Abstract
This quality improvement (QI) project provided Hepatitis C virus (HCV) treatment at a Federally Qualified Health Clinic (FQHC) for persons who use illicit drugs. Many of these individuals sought treatment at the local Infectious Disease (ID) clinic but were denied care as they require a patient be drug-free for 6 months prior to HCV treatment. These individuals expressed a desire to be cured of HCV, which can lead to liver failure or cirrhosis if left untreated. This project bridged the existing gap in HCV treatment that currently exists for substance-users in this city. Pre-treatment HCV levels were obtained from 20 participants who completed an 8-week daily regimen of Mavyret, a direct-acting antiviral (DAA) agent, prescribed by a primary care Nurse Practitioner (NP), trained to treat HCV. Pre-treatment HCV loads were compared to the sustained viral load, obtained 12 weeks post-treatment (SVR-12), which is considered the “test of cure.” The results indicate that 100% of returning patients were considered cured of HCV. This program successfully integrated HCV treatment at a community health center, in a population affected by substance use. Adoption of similar programs in primary care clinics can help meet the clinical needs of this often stigmatized and vulnerable population as well as cure them of HCV.
Keywords
Hepatitis C virus (HCV) is known as the silent epidemic as many individuals living with HCV are asymptomatic. 1 The population of people who inject drugs (PWID) has been identified as the largest group of those with HCV infections. 2 Active injection use in a positive HCV individual is an assured means of HCV community transmission. Therefore, treating an HCV-positive individual has an undisputed public health benefit in reducing HCV secondary transmission, especially in those actively injecting drugs. 3 Challenges exist with HCV education, treatment, and management of this high-risk population, especially in vulnerable and underserved communities. Furthermore, there are stigma and negative perceptions of PWID. In 1 city in central Virginia, the local Infectious Disease (ID) clinic specialists will not treat HCV in patients who submit a positive urine drug screen (UDS). Instead of treating locally, these individuals are referred over an hour away to a university ID specialty clinic. This quality improvement project will address this gap in care for PWID and provide expanded treatment at a local FQHC. Barriers such as unreliable transportation, lack of insurance, housing, food insecurity, addiction, and mental health disorders can also be addressed by the FQHC staff. Specifically targeting the subpopulation of positive HCV individuals who use drugs can greatly decrease the spread of HCV in this community as well as provide multidisciplinary, holistic services to PWID.
Background and Significance
Hepatitis C viral infection (HCV) has quietly become a public health concern in the past 2 decades, increasing its incidence among PWID. 3 Effective antiviral treatment provides the cure for HCV; however, millions of people remain infected. HCV-positive patients have not been able to take advantage of these new antiviral regimens due to patient, provider, and/or system barriers. 3 In 1 region of central Virginia, the local Infectious Disease (ID) specialists will not treat HCV patients who test positive on a UDS. Hepatitis C guidelines have repeatedly stated that active or recent drug use or concern for reinfection is not a contraindication to HCV treatment. 3 Therefore, a large segment of the drug-using population is continuing to spread HCV in this community. Research has shown that properly educated primary care providers can effectively treat HCV-positive patients in their community without the need to refer them to specialty clinics farther from their homes.3 -5
Problem and Purpose Statements
Patients are unable to successfully be linked to HCV treatment, particularly for PWID or those who use drugs, due to the current referral process in place in this community. The key to enhancing linkage to care, as well as treatment retention is evident, especially for vulnerable populations, as they should be treated locally in primary care settings. 4 The overall purpose of this project was to invite PWID or who use drugs to be treated for HCV in their community, at a clinic located on the bus route, as opposed to being referred a much longer distance for specialist care. A nurse practitioner was empowered to provide HCV comprehensive education/treatment for the patient as well as the family. The patient and family were given opportunities for linkage to primary care services, mental health treatment, stable housing, clothing closets, food pantries, Medicaid application assistance, and pharmacy services located adjacent to the clinic. This integrated, multidisciplinary team approach had the possibility to enhance the patients’ overall quality of life as well as treat their HCV.
