Abstract
Rational medication use means taking medication appropriately for curing and relieving the symptoms of disease on the basis of evidence and sound judgment. We compare US policy experience on rational use of medications with the World Health Organization (WHO) list of interventions designed to promote such use. Current US performance and educational, managerial, and regulatory interventions to improve it are discussed. We conclude that, while most of the WHO guidelines for rational medication use are practiced in some form in one or more of the various US health care subsystems today, overall performance based on outcomes is not comparable with that of other industrialized countries. This is due to the absence of a national drug policy, the presence of a few strong stakeholders with committed policy preferences, and the altogether fragmented character of the US state and federal health systems. Practical suggestions are offered as to how the US could improve its overall less-than-optimal policies on rational medication use.
Introduction
The 2008 Health Data from the Organization for Economic Co-Operation and Development (OECD) indicates that the US spends $843.00 per capita on pharmaceutical products, more than any of the other 30 countries in the database; however, the US is still below the median for all the major health status outcomes of importance. 1 The rational use of medications is aligned with effectively achieving the goal of curing, relieving the symptoms of disease, or the goal of prevention and prophylaxis, through therapy based on scientific evidence and well-studied clinical guidelines. 2 It aims at broad, appropriate use of medications that are effective, proven, available, affordable, easily administered, conveniently stored, and with few dangerous side effects. 3 The World Health Organization (WHO) has encouraged policies for rational medication use for the past 20 years. It developed the first Model List of Essential Medications in 1977 to assist in the creation of formularies within independent countries. The current WHO definition of rational use of medications was developed in 1985. It states, “the rational use of medicines requires that patients receive medications appropriate to their clinical needs, in doses that meet their own requirements, for an adequate period of time and at the lowest cost to them and their community”. 4
The rational use of medication receives little attention in the US from a national perspective, but it is becoming more important as health care costs continue to soar, affecting economic growth in this country and as the US debates health care reform. 5 Interest in the rational use of medications comes from diverse directions, such as insurance companies, physician groups, politicians, and patients. 6 In the census bureau report published in 2007, for the sixth consecutive year, the number of Americans living without health insurance has risen, to the largest number ever, 47 million people or 15.8% of the US population. 7 Addressing the pharmaceutical needs of this population, in addition to those with inadequate insurance coverage, will result in future growth in the public expenditures on medications. The implementation of Medicare Part D for pharmaceutical benefit for the elderly illustrates this trend. 8 Although some of the price difference is due to income disparities, there are studies which indicate that the higher spending on pharmaceutical products per capita is not wholly dependent on increased per capita income or per capita costs. 9
WHO advocates 12 interventions to promote increased rational use of medications, as listed in Table 1; these serve as the criteria for this assessment of US rational medication use practices. 10 These interventions are educational, managerial, and/or regulatory, in nature. This study reviews and evaluates specific elements of each interventional measure to elucidate the extent and impact of these measures in the US and to determine if the goal of rational medication use is attained in the US.
