factors, side effects, and/or the belief that the vaccine is unnecessary as reasons for choosing not to vaccinate their children against HPV.11,14,15 Additional factors that affect parental hesitancy include stigma around vaccinations, lack of knowledge about HPV vaccination, and beliefs that children could become sterile due to HPV vaccination. 16 Among parents in rural settings, religiosity and spirituality, and an even greater lack of knowledge about HPV vaccination affect their vaccination decisions.16,17
Several studies have aimed to improve HPV vaccination rates by addressing knowledge and attitudes among parents.18,19 Clinic-based interventions, such as the use of health information technology tools, 20 reminder letters, standing orders, 21 and strong provider recommendations 22 have been found to increase HPV vaccination rates. Additionally, school-based interventions19,23 and social marketing campaigns24,25 have resulted in increased vaccination rates at the community level. What is less well understood is how partnerships between clinic and community groups could affect parental knowledge and attitudes about HPV vaccinations.19,26-30
We conducted a 5-year stepped-wedge randomized controlled trial that aimed to increase HPV vaccination rates among rural Oregon adolescents by engaging both primary care clinics and community organizations in the development and implementation of interventions to increase HPV vaccination. 31 In this manuscript, we specifically describe the results from a survey of parents—following their child’s healthcare visit—to understand their information-seeking behavior and knowledge about cancer risk reduction with HPV vaccination and whether our intervention appears to influence these factors.
Methods
The Rural Adolescent Vaccine Enterprise (RAVE) study was conducted with the Oregon Rural Practice-based Research Network, a statewide network of primary care clinicians, community partners, and academicians dedicated to studying the delivery of health care, improving the health of Oregonians, and reducing rural health disparities. Family medicine and pediatric clinics enrolled in RAVE were based in rural communities with populations of less than 50 000. 31
RAVE included an intervention that partnered each primary care clinic with a practice facilitator for 18 months, over which time they utilized quality improvement methodology to work toward improving HPV vaccination. 31 Many clinics focused on strategies to identify vaccine-eligible patients, conduct reminders/recalls for follow-up vaccination visits, and improve provider communication around vaccination. For the last 12 months of the 18-month intervention, clinics were tasked with identifying and collaborating with a community partner to conduct a social marketing campaign. 31 Clinics collaborated with diverse community-based organizations, such as libraries, schools, coffee shops, and local public health departments. Findings regarding these partnerships are reported elsewhere. 32
We developed a 21-item survey to assess associations between the clinic and community partnership intervention and rural parents’ information-seeking behavior, knowledge about HPV cancer risk reduction, and the HPV vaccine series. The survey was designed to be completed within 48 h of a healthcare visit. Questions ascertained: (1) the reason for the healthcare visit; (2) whether there was any discussion of the HPV vaccine; (3) the information seeking behavior of the parent with regards to the HPV vaccine; (4) and parent knowledge and attitudes about the HPV vaccine and cancers it can help to prevent, including the 8-item Vaccination Confidence Scale developed by Gilkey et al, 33 which measures the benefits and harms of vaccinations, as well as trust in healthcare providers. 33 The survey also assessed whether parents had considered getting their child vaccinated for HPV (including if their child had already initiated or completed the series), parents’ comfort level when asking about the vaccine with their child’s provider and office staff, and demographic information for both the parent and child.
Author PAC conducted 5 rounds of survey pilot tests using cognitive interviewing techniques with parents of age-eligible children, initially with those who did not have a recent healthcare visit to assess general understandability, and then with parents who had just taken their child to a healthcare visit at a family medicine clinic in rural Oregon. 34 The survey was revised after each round of pilot testing to ensure questions were understandable by participants and the order of questions was not leading them to respond in biased ways. The final version of the survey included 21 items and took parents approximately 10 min to complete.
Between September 2020 and January 2021, 27 RAVE clinics participated in parent survey distribution, including identifying the names and addresses of parents of 30 adolescents who were age-eligible for the HPV vaccine and had a clinic visit within the prior 48 h. The study team or the clinic’s management then mailed these parents a survey which included a standardized letter from the clinic and a pre-paid return envelope. The RAVE team used REDCap (Research Electronic Data Capture) to enter survey data and a study team member verified data entry accuracy.35,36
The COVID-19 pandemic occurred during the study period, yet the study team decided to proceed with as many parent surveys as could be captured. Approximately 497 surveys were sent to parents of age-eligible adolescents. Seventy-eight surveys were returned (15.7% response rate) and 6 surveys were subsequently excluded (1 blank and 5 included minimal data), leaving 72 surveys included in the analyses.
