Abstract
Introduction:
Canadian Indigenous populations have a high incidence of poisoning; it has been suggested that care provided to the population living in remote areas is suboptimal. Our study aims to compare the continuum of care of poisoned people in Indigenous communities with those in non-Indigenous communities located in rural regions in the province of Québec.
Methods:
We conducted a multicenter retrospective cohort study using data from the Center Antipoison du Québec (CAPQ) over a 2-year period (2016-2017). We evaluated the care trajectory of Indigenous patients suffering of poisoning as compared to non-Indigenous patients living in rural areas. Our primary outcome was the duration of CAPQ involvement in case management. Our secondary outcome was the symptoms severity at the end of case management.
Results:
Among 491 identified poisoned patients (238 Indigenous/253 non-Indigenous), the duration of CAPQ involvement in case management was 9.4 h [2.9-21.3] for Indigenous patients versus 5.5 h [0.1-14.4] for non-Indigenous patients. No statistically significant difference was found between groups (geometric means ratio (GMR) adjusted = 1.08; [0.84; 1.38]). Results were consistent by age and sex groups. Most patients, in both Indigenous and non-Indigenous, showed mild to moderate symptoms at follow-up (59% vs 54%). One death was registered in each group. The CAPQ received a limited number of calls from the non-conventioned First Nations during the study period.
Conclusions:
We did not observe differences on the duration in case management. Perceptions of suboptimal care provided to rural Indigenous population are likely to be related to geographical remoteness rather than ethnicity. Further research is needed to better identify potential factors involved in the continuity of care provided in emergency situations. Another study will be carried out to describe the Indigenous realities and to better understand the results of this study.
Introduction
Acute poisoning is a major public health problem worldwide.1,2 In Canada, the number of poisoning cases has increased in recent years, with poisoning now among the top 10 causes of medical admissions to an intensive care unit. 3 Recent evidence shows that poisoning was responsible for the deaths of 5516 Canadians and 24 024 hospital admissions, with an associated healthcare cost estimated at CA $4.2 billion in 2010.4-6 The distribution of intentional and unintentional poisoning cases in Canada is not uniform between regions. 7 Data from the Centre Antipoison du Québec (CAPQ) and the Nunavik and James Bay (Eeyou Istchee) social-health regions both of which include predominantly Indigenous territories, reported 1160 and 1021 poisonings respectively per 100 000 persons per year between 2008 and 2014. 7 A similar pattern of poisoning has been observed in other parts of Canada like Yellowknife, Fort Smith, Inuvik, and British Columbia localities.8,9 In contrast, some main social-health regions in the Quebec province, such as Montréal and Capitale-Nationale, report significantly fewer poisoning cases, with 437 and 606 cases respectively per 100 000 persons per year. 7
The higher incidence of poisoning among Indigenous populations may be an artifact of the organization of healthcare services.7,10-12 In fact, in Indigenous communities, populations only have access to primary care services on site, all cases are referred to specialized centers; whereas large hospital centers have the resources to manage cases in-house. Suboptimal provision of health care was also suggested by previous studies to explain poisoning complications burden among Indigenous population.7-9 Nevertheless, limited data exists describing poisoning outcomes among these populations. To that end, we aim to compare the continuum of care of poisoned people in Indigenous communities with those in non-Indigenous communities living in rural regions. Considering the precarious living conditions experienced by some Indigenous populations, including difficult social environment and health care conditions,11,13,14 we hypothesize that Indigenous populations might experience a longer duration of poisoning case management and poorer overall clinical outcomes than non-Indigenous patients living in similar rural areas.
Methods
Study Design
This multicenter retrospective cohort study is part of the larger Canadian CARe for Indigenous Poisoned Patients research program (CARIPP), developed «to support the health, wellness goals of Indigenous peoples through contributions to evidence-informed, culturally sensitive, health, and social policy interventions».
