Abstract
Background:
Chlamydia trachomatis and Neisseria gonorrhea are the most reported sexually transmitted infections in the United States. Testing rates remain suboptimal and may be subject to implicit bias. We evaluated the effectiveness of an opt-out chlamydia and gonorrhea testing program for adolescents and young adults in improving testing rates and promoting equity.
Methods:
An opt-out testing program that standardized testing to once annually for 14 to 24-year-old patients was implemented across 28 federally qualified health centers spanning 4 specialties. A quasi-experimental design using interrupted time series analyses evaluated testing and infection rates between baseline, intervention, and pandemic-associated test shortage periods. Reduction in testing inequities based on sex, race, ethnicity, insurance, and language preference were also examined.
Results:
A total of 57 452 encounters during the baseline, 17 320 during the intervention, and 26 993 during the test supply shortage periods were included. Testing increased from 66.8% to 81.0% (14.2% absolute increase) between baseline and intervention periods. Pediatric clinics demonstrated the largest improvement compared to other settings (absolute increase 30.9%). We found significant reductions in testing inequities for language preference (P < .001), and un-insured and public insured individuals (P < .001). More cases of chlamydia and gonorrhea were detected in the intervention period (chlamydia-29.7; gonorrhea-7.4 per 1000 patients) than in the baseline period (chlamydia-20.7; gonorrhea-4.4 per 1000 patients; P < .001).
Conclusions:
An opt-out approach increased testing, reduced inequities between some groups and detected more infections than a risk-based approach. Opt-out testing should be considered as an approach to increase detection of chlamydia and gonorrhea and promote equity.
Background
Chlamydia trachomatis (CT) and Neisseria gonorrhea (GC) are the 2 most reported sexually transmitted infections (STIs) in the United States and the prevalence has markedly increased over the past 2 decades. 1 In alignment with national trends, the Denver Metro area reported a 24% increase in CT and 150% increase in GC from 2014 to 2018 with the highest prevalence of CT and GC among adolescents and young adults.2,3 Among patients tested at our organization from 2018 to 2019 (Denver Health and Hospital Authority [DHHA]), 15% of women and 12% of men were positive for CT or GC at least once. 4
Many CT and GC infections are asymptomatic; thus, screening is essential to reduce infection rates and prevent morbidity. Currently the United States Preventative Services Task Force (USPSTF) recommends annual screening of sexually active females under age 25 and selective screening of those deemed to be high risk. 5 Similarly, the American Academy of Pediatrics recommends annual screening of sexually active females who are under 25 years of age. 6 Thus, choosing to screen a patient requires that the provider obtain an accurate sexual history and determine risk status. This can be challenging with adolescents and young adults and is infrequently completed by providers. 7 We previously evaluated CT and GC screening rates among over 40 000 primary care patients at DHHA and found that only 20% of males and 48% of women aged 14 to 24 years old were tested in a 2-year period. 4 Additionally, we identified CT and GC cases in patients that were documented as “not sexually active.” These data demonstrated significant inequities in testing rates with disproportionate testing of women who identified as Black or African American compared to those who identified as White. This is concerning because significant social stigma is associated with STI testing.8-10 Sex, racial, and ethnic inequities in testing that disproportionately target particular groups further contribute to negative social stigma and structural racism which may limit the willingness of patients to seek care for testing.10,11
Of interventions designed to improve testing, universal testing or opt-out approaches have the greatest impact on testing rates in select settings. 12 These programs have also been shown to be acceptable by patients, cost-effective and reduce STI associated complications.13-15 Opt-out testing may decrease bias by removing the need for providers to determine who is high risk because determination of risk is vulnerable to implicit bias.14,16 Opt-out testing may consequently lead to more equitable care. However, to date most studies evaluating opt-out testing approaches have been restricted to females15,17-20 and have only occurred in selective clinical environments including adolescent specialty clinics, 18 STI and family planning clinics, children’s hospitals, 17 detention centers, and emergency departments.21,22 Given that most adolescents and young adults receive medical care in community-based primary care clinics, 16 understanding if opt-out testing is effective in improving testing rates and providing more equitable care in these settings is essential. In 2019 we piloted opt-out testing for adolescents in a pediatric and family medicine clinic and demonstrated significant improvements in testing rates and high potential for sustainability. 23 The program was also found to be cost-neutral for our organization. Based on these results opt-out testing was expanded to all 28 federally qualified health centers (FQHCs) in our system in March 2020. We aimed to evaluate the effectiveness of the universal opt-out CT and GC testing program in improving testing rates and promoting health equity.
