Abstract
Introduction
Lyme disease is the sixth most common Nationally Notifiable Disease in the United States.1,2 There were approximately 24,000 confirmed cases and almost 9,000 additional probable cases reported to the Centers for Disease Control and Prevention (CDC) in 2011. Recent data from the CDC indicates that there are an estimated 300,000 people in the United States diagnosed with Lyme disease each year. 3 Many physicians and patients are familiar with acute Lyme disease and the erythema migrans or “bulls-eye” rash, but the existence of persistent symptoms associated with Lyme disease is often debated. The controversy over the existence of the persistent symptoms is exacerbated by the lack of a standard clinical or microbiologic definition of the disease, as well as the commonality of persistent symptoms in the general population. 4 To date, there is no systematic evidence that the Lyme disease–causing bacteria, Borrelia burgdorferi, can be identified in patients with persistent symptoms following appropriate treatment for Lyme disease. Therefore, while many patients present with these symptoms, most physicians believe that persistent symptoms, sometimes referred to as “chronic Lyme disease,” are due to other causes. The CDC acknowledges that a subset of patients who are treated appropriately, particularly those diagnosed in later stages of disease, may experience limited persistent or recurrent symptoms. 5
To address the public health need for more research on Lyme disease and its persistent symptoms, Western Connecticut Health Network (WCHN) developed the Lyme Disease Registry, the only hospital-based Lyme Disease Registry in the United States. Guided by a community-based participatory research (CBPR) process, the WCHN Lyme Disease Registry was created in collaboration with a local tick-borne disease task force composed of community members affected by Lyme disease. The registry’s goal and design was created together with this group and its members are a critical source of patient referrals. As such, the goal of the registry is to provide meaningful data about Lyme disease to be an objective source of data for researchers. CBPR approach has maximized the community’s experience to produce a successful registry.
The primary objectives of the WCHN Lyme Disease Registry are to clinically define the different stages of Lyme disease, develop better diagnostic assays and investigate the cause of Lyme disease’s reported persistent effects. Broad inclusion criteria are designed to encourage and support enrollment of patients who are typically excluded from Lyme disease studies, in addition to patients with acute cases. With a large and widely inclusive sample size, the registry is able to identify patterns among patients diagnosed with Lyme disease at all stages. The CBPR focus of the Lyme Disease Registry and the collection of blood samples enable researchers to combine, epidemiologic, clinical, and biomedical data to help answer novel research questions. The purpose of this study was to identify a core set of symptoms common to patients who report experiencing persistent symptoms due to Lyme disease and to compare these symptoms to those experienced by patients with an acute infection. In turn, these data could help primary care and other specialty providers recognize the nonspecific symptoms that may be associated with multiple stages of Lyme disease. A provider may then consider testing for Lyme disease among other standard tests to help provide a diagnosis for the patient.
Methods
The WCHN Lyme Disease Registry includes patients at least 5 years old who have ever had a Lyme diagnosis documented by a health care provider. Patients must complete the initial enrollment appointment at one of the registry’s locations in southwest Connecticut. Verification of a Lyme disease diagnosis includes copies of positive blood work reports and/or documentation in the patient’s medical record of a Lyme disease clinical diagnosis. Based on this information, participants are categorized as either acute (diagnosis within the past 6 months and still symptomatic), recovered (diagnosis at any point but no longer symptomatic), or persistently symptomatic (diagnosis more than 6 months ago and still symptomatic).
At the initial enrollment appointment, an extensive survey pertaining to the patient’s history with Lyme disease is administered. Information about the patient’s Lyme diagnosis (or diagnoses), treatment, symptoms, and laboratory testing, if applicable, is collected. The intake questionnaire incorporates Quality Metric’s SF-12v2 Health Survey, a 12-question survey that measures functional health and well-being from the patient’s point of view. 6 This information is the patient’s baseline health status for the Registry and reflects his/her health status at the time of their intake. At the initial and only visit, a blood sample is also taken from all participants and is banked for future research.
Following the initial appointment, participants are asked to complete a short follow-up survey every six months by mail or e-mail. This follow-up survey asks patients to update their health information pertaining to the prior 6 months, including symptoms the participant may have experienced, new diagnoses of Lyme disease, and any treatment for Lyme disease the participant may have completed. The 6-month follow-up survey incorporates the SF-12v2 for comparison to the baseline. Completion of these follow-up surveys enables longitudinal study of participants’ Lyme disease history and general physical and mental health. If necessary, up to 4 attempts are made to contact each participant for completion a given 6-month follow-up form. Should a participant fail to respond to 2 consecutive follow-up periods, no additional attempts are made to contact that participant.
All initial intake and follow-up data are recorded using Microsoft InfoPath. Given that privacy is of the upmost concern, all data are encrypted and saved on a designated registry server with access granted to study staff only. Using the InfoPath system, deidentified data are easily extrapolated and viewed using any statistical software package. JMP v.9.0 was used to conduct statistical analysis. Wilcoxon rank sum test was used for continuous variables and Pearson’s χ2 test was used for categorical variables. Statistical significance was set at P < .05.