Literature Review Methodology
It was important to limit the research to the past 5 years as this is when the single daily dose anti-viral therapy was approved for use instead of complex, multiple dosing regimens that had numerous adverse side effects. The newer direct-acting antivirals (DAAs) revolutionized HCV treatment by demonstrating high efficacy, low toxicity, and minimal side effects and were approved by the Federal Drug Administration (FDA) 6 at the end of 2015.
Literature Review Summary
Substance users who have HCV may present with compounding issues including mental health disorders, alcohol abuse, and lack of stable housing/transportation which can be perceived as risks for non-adherence to DAA treatment modalities. 7 Several robust research studies have shown that HCV treatment programs can successfully be integrated into local primary care community health centers that use a multi-disciplinary team approach and focus efforts on HCV patients with high rates of substance abuse and mental illness.5,7 -10
Results also showed cost-savings for HCV treatment in primary care at close to half the cost of specialty hospital-based treatment which is a substantial saving for stakeholders. 11 The new DAA medications are proven effective and well-tolerated with few side effects and demonstrate a cure rate of 95%, which increases adherence to therapy. 9 Research show that patients express positive outcomes during and after HCV treatment such as increased energy, improved physical health, decreased feelings of shame in possibly spreading HCV, and a sense of optimism for the future. 5
Project Methods
Implementation of the project followed the guidelines and recommendations from the American Association for the Study of Liver Diseases-Infectious Diseases Society of America. 3 This QI project was viewed as a delivery model change in that uncomplicated positive hepatitis C virus (HCV) patients with a positive urine drug screen (UDS) will now have the option to receive treatment in the local community. If patients were identified as complicated per AASLD/IDSA guidelines, they were referred to the university ID clinic and eligible for telemedicine visits, facilitated by the project manager.
Project Setting
The project took place in a central Virginia Federally Qualified Health Center (FQHC), located in the downtown area of the city. The community clinic is a multidisciplinary center that focuses on primary care as well as mental health. Lab services were provided by the clinic.
Participants
The initial cohort included 25 participants >18 years of age, testing positive for active HCV and positive on a UDS. The FQHC electronic medical record (EMR) was reviewed to identify individuals meeting this criterion and seen in the last 2 years. Due to the population characteristics of this project, it was submitted and approved by the hospital IRB. Participants were offered to be treated for HCV locally, at the clinic, for 8 consecutive weeks. Mavyret is the brand name of the generic formulation known as GLE/PIB (glecaprevir/pibrentasvir), a viable direct antiviral agent (DAA)-approved therapy for HCV and chosen as the DAA prescribed for this project as treatment is over an 8-week period. 12 Of the initial 25 participants who agreed to be treated, 2 became incarcerated, 2 were unable to be reached by phone and 1 entered a drug rehabilitation center prior to the start of treatment. This left 20 participants starting the Hep C treatment program. Aggregate data reveal the mean age for this cohort was 48, with 71% male and 29% female. Of the participants, 7 (35%) were African American and 13 (65%) were Caucasian.
Implementation Steps
The 20 participants were divided into 2 groups and scheduled for an initial appointment and one 12-week post-treatment lab appointment. The initial appointment included a complete history/physical examination, HCV education, treatment overview, and baseline lab work. Patients identified with mental health disorders were referred to behavioral health. Once insurance approved the DAA and medication was obtained/delivered to the patient, a start date was initiated, and adherence education was emphasized to the patient. The treating NP called patients halfway through treatment, to ensure adherence to treatment regimen and to answer questions. Patients were referred to the clinic community health workers for additional resources related to food insecurity, lack of stable housing, phone, and clothing needs, as well as ongoing transportation issues. If desired, the patient was directed to a Medically Assisted Treatment (MAT) program for substance abuse, but this was not a requirement. When the individual completed the 8-week DAA treatment regimen, they were scheduled to return for a 12-week post-treatment lab draw to confirm the HCV RNA was undetectable. The patient was counseled to continue practicing lifestyle changes and risk reduction to decrease the possibility of reinfection.