Data Collection
Participants
We identified all poisoning cases meeting the following inclusion criteria: coming from rural regions of Québec, seen in hospital and reported to the CAPQ from June 2016 to October 2017 using the TOXIN database. 15 This database includes approximately 1 500 000 patient records since implementation in 1986, with 24 300 patient records added during the last 5 years (2014-2018). We use the residency location as a proxy for the ethnic origin. We began by identifying Indigenous patients indexed in the database with the names of targeted First Nations communities and Inuit municipalities.16,17 Secondly, we identified non-Indigenous populations as all other communities located in the same regional county municipality (RCM) as the previously determined Indigenous communities. 18 Of these, we selected patients living in small towns or rural areas.19,20 We subsequently excluded non-poisoning cases, minor clinical effects cases (situation not requiring treatment in a healthcare setting other than a decontamination), chronic exposures and nations close to urban regions (<50 km), such as the Mohawks (Montréal area), Abénaquis (Montréal area), and Hurons-Wendats (Québec City area). We developed an anonymized database using medical records from the CAPQ. In fact, each report to the CAPQ is recorded electronically or manually in the TOXIN database, which includes medical records from individuals and health professionals who call seeking help for a poisoning emergency. From this database, the data of our study were collected independently by 4 individuals (ED, AC, KGL, and DRC) on a standardized case report form. Before commencing data collection, we assessed the reliability of data extraction between each pair of data abstractors after training with 30 patient records using a Cohen’s kappa statistic reaching at least 0.80. One researcher with clinical training in toxicology (MSO) verified randomly 10% of the collected data. Disagreements were resolved by reviewing data with the other data abstractors.
Outcomes measures
Primary outcome
Our primary outcome was the duration of care provided by the CAPQ in the management of poisoning cases. This measure was defined as the total time between the first and the last call to or by the CAPQ, assuming that toxicological treatment had been completed.
Secondary outcome
Our secondary outcome was the severity of the symptoms measured at the end of the episode of care. We used the classification used by the CAPQ which is defined as follows: (1) asymptomatic, when patient remained asymptomatic throughout the poisoning episode; (2) mild symptoms, patient presenting or having presented symptoms that did not threaten organ integrity and/or patient’ life and requiring nothing beyond simply decontamination as treatment (ie, cough and brief respiratory symptoms following ingestion of paraffin oil, asymptomatic thereafter); (3) moderate symptoms, requiring more advanced treatment in healthcare setting and did not threaten organ integrity and/or patient’s life (ie, acetaminophen ingestion successfully treated with N-acetylcysteine); (4) severe symptoms, for organ or life-threatening poisoning (ie, hepatic failure following acetaminophen poisoning requiring life support measures); and (5) death. We also looked at the effect of sex for both primary and secondary outcomes.
Statistical analyses
Based on a previous preliminary study carried out by the CARIPP program, a sample size of 150 patients per group was required to detect a 2 h difference in the duration of care (alpha error 5%, power of 80%). Patient characteristics were presented as proportions and medians (interquartile range). Chi2 and Wilcoxon-Mann-Whitney tests were used to compare patient characteristics according to their ethnic origin. We used the ethnic origin as the independent variable based on the self-reported location of the poisoning or the patient’s self-reported place of residence when this information was not available. The Indigenous consisted of 2 distinct groups: First Nations (conventioned and non-conventioned) and Inuit populations.21,22 The convention refers to a land claim settlement approved by the Cree and Inuit of northern Quebec, and later slightly modified by the Northeastern Quebec Agreement, through which Quebec’s Naskapi First Nation joined