Areas of Attempted Interventions to Increase Rational Medication Use in the United States
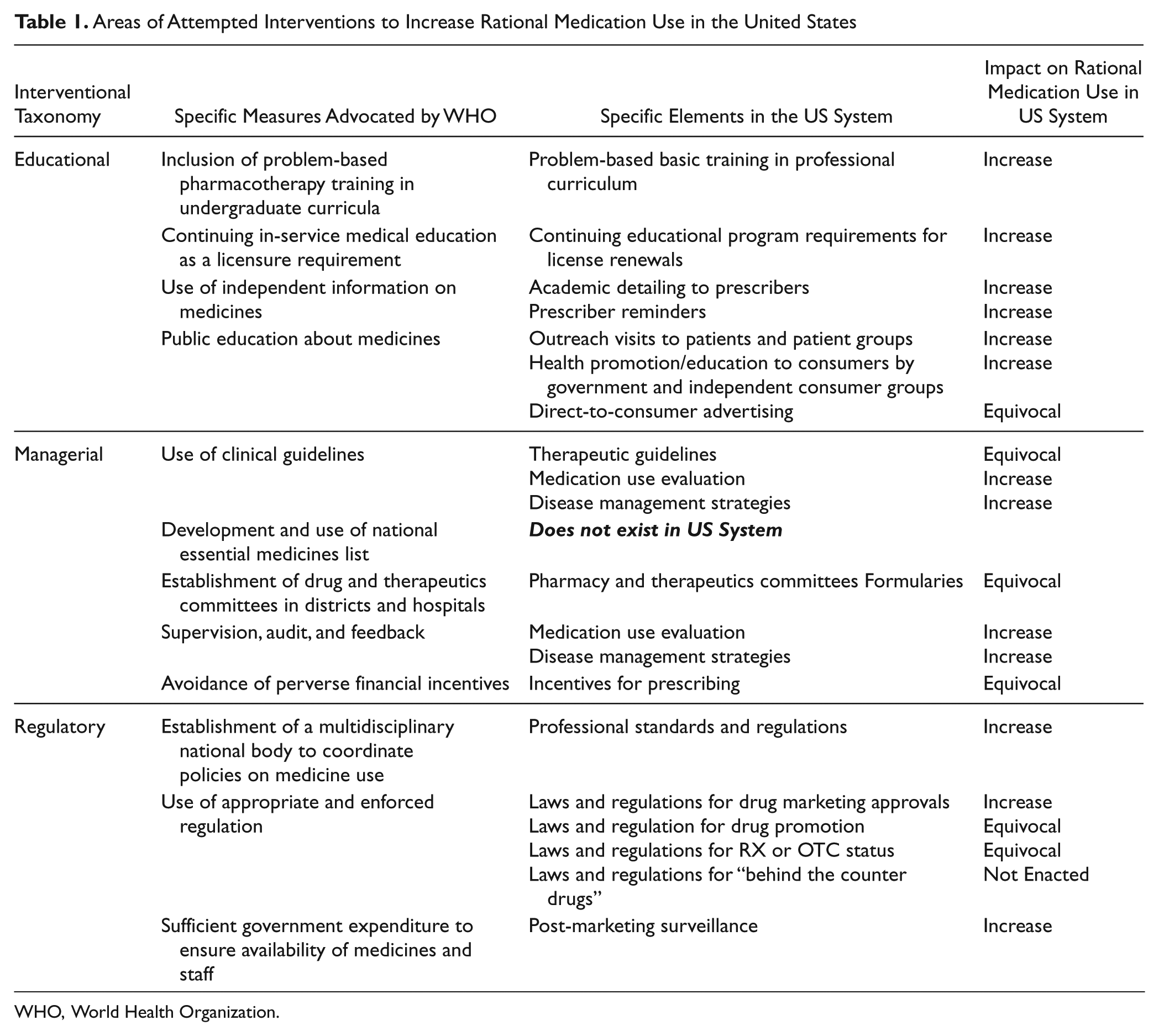

WHO, World Health Organization.
Interventions to Promote Rational Medication Use in the United States
Educational Measures
Educational measures to increase the rational use of medication may target professionals, the general population of patients and consumers, or both, at the same time. The interventions include academic detailing or outreach visits; prescriber reminders; health promotion/education to consumers through various media, including direct advertising; problem-based basic professional training; and continuing education. For example, with its “Get Smart campaign” the Centers for Disease Control and Prevention promotes “appropriate prescribing guidelines among providers” at the same time that it tries to educate the public to refrain from demanding inappropriate antibiotics from their doctor. 11
Evidence indicates the positive impact of educational methodologies in many, but not all situations, whether they target the health care professional or the patient. A study by Juzych et al evaluated the impact of an interactive case-based educational program for physicians and staff on the management of acute upper respiratory tract infection in adult and pediatric patients. Twenty-one primary care physicians at clinics where the educational program was implemented were compared with a group of physicians at 9 primary care clinics where no educational program was implemented in Detroit, Michigan.