Methods
Setting
DHHA is an integrated, safety-net healthcare system located in Denver, CO comprised of 28 FQHCs (10 multi-specialty clinics and 18 school-based health centers [SBHC]), a level 1 trauma center, and encompasses the Public Health Institute at Denver Health and the regional STI clinic. 24 Similar to other FQHCs, nearly 3 quarters of patients have Medicaid or are self-pay. 25 Among all DHHA patients, 16.0% were uninsured and 60.9% had Medicaid or Children’s Health Insurance Program. In 2020, racial and ethnic demographics for all patients who presented to DHHA were 29% white, 17% black, and 54% of patients identified as Hispanic.
Intervention
In 2020, CT and GC testing became an institutional strategic target at DHHA. The opt-out CT and GC testing program recommended annual nucleic acid amplification testing (NAAT) of male and female patients 14 to 24 years old regardless of risk factors and was implemented across all primary care divisions including pediatrics, family medicine, school-based health centers, general internal medicine, and women’s care (obstetrics and gynecology). For patients 14 to 17 years of age testing occurred at well child visits if it had not been completed within the past year. For patients 18 to 24 years of age testing occurred at any primary care visit if it had not been completed in the prior year. All patient and parents of adolescents were provided a comprehensive education sheet at check-in that discussed sexual health, sexual health resources, and the opt-out testing program. Medical assistants ordered tests when indicated based on an electronic health record alert that appeared during rooming, collected urine, and verified a confidential phone number when indicated. The workflow was designed to appropriately protect confidentiality for adolescents. Standard scripting was provided for medical assistants to explain universal testing to patients. All patient questions or concerns were deferred to the provider. The workflow and scripting were created with medical assistant input and approved by nursing leadership. When a test was positive, patients were contacted and treated according to institutional clinical care guidelines. For adolescents, results were confidentially reported directly to the adolescent using the confidential phone number provided at test ordering. Appropriate confidential care for treatment was arranged at a DHHA clinic. Order sets were created in the electronic health record to provide real-time decision support based on test results to guide ordering of treatment and follow-up laboratory tests.
Data
This was a quasi-experimental analysis consisting of multiple intervention periods. We used DHHA’s enterprise data warehouse to collect data for testing encounters (using codes for lab tests) and patient demographics. We collected baseline data from January 1, 2019 to February 29, 2020, intervention data from March 1, 2020 to August 31, 2020, and COVID-19 associated CT and GC test shortage and recovery data from September 1, 2020 to April 20, 2021 (Figure 1). Unprecedented demand for testing reagents resulted in critical CT and GC testing supply shortages starting in September 2020. This required restriction of testing to those deemed high risk from September 1, 2020 to December 1, 2020. After December 2020, testing was gradually increased back to universal testing. Using the available data, we used an internal algorithm to determine whether the patient was tested within the past year during the evaluation month. Only patients with in-person encounters in a primary care setting were included in the analysis.

Impact of universal CT and GC testing on monthly testing rates (January 2019-April 2021).
Outcome Measures
The primary outcome was CT and GC NAAT testing completed on the day of the office encounter or within the prior year. Eligible encounters included in-person primary care visits for 14 to 24-year-old patients during the designated period. Secondary outcomes included number and rates of CT and GC cases detected among primary care patients with visits during the time period.