the agreement. The agreement covers economic development and property issues in northern Quebec, as well as establishing a number of cultural, social, and governmental institutions for Indigenous people who are members of the communities involved in the agreement. The conventioned communities are those governed by these agreements and the non-conventioned communities are those not governed by these agreements. Because they are important risk factors for the occurrence of poisoning, multivariable generalized linear regression models were performed for our primary outcome adjusted for potential confounding factors, such as age, sex, type of substance involved, poison severity score (1 = asymptomatic to 5 = death). 23 We also adjusted for distance between village and first referral medical center, and distance between referral and last admitting medical center because distance from a patient to a provider intuitively seem to be a barrier to health care access. These distances were calculated by using google maps. In order to improve the regression coefficient estimates, we planned to perform multiple imputation for variables which presented more than 5% of missing data. We used the inverse probability censored weighting (IPCW) methodology in our models to account for potential selection bias from lost to follow-up patients. 24 The association between duration of care and ethnic origin was examined with geometric means ratios (GMR). A log-transformation was used to satisfy the postulates of the regression model. A constant of 1 for all patients was added to perform the log-transformation. We also explore whether sex and age are effect modifiers of the continuum of care and ethnic origin association by using interaction terms and, when significant, the Bonferroni method was used. 25 Confidence intervals were reported at 95%. We planned performing subgroup analyses based on the type of poisoning (voluntary vs not and acetaminophen vs not) and time from exposure to the first call to test the robustness of our results. To estimate the mean time from exposure to first call, we used the mean time between the earliest and the latest time before call to the CAPQ, reported in the patient’s medical record. For missing data for the time from exposure to the first call, we performed 2 analyses based on the imputation of the (i) median mean time and (ii) median of the latest time available. We performed other sensitivity analyses by estimating adjusted results with and without the IPCW methodology. All statistical analyses were conducted using SAS software, version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Patients and Poisoning Characteristics
A total of 950 medical records were reviewed and 491 patients were included in our study (238 Indigenous and 253 non-Indigenous). Of those, 126 patients were lost to follow-up. Thus, 365 patients were included in analysis (Figure 1 and Supplemental Material 1). Crees and Innuits were the most representative Indigenous communities (91%) with very limited consultations for the non-conventioned First Nations (7%). The median patients age was 21 years (16-27) in Indigenous and 30 (17-48) in non-Indigenous. Women were over-represented (65% in Indigenous and 55% in non-Indigenous). In terms of substances, medicines were the most common source of poisoning in Indigenous population (85%), followed by alcohol (43%) and illicit drugs (18%); in the non-Indigenous population, the most common were medicines (72%) and alcohol (14%). Most poisoning cases were voluntary in both groups (72% vs 51%). We observed a shorter delay between exposure and first call to the poison center in Indigenous as compared to non-Indigenous (1.3 h [0.8-3.5] vs 3.5 h [0.8-3.5]). The call to the poisoning center is mostly made by a healthcare professional, 95% in Indigenous versus 71% in non-Indigenous. Forty-seven percent of Indigenous patients were first evaluated by a physician as compared to 70% of non-Indigenous patients. A larger proportion of patients in the Indigenous group were transferred to another healthcare facility for further medical support (55%vs 11%). The median distance between the first and the last medical contact during transfers was 179 km [87-334] as compared to 70 km [44-332] in the non-Indigenous group (Table 1 and Supplemental Material 2).