For analyses, parent survey questions were organized into 3 sections: (1) Recent Healthcare Visit, (2) Vaccination Confidence Scale, and (3) Patient Demographics. Relevant healthcare visit and demographic items were tabulated by counts and percentages and continuous variables were summarized by their mean and standard deviation. Differences in categorical items were assessed by Chi-Square tests and continuous variables were assessed using t-tests.
We calculated mean scores and standard deviations for the Benefits, Harms, and Trust factors as well as for each individual item from the Vaccination Confidence Scale. The 2 variables that reflect the Harms factors were reverse scored. Higher scores indicate positive attitudes toward vaccinations, whereas lower scores indicate negative attitudes about them. Lastly, an overall mean and standard deviation were calculated for all 8 items. For this exploratory analysis, we grouped data according to whether they had received a study intervention (Group 1) or not (Group 2).
Univariate models of the association between the overall mean score of the Vaccination Confidence Scale and relevant healthcare visit and demographics were analyzed and stratified according to study group. We calculated group means, standard deviations, and standard errors. We used Tukey’s test to assess differences between study groups and report P-values for each variable. Alpha levels for identifying statistically significant findings were set at <.05.
Results
A total of 72 participants completed the survey. Intervention clinics included 43 participants (59.7%), and controls included 29 (40.2%). The patients averaged 13.4 years of age and over half were female (56.9%), white, and non-Hispanic (77.8%; Table 1). Parents were represented primarily by mothers (81.9%) over 45 years of age (45.3%), and over half had a college degree or more (56.9%; Table 1). There were no statistical differences between characteristics of children and parents in the 2 study groups. Over 90% of healthcare visits were illness, injury, sports physical, or well-child visits (n = 67 or 93.1%; Table 1). Over half the parents reported having discussions about HPV and the HPV vaccine (54.5%), 31.3% had recently learned about HPV, HPV risks, and the HPV vaccine prior to the visit, 83.1% were knowledgeable about cancers associated with HPV, and 79.2% were either considering the HPV vaccine for their child(ren) or had already initiated or completed the vaccine series (Table 1).
Child and Parent Demographic and Health Care Visit Findings According to Study Group.

Vaccination Confidence Scores for individual items ranged from 8.2 to 9.9. The only statistical difference among the individual items between study groups was for “If I do not vaccinate my older child or teenager, he/she may get a disease such as meningitis or human papilloma virus and cause other teenagers or adults to also get the disease” which was 8.0 in the intervention group and 9.9 in the control group (P = .01). When the factor structures for vaccine Benefits, Harms, and Trust were analyzed, the Trust factor scored highest overall (Mean = 9.6, SD = 2.3) and the Harms factor scored lowest (Mean = 8.5, SD = 3.4; Table 2). We found no statistically significant differences according to study group for vaccine confidence factor structures.
Vaccine Confidence Findings According to Study Group.
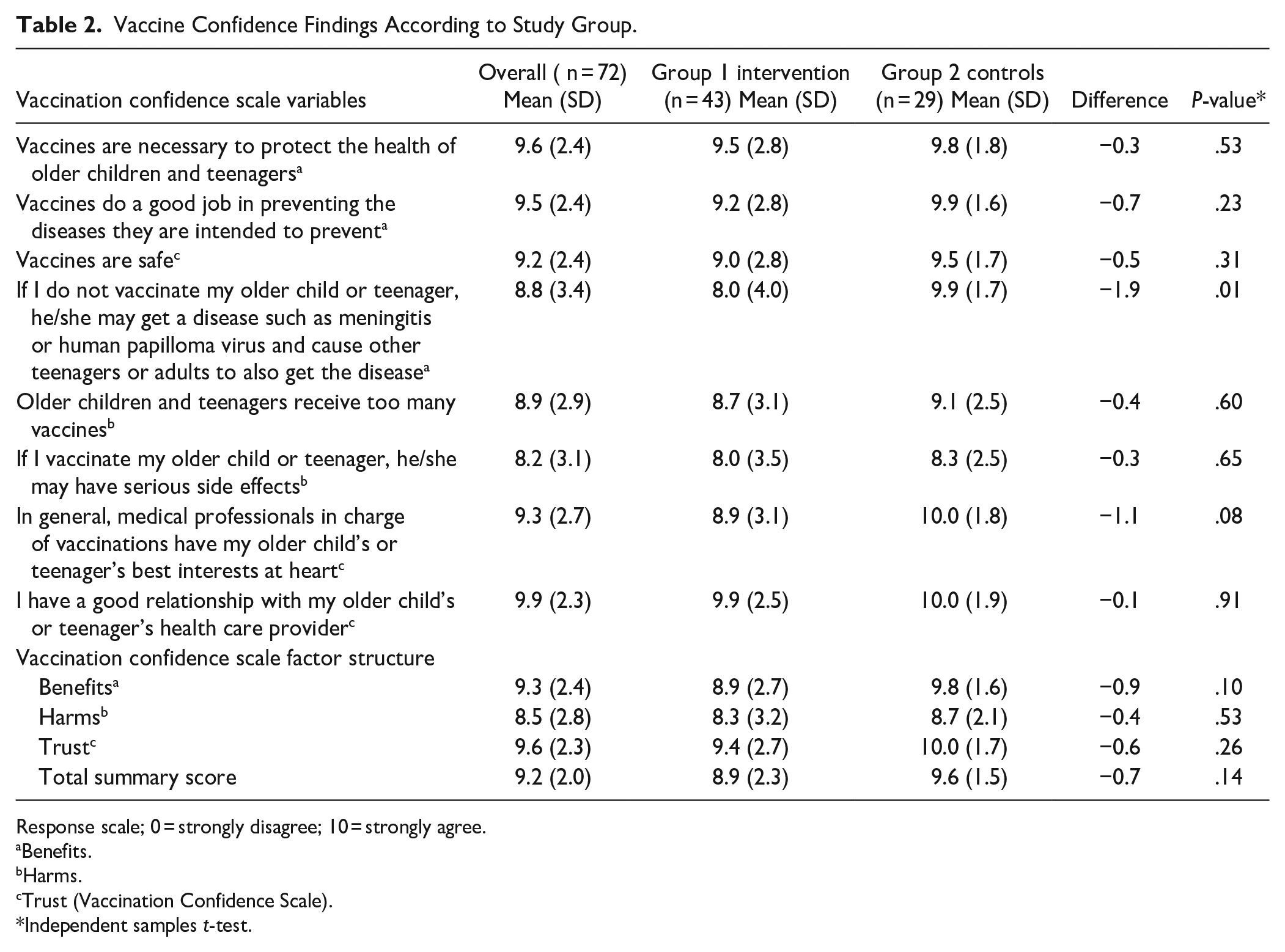
Response scale; 0 = strongly disagree; 10 = strongly agree.