12
The antimicrobial prescribing among the intervention group of physicians decreased 24.6% from the baseline to the postintervention period (
Medical schools are also now focused on teaching rational prescribing through problem-based learning (PBL) modules; courses are specifically designed to compare recently approved medications with currently available therapy in terms of efficacy, safety, and cost. 14 The American Association of Colleges of Pharmacy (AACP) has indicated that the future of pharmacy lies in the ability of pharmacists to contribute to the rational use of medications in health care and has created curricula with outcomes centered on this goal. 15
Direct-to-consumer advertising (DTCA) by pharmaceutical companies, until the last year, has been the fastest growing source of drug information for consumers in the US, even outstripping promotion to physicians, but its educational impact is a double-edged sword. 16 Some suggest that it increases communication between patient and prescriber regarding drug therapy and increases the awareness of medications as a treatment option, but it also increases demand for the advertised medications and not all of these were appropriate to the patient’s condition. 17 A review of largely qualitative evidence on direct-to-consumer advertisements indicates that it is “unlikely that advertising will serve an unbiased and strictly informative function.” 18 A survey conducted by Kaiser Family Foundation and the Harvard School of Public Health in January of 2008, found that prescription drug advertisements prompt nearly one third of US adults to ask their physicians about advertised medications, and 82% of those who ask are given a prescription. 19 Spending for DTCA increased from $1.1 billion in 1997 to approximately $2.7 billion in 2001, an average annual increase of 28% and a high of $4.7 billion in 2007.20,21 Studies have indicated that, on average, a 10% increase in DTCA of drugs within a class results in a 1% increase in sales of drugs in that class, an advertising elasticity of 0.10. 22 The group of drugs known as proton pump inhibitors (PPIs) is a good example. Between 1998 and 1999, DTCA spending for PPIs increased by 60% (from $49.7 million to $80.1 million) and PPI sales subsequently increased 36% (from $4.2 billion to $5.7 billion). Application of the estimated elasticity of 0.10 to the results indicates that approximately $252 million, or 17% of the 36% increase, was attributable to DTCA. So, though DTCA is not the primary driver of growth in prescription drug spending, it produces a significant return of $4.20 in sales for every $1.00 spent. 20 The World Health Organization’s stand is that DTCA poses an inherent conflict of interest between the business goals of the pharmaceutical companies and the medical and economic needs of the providers and the patients to select and use drugs in the most rational way.23,24
In 2008, the pharmaceutical industry spent $4.4 billion on direct-to-consumer ads, whereas the public health system had little or no money to educate the lay public. The US Food and Drug Administration (FDA) Division of Drug Marketing, Advertising, and Communications, the regulatory division responsible for reviewing the accuracy of claims, had a budget of only approximately $2 million for the same period. 25 Congressional efforts to increase drug safety by banning DTCA during the initial 2-year post-FDA approval period for a new product, which would allow time for accumulation of safety data in real-world usage, have failed repeatedly. These legislative attempts are controversial because advertising is viewed as a form of free speech protected by the First Amendment. Precedence in the court indicates that even if the legislative process is adopted, it may not survive judicial review. 26
The current delivery of pharmaceutical education in a fragmented health care delivery system with mismatched funding by the interested stakeholders, leads to an educational process that is patchy at best, with limited impact, extent, and outcomes.
Managerial Measures
Managerial measures provide infrastructure to the health care environment to facilitate rational use of medications; they are often combined with educational measures to reinforce the impact of the change. Managerial measures include therapeutic guidelines, pharmacy and therapeutics (P&T) committees, institutional and/or managed care formularies, medication use evaluations, disease management strategies, and incentives for prescribing. Clinical guidelines have been defined as “systematically designed statements to assist practitioner and patient decisions about appropriate health care for particular clinical circumstances”.27–29 The National Guideline Clearinghouse (NGC) is a comprehensive database of evidence-based clinical practice guidelines and related documents. NGC is an initiative of the Agency for Health care Research and Quality (AHRQ), an agency of the US Department of Health and Human Services, that was originally created by AHRQ in partnership with the American Medical Association and the American Association of Health Plans (now America’s Health Insurance Plans or AHIP). The database contains over 2000 separate guideline summaries, the majority of which focus on pharmaceutical therapies for specific medical conditions. 