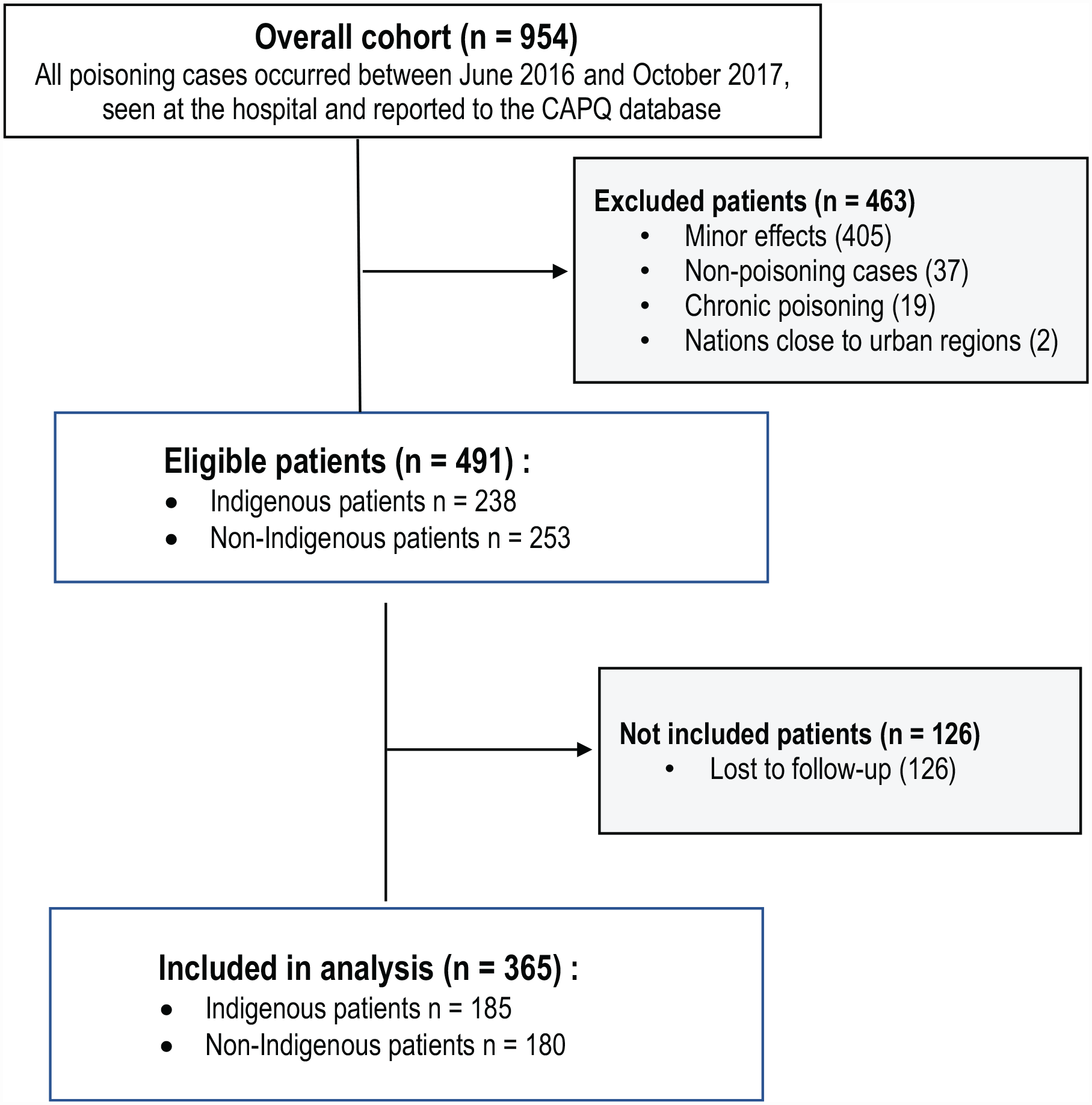
Study flow diagram.
Distribution of Indigenous and Non-Indigenous Socio-Demographic and Medical Characteristics.

Data are presented as the median [interquartile range] or n (%) of case patients.
P-value: chi2 test was used for categorical variables and Wilcoxon-Mann-Whitney test for continuous variables.
Missing Value (Age, n = 0 Indigenous and n = 1 non-Indignous/Weight, n = 45 Indigenous and n = 65 non-Indigenous).
Non-mutually exclusive categories (Each category was coded Yes/No).
Primary Outcome
Following risk adjustment for important potential confounders, no statistically significant difference was found between Indigenous and non-Indigenous patients for the duration of the management of care (GMR adjusted = 1.08; [0.84; 1.38]) (Figure 2, Table 2 and Supplemental Material 7). Results were not influenced by the age and sex of the patients.

Distribution of the duration of care provided by the CAPQ in poisoning cases management.
Crude and Adjusted Associations Between Ethnic Origin and Duration of Care Provided by the Poison Center in the Management of Poisoning Cases.