Statistical Model
We used a multi-interrupted time series model to assess the impact of the intervention on the population level across the clinics. 26 The model took the general form:
where month is the incremental increase in months starting with the baseline period, interventionint is a dummy variable (0/1) that identifies the period in which the universal intervention occurred, timeint is the incremental increase in time with respect to the beginning of the interventionint period, interventionshortage is a dummy variable (0/1) that identifies the period in which the shortage occurred, and timeshortage is the incremental increase in time with respect to the beginning of the shortage period.
We modeled in 3 separate time frames, baseline, intervention, and test shortage and recovery, and assessed changes in testing rates and trends for each time frame. To determine whether the intervention decreased inequities among the patient population, we analyzed differences in screening rates between subgroups during the baseline and intervention periods. We calculated the difference between the screening rate for the reference group and the subgroup for each time frame. We subsequently assessed whether there was a significant difference in these inequities between the time frames. The number and rates of CT and GC cases detected were plotted by month against the community prevalence in the Denver metropolitan area during the same time frame. The rates of cases of CT and GC detected were compared between time periods using Pearson’s chi-squared tests. An alpha of .05 or less was used to determine statistical significance.
The project was reviewed by the Quality Improvement Committee of DHHA, which is authorized by the Colorado Multiple Institutional Review Board at the University of Colorado, Aurora, CO, and the DHHA Ethics Committee, Denver, CO, and was exempted as it was determined not to be human subjects’ research.
Results
There were a total of 57 452 eligible encounters during the baseline period, 17 320 encounters during the intervention period, and 26 993 during the test supply shortage and recovery phase (Table 1). Included patients tended to be female, white, and Hispanic and had public insurance. More than 20% of the encounters were for patients with a preferred language of Spanish. Over 40% of encounters occurred in family medicine clinics.
Patient Demographics for Encounters Stratified by Intervention Period.

Testing
Table 2 contains the unadjusted changes in testing rates, stratified by subgroups. There was an increase of 14.2% between the baseline (66.8%) and intervention (81.0%) periods and a subsequent decrease of 12.6% between the intervention (81.0%) and the shortage (68.4%) periods (Figure 1). Pediatric clinics demonstrated the largest improvement in testing rates from baseline to intervention compared to other clinical settings (absolute increase 30.9% vs family medicine 11.9%, internal medicine 5.2%, women’s care 1.1%; Table 2).
Unadjusted Testing Rates During Baseline, Intervention, and Test Shortage Time Periods.

In the adjusted interrupted time series model, there was an 8.7% (P = .021) increase in testing rates between the baseline and intervention periods and a decrease of 20.6% (P < .001) between the intervention and test shortage and recovery period. During the test shortage and recovery period, after an initial decrease in testing there was an adjusted 2.6% (P < .001) increase in testing per month as the shortage resolved (Figure 1).
Health Equity
We found significant reductions in testing inequities for sex, language preference, and insurance. The difference in testing rates decreased from 37.5% to 22.7% (P < .001) between male and female patients and from 16.8% to 4.7% (P < .001) between English and Spanish-speaking patients (Table 3). Differences in testing rates between patients who were self-pay or utilized financial assistance and those with public insurance decreased, though the difference between those with public and commercial insurance increased. Though not statistically significant there was decrease in the difference in testing rates between White patients and Black or African American patients with universal testing from 4.2% to 2.6% (P = .22).
Changes in Testing Rate Inequities by Subgroup.

Number of Cases Detected
The number of CT cases detected at DHHA increased from 20.7 cases per 1000 patients at baseline to 31.1 cases per 1000 patients during the intervention period and decreased to 23.3 per 1000 patients during the period of COVID-19 associated test shortage and recovery (P < .001). Similarly, the number of GC cases increased from 4.4 cases per 1000 patients at baseline to 9.6 cases per 1000 during the intervention period followed by a decrease to 5.3 per 1000 during the test shortage and recovery period (P < .001). The prevalence of CT and GC in the Denver metropolitan area remained stable during these periods (Figure 2a and b; Supplemental Table).