Benefits.
Harms.
Trust (Vaccination Confidence Scale).
Independent samples t-test.
Univariate associations between the overall mean of the Vaccination Confidence Scale and demographics and healthcare visit questions showed no statistical differences in scores between the intervention and control groups (Table 3). Vaccine confidence scores ranged from 6.7 for race/ethnicity to 11.0 among those who were Hispanic in the intervention group, and from 5.6 for fathers to 10.0 among mothers, non-Hispanic whites, and those with a college degree in the control group. Of the 25 variables and their respective levels, differences in survey scores were primarily between –1.0 and 1.0, whereas the remaining differences in survey scores were greater than –1.0 or 1.0, indicating minimal differences.
Estimated Mean Vaccination Confidence Scale Score According to Demographics Healthcare Visit and Study Group.

Discussion
We conducted this study to determine what occurs between healthcare providers and parents of children age-eligible to receive the HPV vaccine during a healthcare visit, what level of knowledge parents have about HPV and the HPV vaccine and its preventive benefits, and to explore whether this was affected by our intervention. To our knowledge, this is the first study to assess HPV-related conversations that occurred during healthcare visits in rural clinics. Findings suggest that more than half the parents, primarily mothers, discussed HPV during the visit and that the majority of participants reported being knowledgeable about cancers associated with HPV and many of these were considering having their child be vaccinated.
This study was unavoidably impacted by the COVID-19 pandemic in a variety of ways. First, the response rate was low, likely because the timing of the survey coincided with school closures and stay-at-home orders and parents may have been simply too busy or distracted to respond to a survey. Second, many parents were out of work during this phase of the pandemic, causing unforeseen stressors in rural communities that are already under-resourced. Third, conducting a study on vaccines during a pandemic while awaiting emergency use of a vaccine for COVID-19 likely led to response bias, where those who responded to the survey may have had unique ideas, either positive or negative, about vaccines. Fourth, the pandemic stressed the clinics and community organizations participating in the study, and many of them chose to pause their work with the RAVE study to allocate their time and resources elsewhere during the pandemic. Other studies have reported on smaller response rates during the COVID-19 Pandemic.37,38 Krieger et al 37 reviewed survey completion rates from 5 national surveys, conducted during COVID. They showed a 29% lower response rate during 2020 compared to 2019. 37 Another study by de Koning et al 38 cite survey fatigue as a reason for a lessened response rate among neurosurgical-specific surveys issued since the start of the COVID-19 pandemic. Through their review of 255 articles, they found that more neurosurgical surveys were issued during the pandemic—leading to more total survey responses, however, overall survey response rates decreased to 34.5% from 51% pre-pandemic. 38 It would have been helpful to know the characteristics of responders and non-responders, which would have allowed us understand potential biases among those who did complete the survey relative to those who did not.