Evaluation
The outcome tracked was the HCV viral load at the end at 12 weeks post-treatment which indicates sustained viral response (SVR) or HCV cure. Goal virologic response was defined as the absence of quantifiable HCV RNA in serum (<12 IU/mL). 3 Patients returning for the 12-week SVR were expected to decrease as the participants were no longer taking DAA mediation and had moved on with their lives. However, they were called and advised to return for this final lab test.
Outcomes
The primary project endpoint was to establish if there had been a sustained viral-load reduction (SVR-12) in participants. Descriptive statistics were used to analyze the pre-and post-treatment response to DAA treatment. Data outcomes were measured in percentages of patients who attained an undetectable viral load, which is defined as being <25 IU/mL. An HCV-RNA (Hepatitis C virus ribonucleic acid) test with a detection level of <25 IU/mL is the defined benchmark used to detect HCV RNA. 3
Overall, viral rates declined throughout the duration of the project, as demonstrated in Figure 1. The HCV load measured at 12 weeks post-treatment was measured undetectable in 100% of returning patients, which is considered a cure of HCV. Three patients lost to follow-up were unable to be included in the test of cure, as they were unreachable, despite being called multiple times. Overall, it proved to be challenging to reach many participants due to disconnected phone numbers, incarceration, or entering rehab facilities.
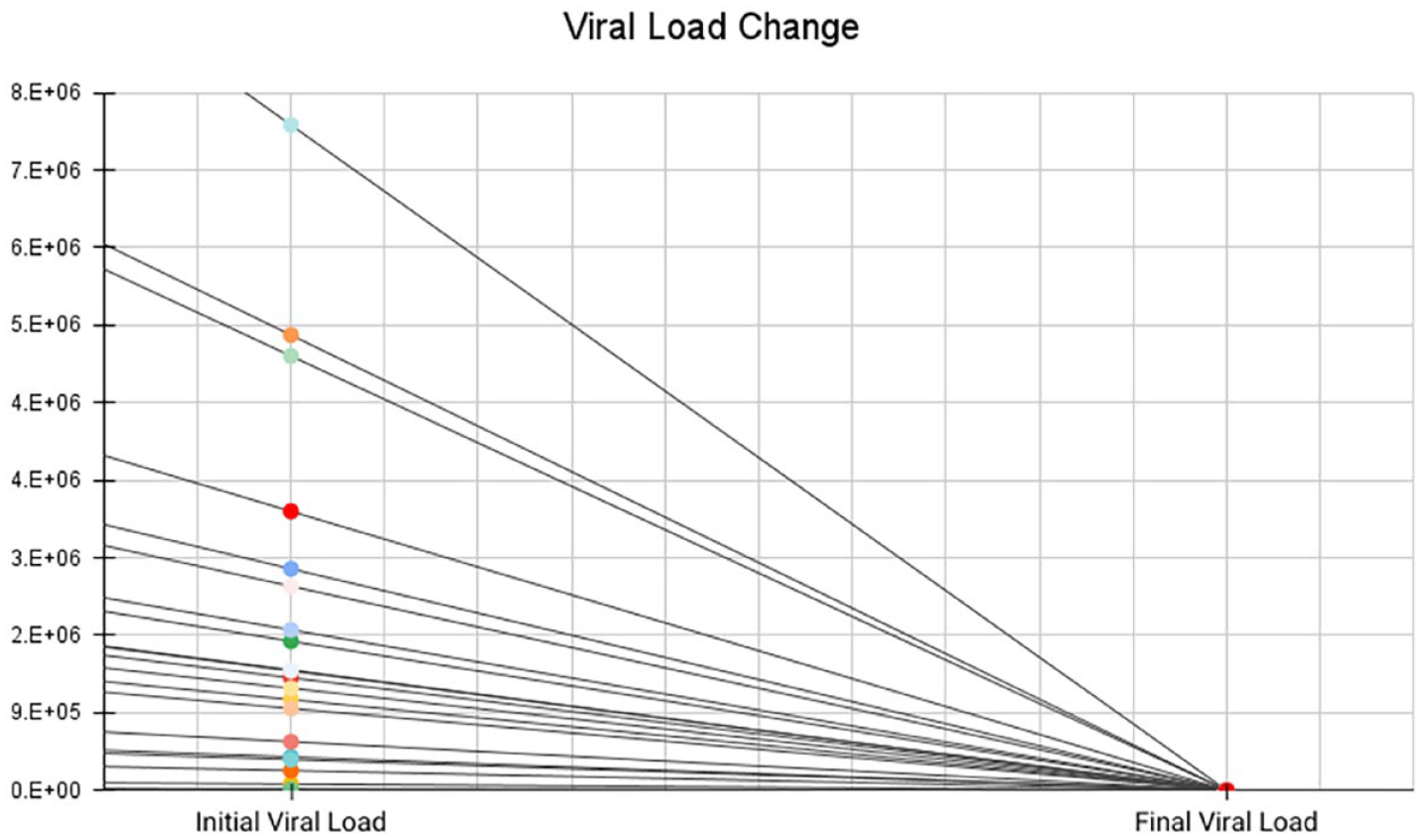
Sustained viral load 12 (SVR-12).