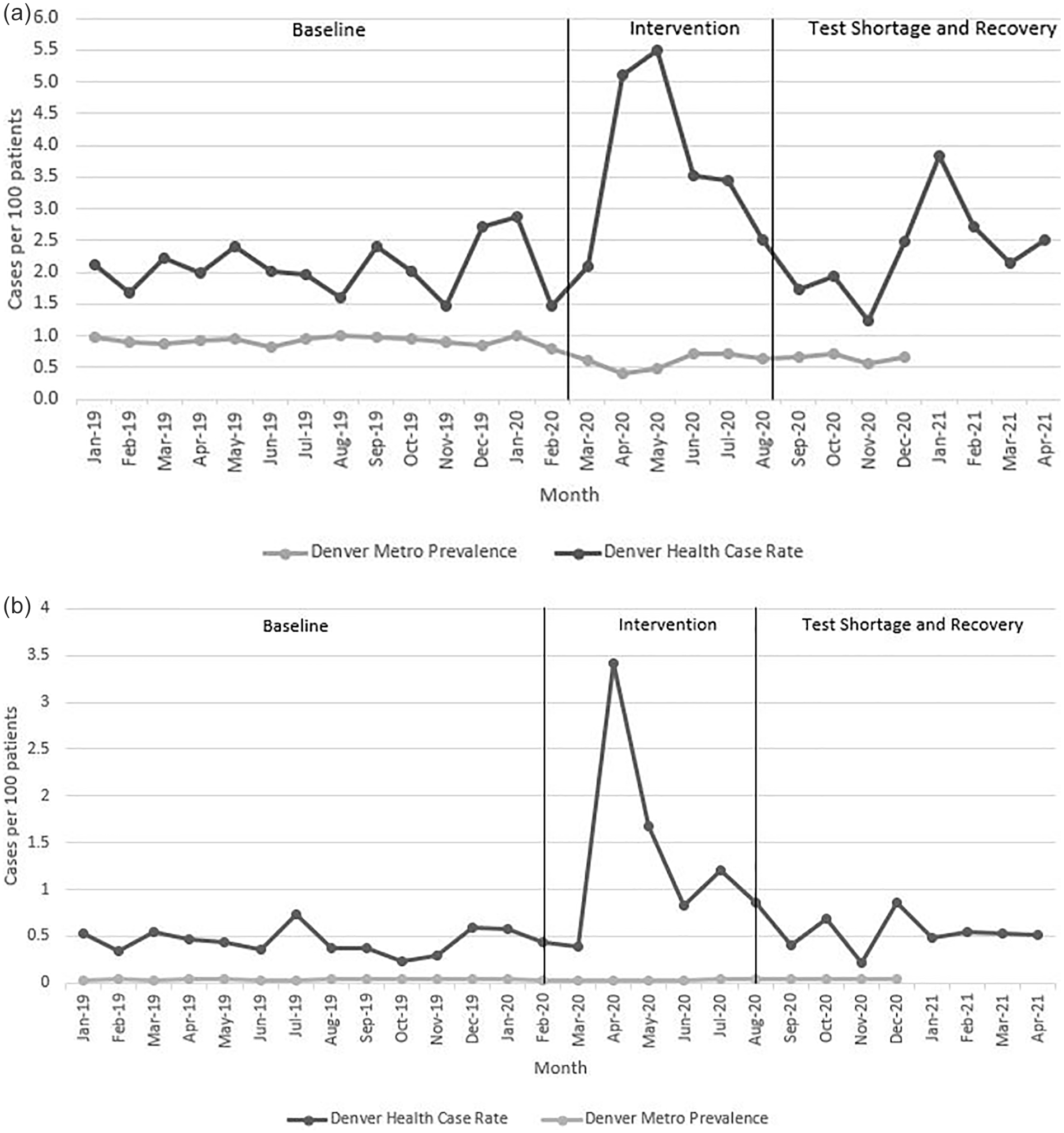
(a) Monthly prevalence of chlamydia at DHHA and Denver metropolitan area by time period and (b) monthly prevalence of gonorrhea at DHHA and Denver metropolitan area by time period.