Regardless of challenges with our study, this work helped us understand the nuts and bolts of working with clinics to undertake a survey of parents designed to assess what occurs during a recent healthcare visit. Such surveys are increasingly being used to study preventive and non-urgent healthcare visits for children and adolescents. One 2021 survey examined sexual and reproductive health discussions that occurred with adolescents, their parents, and healthcare providers within 2 years of a healthcare visit. 39 Another study examined parental use of emergency rooms for non-urgent care issues, which used key informant interviews immediately after care was received to collect data. 40 Our study is unique in that we surveyed parents within days of their healthcare visit to mitigate the influence of recall bias that can be introduced when participants do not remember prior events or omit important details.
Our findings about parental knowledge and attitudes revealed that more than half the parents discussed HPV during the visit and the majority of participants reported being knowledgeable about cancers associated with HPV, with many considering having their child be vaccinated. These findings are similar to a 2016 study which found the top sources of information about the HPV vaccine reported by parents were a doctor/medical professional at 80% and 64% was television. 41 Importantly, this study found that sources of information about the HPV vaccine are associated with parental attitudes about the HPV vaccine, which are associated with vaccine uptake among adolescents, further underscoring the need for impactful communication between healthcare providers and parents.
We found no statistical differences for knowledge and vaccine confidence factors between the control and intervention groups. The vaccine Benefit variable “If I do not vaccinate my older child or teenager, he/she may get a disease such as meningitis or human papilloma virus and cause other teenagers or adults to also get the disease” was statistically higher in the control group compared to the intervention group, indicating higher agreement with the statement in the control group. This was an unexpected finding that suggests that those parents visiting healthcare providers unexposed to an intervention designed to increase HPV vaccination during the pandemic were more aware of disease spread compared to parents seeing providers in the intervention group. This may be a spurious finding that resulted from our small sample size.
Conclusions
Despite this small exploratory finding that parents and providers discussed the HPV vaccine in more than half of the visits as well as that parents were knowledgeable about the vaccine and the cancers it prevents, it also raises many questions. For example, it may be that the rapid COVID-19 vaccine development and administration under emergency use laws, affects confidence in vaccines overall. Vaccine hesitancy does appear to have increased during the COVID-19 pandemic, and more research is needed to understand how beliefs about hesitancy form and what causes these to change over time.42,43 It may be that several different interventions need to occur in multiple settings to overcome hesitancy, not only to the HPV vaccine but other vaccines as well.
In addition to high vaccine hesitancy rates for the HPV vaccine among parents, rates of vaccine hesitancy are high for vaccination overall and may be increasing.44,45 For instance, Alimoradi et al 46 reviewed 98 peer-reviewed papers from nearly 70 countries in order to understand parental acceptance to the COVID-19 vaccine. Their findings show a low prevalence of such acceptance, with a pooled estimated prevalence of 57% (far lower than what may be necessary to reach herd immunity for COVID-19). In 2019, Kempe et al 47 modified the Vaccine Hesitancy Scale in order to test for vaccine hesitancy among U.S. parents. Survey results from 2052 eligible respondents indicated that 6.1% of parents were hesitant about routine childhood vaccination and 25.8% were specifically hesitant about influenza vaccination. 47 Further, reviewing data from a vaccine hesitancy module in the 2018 and 2019 National Immunization Survey-Flu, Santibanez et al 48 found that in 2018, 25.8% of U.S. children ages 6 months through 17 years had a parent who expressed hesitancy about childhood vaccinations.
As noted through our work, most parents accompanying patients to visits were mothers. However, we found lower vaccine confidence rates among fathers. While beyond the scope of the current study, it may warrant future research to understand how paternal influence affects vaccine delivery, especially for HPV vaccination. Our low response rate produced a small sample that included only 29 participants in the control group and 43 in the intervention group, which is an inadequate sample size to fully test our interventions. Regardless, we believe it is clear that more work needs to be done in rural communities to help influence HPV vaccination rates, which has implications for receipt of vaccines for other diseases as well. Additionally, more research is needed on how vaccines are discussed in the context of wellness and illness healthcare visits, given the existing literature that shows how influential healthcare providers are when they recommend vaccines for patients.
Footnotes
Acknowledgements
The RAVE team is grateful to the clinics that enrolled in the study. The participating clinics worked hard to improve adolescent HPV vaccination rates during a very tumultuous time in medical history. We also extend appreciation to the parents who completed the RAVE parent survey.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the American Cancer Society [RSG-18-022-01-CPPB].
Prior Presentations
None.
Clinical Trial Registration
NCT03604393.