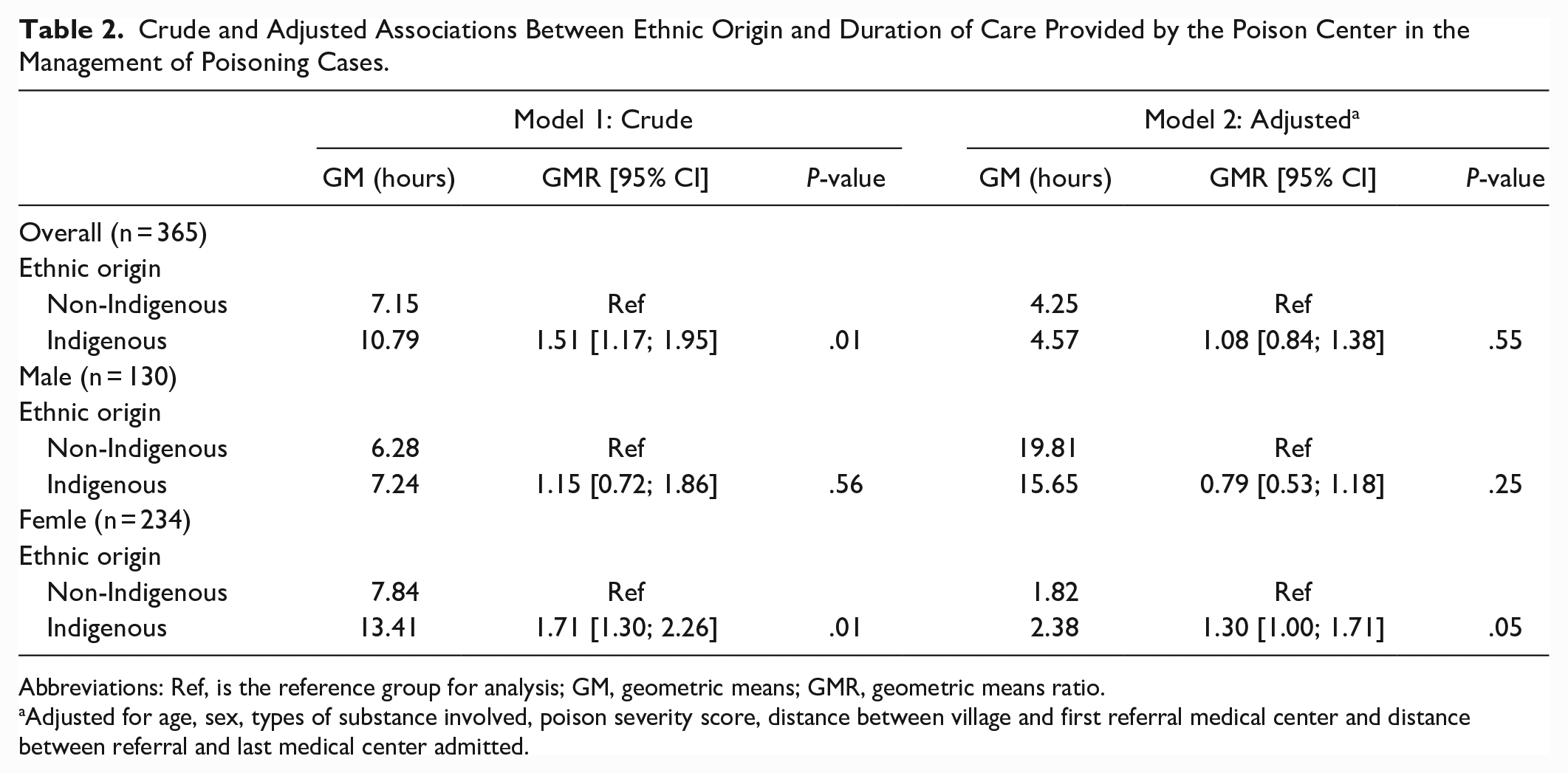
Abbreviations: Ref, is the reference group for analysis; GM, geometric means; GMR, geometric means ratio.
Adjusted for age, sex, types of substance involved, poison severity score, distance between village and first referral medical center and distance between referral and last medical center admitted.