Discussion
An opt-out CT and GC testing approach in male and female adolescents and young adults presenting for primary care significantly improved testing rates and reduced testing inequities in several subgroups across pediatric, internal medicine, women’s care, and family medicine clinical settings. Prior to the implementation of universal testing and during the period of testing supply shortage, testing rates were highest among patients who were female, Black or African American, Hispanic, English speaking, or had public insurance. However, during the intervention period and during the resolution of the test supply shortage testing inequities were reduced. Data suggest that more cases of CT and GC were detected with opt-out testing than with traditional risk-based testing.
While universal testing improved testing rates in all clinical settings, the greatest improvement was seen in pediatric clinics. Because adolescents account for over half of CT and GC cases and they frequently seek care in pediatric settings, opt-out testing may be a particularly beneficial approach to improving STI care in these locations. Well adolescent visits are a natural fit for implementation of opt-out testing protocols because anticipatory guidance and confidential care are already integral parts of these visits. Testing rates were higher at baseline in women’s care and family medicine clinics likely due to increased comfort level with providing sexual healthcare and fewer confidentiality concerns associated with adolescent patients.
Though providers were not systematically surveyed, anecdotal reports from providers in all specialties indicate that opt-out testing could reduce stigma and bias since the testing is automatically done at least annually on all patients presenting for a primary care visit regardless of risk. Because testing was considered standard for everyone providers could normalize testing when talking with patients, which has been shown to reduce stigma and shame. 11 We recently reported that opt-out testing also aligns with the values of patients. Of patients surveyed in our system 93% preferred opt-out testing over risk-based testing and opt-out testing was associated with reduced feelings of stigma and discrimination compared to risk-based approaches. 27 A key aspect of the success of the implementation of opt-out testing was shifting the CT and GC testing workflow to the medical assistant rooming procedures rather than relying on a provider-based workflow. In contrast, the baseline workflow relied on a provider first completing a sexual history and making a clinical judgment to assess need for STI testing. This workflow was complex and time intensive which resulted in missing testing and infections due to unreliable reporting of sexual history. Additionally, the decision to obtain a test was often made under the time pressures of a busy primary care clinic session without a full understanding of patient risk, which may further increase implicit bias. Opt-out testing created a streamlined and equitable approach and assured that testing did not rely on a provider appropriately assessing risk or patients accurately disclosing their sexual histories.
While the opt-out testing program did not eliminate inequities in testing, we found a significant reduction in inequities between patients of different sexes, language preferences, and insurance types with this approach. In alignment with the opt-out testing pilot sites mentioned in the background, data on race indicated a trend toward more equitable care. Based on data from the 2 pilot sites at DHHA, we anticipate seeing a further reduction in inequities by race and ethnicity in the future. For example, the pediatric pilot site demonstrated a 27% absolute reduction in the difference in testing rates between patients who were White and those who identified as Black or African American from January 2019 to July 2021. 23 Thus, opt-out testing may be a valuable tool to prevent stigmatization of minority groups that have historically been disproportionately tested for STIs.4,10,11
Though we did not randomize clinics to opt-out versus risk-based testing approaches the data strongly suggest that universal testing detected significantly more CT and GC cases than risk-based testing. This is evident by the increased number of cases detected seen during the intervention period compared to the baseline and test shortage time periods. Additionally, public health data from the Denver metropolitan area indicate that the community prevalence did not change considerably during these time periods. Ultimately, we expect the prevalence in our population to fall as we continue to improve prevention, detection, and treatment of CT and GC.