Results
As of January 2014, the registry has a total of 256 participants, representing the 3 previously defined categories of Lyme disease. Based on categorization at the time of enrollment, approximately one quarter of participants are acutely infected, 31% have recovered from Lyme disease, and 43% are experiencing persistent symptoms associated with Lyme disease (Table 1). Of the 256 consented patients, 98 have been lost to follow-up. There were no statistically significant differences between the acute and persistently symptomatic patients for age and gender.
Western Connecticut Health Network Lyme Disease Registry Participant Demographics.
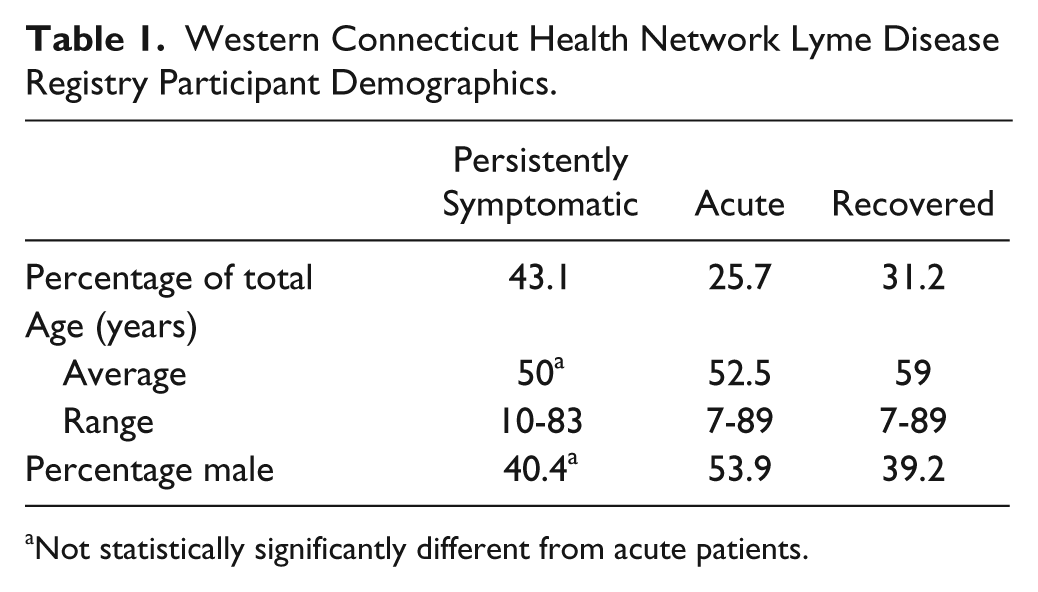
Not statistically significantly different from acute patients.
As part of the initial intake survey, registrants were asked about symptoms they experienced during the course of their disease. Table 2 displays the top 20 symptoms reported as “current” at the time of enrollment. The percentages are reported as the number of patients within the given disease state that experience the described symptom. The only symptoms that do not overlap from the persistent symptoms group to the acute are marked with superscript “a” and include mood swings, difficulty in word finding, confusion, and inattention. More notable is that the remaining 16 persistent symptoms are consistent and prominent throughout both stages of disease.
Top 20 Current Symptoms as Reported by Persistently Symptomatic Patients and Associated Percentage of Acute Patients.

Represents symptoms not reported in the Acute Top 20.
Discussion
Since the inception of the WCHN Lyme Disease Registry, the impact on the institution and community has been strong. With greater recruitment, we are able to more confidently report registrant symptoms and describe the overlap and discrepancies. Our current reported data suggest there is not a large discrepancy of symptoms between the acute and persistently symptomatic patients. Previous reports of persistent Lyme disease symptoms include the evolution of neurologic and psychological deficits, findings that our data do not strongly favor.7,8 The symptoms among our persistent patients, however, seem to be more similar to those reported by our acute patients. In fact, more than half of acute patients reported symptoms persisting for at least 6 months to a year post enrollment, with 45% reporting persistent sleep problems and approximately three quarters of patients reporting persistent cognitive deficits (data in the appendix). Alternatively, symptoms such as Bell’s palsy and fever were resolved by six months post enrollment. Continued recruitment will help to provide a more precise case definition for the different stages of Lyme disease. This, in turn would help guide health care providers to make evidence-based diagnosis and treatment decisions.
The WCHN Lyme Disease Registry’s design serves as a model for other institutions. Hospital-based disease registries provide detailed access to acutely and chronically ill patients not available elsewhere. Typically, hospital-based registries are limited to data reflecting experiences occurring within the institution. The data produced by hospital-based registries often experience socioeconomic biases because of differences in patient access to health care. 9 The WCHN Lyme Disease Registry is thus population-based, enrolling patients diagnosed with Lyme disease by any qualified clinician within the community.
The prospective tracking of patients combined with guidance of targeted research questions from the affected community results in data that are more easily translated into public health action. The community input is highly valuable and can provide observations that may not be as inherent to a clinical researcher. Furthermore, the collaborative nature of CBPR projects enhances an institution’s relationship with its surrounding community and help to support patient recruitment.
Footnotes
Appendix
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Western Connecticut Health Network.