Secondary Outcome
The severity of the symptoms was comparable between groups. Most patients, in both indigenous and non-indigenous (59% vs 54%), showed mild to moderate symptoms at follow-up (Table 3). We observed one death per group during the study period.
Distribution of Primary and Secondary Outcomes Among Indigenous and Non-Indigenous Groups.

Abbreviations: CAPQ, Center Antipoison du Québec.
Data are presented as the median [interquartile range] or n (%) of case patients.
Missing Value : Overall, n = 2 Indigenous and n = 2 non-Indigenous.
Sensitivity Analyses
Patients excluded from our main analyses were comparable for the most important variables to the study participants (Supplemental Material 3). No statistically significant difference was found between groups when considering only voluntary poisoning cases, cases excluding acetaminophen, nor when considering delays since exposure to the last call to the CAPQ as the outcome (Supplemental Materials 4 and 5). Inconsistent variations were observed based on sex in non-acetaminophen poisonings and when data imputation was performed (Supplemental Materials 4 and 5). Moreover, inconsistent variations were observed with and without the IPCW methodology (Supplemental Material 6).
Discussion
In our study, we did not observe a difference in the duration of poisoning case management between Indigenous and non-Indigenous populations. This observation was consistent regardless of age and sex. We also observed no statistically significant difference in clinical outcomes. Very limited number of consultations to the CAPQ were received from the non-conventioned First Nations during the study period.
Our results were inconsistent with those of previous studies conducted in acute care settings in Indigenous populations.26-28 Most studies conducted so far showed that Indigenous population may be subject to suboptimal care such as delays in disease diagnosis, treatment, or referral, poor assessment and inadequate or inappropriate patient management. Inadequate risk adjustment between Indigenous and non-Indigenous populations may explain part of the discrepancies observed as compared to our study. Also, the location of the studies may probably have an impact on results; previous studies for the most part conducted outside of Canada, in Australia. Results from a study carried out to investigate poisoned children aged 0 to 4 years although showed a comparable hospital length of stay between Indigenous and non-Indigenous patients. 29
In our study, we observed a surprisingly high proportion of patients showing mild to moderate symptoms at follow-up and one death in each group. Our findings were not consistent with previous studies conducted in Australia, New Zealand, the USA, or Canada.30-36 However, most studies were designed to compare Indigenous populations to the overall population rather than to non-Indigenous living in remote areas where most of these communities live. The distance from health care services being known to be associated with a greater risk of unfavorable outcome in emergency and critical care this may have led to an inappropriate risk adjustment between groups in these studies. Our results suggest that problems highlighted in the management of poisoning may have more to do with the geographic remoteness of the population as opposed to the ethnicity. On the other hand, patients from the non-conventioned First Nations were underrepresented in our study due to the limited number of consults and calls made to the CAPQ by these populations. The fact that non-conventioned First Nations receive medical care under the federal jurisdiction rather than provincial jurisdiction may explain these observations.
Limitations
We performed a multicenter cohort study in an integrated provincial care system context including patients cared by the medical services of the CAPQ. 37 As compared to previous work, we performed risk adjustment based on remoteness as a potential confounding factor considering that several communities live in remote rural areas. We also accounted for the potential impact of a potential selection bias in our analyses from patients excluded due to lost to follow-up.
Our study is not without limitations. First, we use the residency location as a proxy for the ethnic origin and cannot exclude some potential misclassification. Second, we estimated the distances using Google Maps, taking into consideration the zip code of the area. This method may have led to some imprecision in the distance from the care center used in our analyses. However, considering that we used the zip code of the area, it should have led to an underestimation of distances which could however reinforced the absence of effect between groups. Third, despite the fact that all patients came from remote regions, most Indigenous individuals included lived in the region, “Nord du Quebec,” while the minority of non-Indigenous individuals included did not. This may introduce bias. Fourth, due to our inability to access health records of the primary care centers and the retrospective design of the study, we were unable to collect some missing information and data. Fifth, difference in the mode of transportation (eg, ambulance, airplane, etc.) and seasonality were not considered as potential confounding factors. Sixth, our study was based on a limited sample size, and we cannot entirely exclude a difference in care between Indigenous and non-Indigenous considering the measures of dispersion around our estimates. Moreover, the reliability and validity of the CAPQ database is less well-known. 38 Last, our study was conducted in a province with an integrated trauma and poisoning system with clear corridors of service from primary to tertiary care; our results thus may not apply in other clinical contexts.