This evaluation has several strengths including the ability to analyze the effectiveness of the program across a large multi-specialty system that serves a diverse population. Given the high number of patients, we could assess for changes in testing inequities related to the intervention. COVID-19 associated test shortages and access to Denver public health data afforded us the opportunity to assess if the changes in cases detected that were observed during the intervention were secondary to changes in community prevalence or the intervention. Additionally, the test shortage period allowed us to evaluate if the reduction in testing inequities was predominantly due to the intervention versus other institutional programs to promote overall equity at DHHA.
Limitations
This evaluation also has several limitations. Because of COVID-19 associated test shortages we had to halt universal screening from September to December 2020. Given the significance of this change, the time-period was analyzed as a separate period in the ITS analysis rather than simply adjusting for month as a confounder. We could not distinguish between testing of symptomatic patients and screening. Because this is a single center evaluation the findings may not be generalizable to other health care systems. However, the population evaluated is similar to the 28 million patients seen annually at other urban FQHCs. 25 Though prior studies have determined that opt-out chlamydia testing is cost-effective and we found that this approach was cost-neutral for our organization and did not result in additional expense for our patients, we urge other systems to carefully examine costs in relations to their primary payers before widespread implementation. We were also not able to compare changes in testing or number of cases detected to a similar, primary care, comparison group without the intervention. Thus, we cannot definitively conclude that the changes observed were secondary to the intervention rather than other internal or external factors. Lastly, we cannot quantify how much provider bias versus other factors (eg, patient opt-out rates) contributed to testing inequities.
Conclusions
In conclusion, CT and GC testing rates were significantly improved with an opt-out testing approach. Though the approach improved testing in all settings, it was most effective in pediatric clinics. Opt-out testing significantly reduced health inequities and resulted in a higher detection of CT and GC cases compared to risk-based screening. Given rapidly increasing STI rates along with persistent inequities with current screening recommendations innovative approaches are needed. Universal, opt-out testing is a pragmatic approach that effectively improves testing rates.
Supplemental Material
sj-doc-1-jpc-10.1177_21501319221131382 – Supplemental material for Closing the Equity Gap: An Intervention to Improve Chlamydia and Gonorrhea Testing for Adolescents and Young Adults in Primary Care
Supplemental material, sj-doc-1-jpc-10.1177_21501319221131382 for Closing the Equity Gap: An Intervention to Improve Chlamydia and Gonorrhea Testing for Adolescents and Young Adults in Primary Care by Margaret M. Tomcho, Yingbo Lou, Sonja C. O’Leary, Deborah J. Rinehart, Tara Thomas-Gale, Lara Penny and Holly M. Frost in Journal of Primary Care & Community Health
Footnotes
Abbreviations
CT-chlamydia
DHHA-Denver health and hospital authority
EHR-electronic health record
FQHC-federally qualified healthcare center
GC-gonorrhea
MSM-men who have sex with men
NAAT-nucleic acid amplification test
STI-sexually transmitted infection
Author Contributions
Tomcho-Conceptualized and designed the evaluation, assisted with the initial analysis, drafted the initial manuscript, and reviewed and revised the manuscript. Lou-Conceptualized and designed the evaluation, completed the data analysis, assisted with data interpretation, and critically reviewed and revised the manuscript. O’Leary-Conceptualized and designed the evaluation, assisted with data interpretation, and critically reviewed and revised the manuscript. Rinehart-Assisted with data analysis and interpretation and critically reviewed and revised the manuscript. Thomas-Gale-Assisted with data analysis and interpretation and critically reviewed and revised the manuscript. Penny-Assisted with evaluation design and data interpretation and critically reviewed and revised the manuscript. Frost-Conceptualized and designed the evaluation, assisted with data interpretation, and critically reviewed and revised the manuscript. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: H.F received salary support from the Eunice Kennedy Shriver National Institute of Child Health & Human Development of the National Institutes of Health under Award Number K23HD099925. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Role of the Funder
The funder/sponsor did not participate in the work.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