Discussion
The expectation of this study was that >90% of HCV-infected low-income participants, who use illegal substances or inject drugs, would be successfully cured of HCV when treated by a nurse practitioner at an urban FQHC. In this case, 17 participants returning for the SVR-12 had an undetectable viral load and the remaining 3 participants may be tested when/if they return to the clinic. Currently, PWID or use drugs in this community are unable to obtain HCV treatment by the local ID clinic and are referred to a university ID clinic located over an hour away, inaccessible by public transportation. The results of this project indicated that 100% were cured of HCV in a setting that was easily accessible by car, bus, or on foot. Furthermore, this outcome was not influenced by demographics such as age, ethnicity, or continued drug use while undergoing treatment. The outcome of this study is consistent with previous research that demonstrates nurse practitioners can safely and effectively treat HCV in PWID in a community clinic setting without the need for infectious disease specialists administering the DAA. 13
This project reinforces the fact that patients should not be excluded from treatment simply because they are not ready to face their addiction prior to being treated for HCV. The nurse practitioner, involved in this study, felt uniquely equipped to improve the care and treatment of this group of patients and established a rapport with each participant throughout treatment. Given the study was conducted in an urban FQHC, this provided much-needed access to treatment for vulnerable individuals as well as opportunities to obtain insurance, food, clothing, and housing resources.
Limitations
This study did not randomize patient selection and only 1 provider selected and treated the subjects. This can result in provider bias as potential subjects were chosen from a list of individuals who submitted a positive HCV and UDS test over the past 2 years. There also were COVID limitations as some patients may have been afraid to come into the clinic for lab work or attend the initial intake appointment. Furthermore, this project was conducted on a small sample of substance-using individuals, selected only among the clients previously seen at this clinic. Therefore, multicenter studies, including equal numbers of various ethnicities, are suggested to investigate if HCV treatment in substance users by a nurse practitioner is effective in larger PWID populations.
Conclusion
Injection drug use (IDU) is the most common risk factor for HCV infection in the United States and Europe. 14 Targeting the population of individuals who use drugs is an effective way to decrease the HCV rates in a community. To achieve this goal, innovative strategies are needed to direct these patients to treatment facilities soon after being identified as positive for HCV. To that end, this healthcare delivery QI initiative was aimed to address the lack of opportunity for PWID or use drugs to obtain HCV treatment in this local community, where many patients can walk or take public transportation to the community clinic. Additionally, treating HCV in this FQHC provides access to vital services such as bus passes, insurance, housing, food and clothing pantry, and addiction and mental health services, which can all be addressed by the FQHC staff.
Our country is now in a position where DAA treatment for HCV is simplified for providers and patients can achieve a SVR with few side effects. This study demonstrates that a non-specialist can treat HCV in vulnerable, low-income patients who continue to use illegal substances. They can be cured of their HCV and may return for additional services, even referral to addiction services when they are ready. Most importantly, they are more likely to return to a clinic whose staff treated them with kindness and compassion when treating their HCV.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