Conclusion
In our study, we did not observe evidence of disparities in the clinical management of poisoning cases between Indigenous and non-Indigenous populations living in remote areas. Geographical remoteness more than ethnicity likely explains the issues observed in previous studies in the Indigenous population. However, our study has limitations, and results must be interpreted with caution. Further studies should examine the impact of geographical remoteness on the continuity of healthcare provided in emergency situation, and better describe the continuum of care of Indigenous poisoned patients receiving health care under federal jurisdiction. Another study will be carried out to describe the Indigenous realities in the field and to better understand the results of this study.
Supplemental Material
sj-docx-1-jpc-10.1177_21501319231178654 – Supplemental material for Clinical Outcomes of Indigenous Versus Non-Indigenous Patients: A Multicenter Retrospective Cohort Study in the Province of Quebec
Supplemental material, sj-docx-1-jpc-10.1177_21501319231178654 for Clinical Outcomes of Indigenous Versus Non-Indigenous Patients: A Multicenter Retrospective Cohort Study in the Province of Quebec by Ella Diendéré, Alexis F. Turgeon, Katherine Gagnon-Labelle, Amélie Couture, Xavier Neveu and Maude St-Onge in Journal of Primary Care & Community Health
Footnotes
Acknowledgements
The authors want to thank Olivier Costerousse and Marjorie Daigle for their administrative help, Joëlle Murray Bergeron for her help on start-up data collection with the CAPQ TOXIN database and David Rodrigues Cabrita for his help in collecting the data. They also want to thank Marie-Claude René and Barbara O’Farrell from the Nunavik Regional Board of Health and Social Services (NRBHSS) and Jill Torries from the Cree Board of Health and Social Services of James Bay (CBHSSJB) for their help in the development of the study protocol and the interpretation of the data results.
Authors’ Note
All authors had full access to all data and take responsibility for its integrity and the accuracy of its analysis.
This article has been peer reviewed.
Authors’ Contributions
Maude St-Onge had the original idea for this work. Ella Diendéré, Alexis F. Turgeon, and Maude St-Onge developed the protocol. Ella Diendéré, Katherine Gagnon-Labelle, and Amélie Couture collected the data. Tatiana Ella Diendéré and Xavier Neveu performed the analyses. Ella Diendéré, Alexis F. Turgeon, and Maude St-Onge drafted the manuscript. All of the authors participated in the interpretation of the data, revised the manuscript critically for important intellectual content, gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported in part by the Fondation du CHU de Québec-Université Laval, who had no role in any part of conducting the study or preparing the manuscript. Mrs. Diendéré was funded by a Training Award from the Fondation du CHU de Québec-Université Laval. Dr. Turgeon is the Canada Research Chair in Critical Care Neurology and Trauma. Dr. St-Onge is a recipient of a Salary support Award from the Fonds de recherche du Québec -Santé (FRQS). Dr. St-Onge is also a recipient of the Canadian Critical Care Trials Group/Canadian Critical Care Forum (CCCTG/CCCF) Early Career Research Grant.
Ethics Approval and Consent to Participate
This study was approved by the Research Ethics Review Boards of the Centre Intégré Universitaire de Santé et de Services Sociaux (CIUSSS) de la Capitale-Nationale and of the Centre Hospitalier Universitaire (CHU) de Québec-Université Laval.
Consent for Publication
All individuals named in the acknowledgements section below consented to being acknowledged in this publication.
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article [and its supplementary information files].
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
