Abstract
There are few estimates of the seroprevalence of SARS-CoV-2 antibodies among children in the United States. We measured vaccine and infection induced seroprevalence among nearly 5000 healthy 1 to 17-year-old children in Colorado from 2020 to 2021. By December 2021, 89% of older children, ages 12 to 18, had antibodies detected. The increase was largely driven from vaccination rather than infection.
Introduction
COVID-19 disease, caused by the SARS-CoV-2 virus, results in considerable morbidity among children. Individual and herd immunity are critical to reducing the disease burden among children and preventing transmission to adults. There are few reliable estimates of the seroprevalence of SARS-CoV-2 antibodies among the U.S. pediatric population. We aimed to determine the seroprevalence of vaccine and infection induced SARS-CoV-2 antibodies from 2020 to 2021 among nearly 5000 healthy 1 to 17-year-old children in Colorado, USA and to understand if the change in seroprevalence was driven by natural infection versus vaccination.
Methods
Antibodies to the SARS-CoV-2 were tested in 4717 general population Colorado children aged 1 to 17-years-old participating in the Autoimmunity Screening for Kids (ASK) type 1 diabetes screening study1,2 between July 2020 and December 2021. Children were recruited primarily from outpatient clinics in the context of well child visits. Because the primary goal of recruitment was to detect autoantibodies for type 1 diabetes, children were not recruited based on history of COVID-19 disease or vaccination status. Ill children, including those with SARS-CoV-2 infection in the prior 2 weeks, were not enrolled to protect study personnel. History of prior COVID-19 diagnoses and SARS-CoV-2 vaccination history was obtained from parents. Antibodies to the SARS-CoV-2 receptor-binding domain (RBD) of the spike protein were measured for all children. RBD antibodies are produced after SARS-CoV-2 infection or vaccination. Thus, to evaluate if vaccinated children had also had a prior SARS-CoV-2 infection, we measured antibodies to SARS-CoV-2 nucleocapsid protein (NP) in children that had received at least one dose of the SARS-CoV-2 vaccination. NP antibodies are produced after SARS-CoV-2 infection, but not after vaccination. 3 Detection thresholds for positivity were assessed using the World Health Organization international standard (NIBSC code 20/136, Supplemental Material). Past SARS-CoV-2 infection was defined by the presence of antibodies to SARS-CoV-2 RBD and NP measured by electrochemiluminescent (ECL) assay. An RBD antibody index >5 defined past SARS-CoV-2 infection in an unvaccinated child. In children who had received a SARS-CoV-2 vaccine, an NP antibody index >5 defined past SARS-CoV2 infection. Seroprevalences by time, age group, and antibody type were computed. Demographics and vaccination status were compared between children who did and did not have prior infection using Pearson’s chi-squared tests. Two-tailed P-values <.05 were considered significant. The Colorado Multiple Institutional Review Board approved this research and informed consent was obtained from parents of each study participant.
Results
The Table 1 shows demographic features and vaccination status of included children. Of the 4717 children included, 1524 (32.3%) had serologic evidence of prior SARS-CoV-2 infection, though only 359 (7.9%) reported prior infection. Infection was more frequent in older children (P ≤ .001), Hispanics (P < .0001), and unvaccinated children (P = .0001). The prevalence of SARS-CoV-2 RBD antibodies increased from 0% in July 2020 to 70% (95% CI 63%-76%) in December 2021. Monthly age-specific prevalence estimates during the 18-month study period by age and vaccination status are shown in the Figure 1. By December 2021, the seroprevalence of SARS-CoV-2 antibodies induced by infection and/or vaccination in children aged 1 to 4, 5 to 11, and 12 to 18 years had reached, respectively, 46% (95% CI 32%-61%), 74% (95% CI 64%-81%), and 89% (95% CI 77%-96%). The increase in the prevalence of potentially protective antibodies was driven primarily by vaccination, rather than infections.
Baseline Characteristics of the Study Population, No. (%).

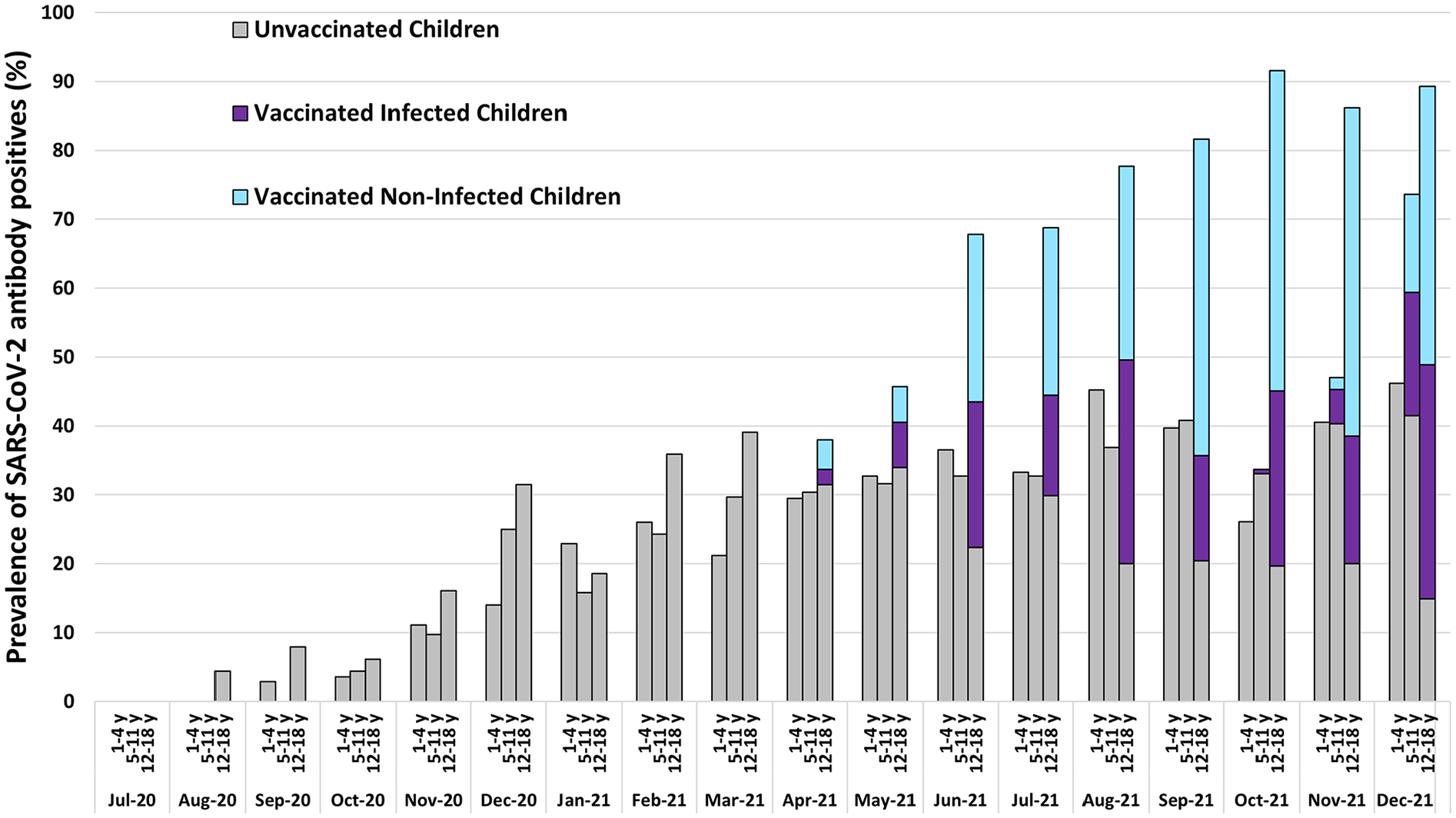
Prevalence (%) of SARS-CoV-2 receptor binding domain (RBD) antibodies in Colorado children (n = 4717) by age, July 2020 to December 2021. Combined prevalence of the antibodies induced by infections and vaccinations may serve as an estimate of herd immunity to SARSCoV-2.
Discussion
The prevalence of antibodies to SARS-CoV-2 among healthy U.S. children may be higher than previously estimated. Prior evaluations that have aimed to assess the seroprevalence of SARS-CoV-2 antibodies have focused predominantly on estimates based on infection-fatality rates 4 or serologic evidence of prior infection (45%-74%) 5 which likely underestimated antibodies generated from asymptomatic infection and/or vaccination, respectively. Vaccination played a critical role in increasing the generation of potentially protective antibodies. Importantly, neither antibodies from infection nor vaccination are fully protective against infections as they may confer incomplete protection against new variants (eg, omicron), protection may wane over time, and protection may depend on other host immune characteristics and viral load. We were limited in that we did not measure neutralizing antibodies that may serve as a biomarker of immunity. Thus, the herd immunity may be lower than that predicted by the seroprevalence. Though we assessed antibodies generated from both infection and vaccination, vaccination is the strongly preferred mechanism to achieve adequate herd immunity because antibodies from vaccination persist longer 6 and vaccination avoids obvious complications from gaining immunity by infection and reduces community transmission of virus. Future studies should assess the longevity of immunity from vaccination and herd immunity among children as they become vaccinated.
Supplemental Material
sj-docx-1-jpc-10.1177_21501319231189147 – Supplemental material for Prevalence of SARS-CoV-2 Antibodies Among Healthy Children From Colorado From 2020 to 2021: A Brief Report
Supplemental material, sj-docx-1-jpc-10.1177_21501319231189147 for Prevalence of SARS-CoV-2 Antibodies Among Healthy Children From Colorado From 2020 to 2021: A Brief Report by Holly M. Frost, Cristy Geno Rasmussen, Hanan Shorrosh, Laura Pyle, Kimberly Bautista, Brigitte I. Frohnert, Marisa Stahl, Kimber Simmons, Andrea K. Steck, Xiaofan Jia, Liping Yu and Marian Rewers in Journal of Primary Care & Community Health
Footnotes
Abbreviations
RBD-receptor-binding domain
NP-nucleocapsid protein
Author Contributions
Dr. Rewers conceptualized and designed the study, drafted the manuscript, completed statistical analyses and obtained funding. Drs. Rasmussen, Shorrosh, Jia, and Yu acquired, analyzed, and interpreted data and critically reviewed the manuscript. Dr. Frost interpreted data and drafted the manuscript. Dr. Pyle completed the statistical analyses, interpreted data, and critically reviewed the manuscript. Drs. Frohnert and Simmons interpreted data and critically reviewed the manuscript. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work (the ASK Study [3-SRA-2018-564-M-N]) is funded by the Juvenile Diabetes Research Foundation (JDRF) International, The Leona M. and Harry B. Helmsley Charitable Trust, and Janssen Research and Development, LLC. Additional support came from the Courtnay C. and Lucy Patten Davis Foundation and the National Institutes of Health (NIH), National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) University of Colorado Diabetes Research Center grant [DK116073]. H.F received salary support from the Eunice Kennedy Shriver National Institute of Child Health & Human Development of the NIH [K23HD099925]. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