30 Publication of other internet resources and manuals has created a massive guideline industry in the US; these guidelines serve as tools for clinicians, patients, and regulatory administrators and third-party insurance payers.31,32 Clinicians, payers, and policy makers view guidelines as a tool for making consistent, efficient, and rational choices and for closely tying evidence to clinical care, and is a popular public research agenda in the US today. However, 2 challenges facing the development and utilization of translation-based guidelines are (1) incorporation of systemic evidence on which to base the development of guidelines and (2) identification of the most effective method(s) for implementation of guidelines in practice. Scientific evidence for guideline development may be lacking, misleading, or misinterpreted; recommendations may be influenced by opinions and the experience of the guideline development group; and the group may have priorities other than patient needs, such as controlling costs or protecting special interests. Stakeholders do influence guidelines and this is an important conflict of interest situation in the US. 32 One possible area for improvement would be a required addition of comparative information to what is currently available, though this may entail additional regulatory authority for the FDA to mandate. 25 The Obama administration has included more funding for comparative effectiveness research, which could add to and improve clinical guidelines in the US. 33 Also, no single effective method is in place for guideline dissemination and implementation. 34 A combination of management strategies targeted at limiting the barriers to implementation of guidelines exist, aimed at the individual prescriber, the peer group, or the medical organization, including the educational methods discussed above. 35
P&T committees, comprising clinicians, pharmacists, administrators, and other stakeholders, are primarily held responsible for creating and managing formularies and medication use evaluations in institutions in the US Managed care and the government Medicaid and Medicare entities also use formularies and medication use evaluations to guide and promote appropriate usage of medications. 36 It is very similar to essential drug lists used by most countries with universal health care coverage, except it focuses on smaller groups and institutions. A formulary system encourages health care practitioners to serve the health interests of a given patient population by establishing policies about use of drug products and therapies, and identifying the most medically appropriate and cost-effective interventions, based on local practices identified by medication use evaluations, scientific evidence-based clinical guidelines, and pharmacoeconomic considerations. 37 Here, again, forces within and outside the health care entities will influence the selection of agents to be included and managed by formulary systems. Medication use evaluations, or drug utilization reviews, are structured initiatives that evaluate patterns of medication use in relation to predetermined criteria and attempt to minimize inappropriate prescribing. 38 Conflicting data on their impact on clinical outcomes indicate that more work must be done to see if these mechanisms improve rational medication use. 39
Therefore, the strength of evidence to create guidelines is lacking, misleading, or misunderstood, creating opportunities for personal preferences and priorities. No single, effective method exists for guideline dissemination. Forces within and outside the health care system influence the selection of agents to be included and managed by formulary systems, which are local in focus, health care system by health care system. Structured initiatives such as medication use evaluation attempt to minimize the inappropriate use; however they are limited in scope and extend.
Regulatory Measures
The FDA is responsible to the general public and can be held accountable for the rational use of drugs. Through the approval process for drugs, the FDA has the long arm to reach large segments of the population and influence drug choice, though current legislation limits its role to determining which drugs are safe and effective; no determination of cost effectiveness is made at the time of approval.
The process of transferring FDA-approved prescription medications to nonprescription, over-the-counter (OTC) status is known as “Rx-to-OTC switch.” This process provides consumers with easy access to safe and effective products without making a visit to a health care professional. The 1951 Durham–Humphrey Amendment provided the FDA with a statutory basis and specific criteria for differentiating prescription from OTC drugs. 40
OTC medication is a very cost-effective first-line treatment for many diseases, but availability of a medication OTC can either increase or decrease its rational use. Currently, over 90 previously prescription-only medications have been approved for use as self-care or OTC products. 41 Examples include antihistamine agents for allergies, histamine-2 blockers, and PPIs for heartburn, antifungals for vaginal infections, and nonsteroidal antiinflammatory agents for pain and fever. 42 The total cost of acquiring a prescription medication may include the professional office visit, travel time and costs, and decreased productivity from missed work or school time, in addition to the costs associated with the medication. OTC availability decreases the cost in most incidences to only the purchase cost of the product, which is usually approximately 50% of the total cost of the same product by prescription. OTC availability has both advantages and disadvantages because increased availability of medications without practitioner oversight may lead to nonrational use of the drug, including overuse, abuse, and inappropriate use, possibly even delaying treatment for potentially serious conditions. 43
The FDA is currently exploring the concept of a third class of medications in the US, called “behind the counter (BTC) drugs,” which would be available to patients without a prescription, but through consultation with and intervention by a pharmacist. 44 In this scenario, the cost reduction benefits of OTC access are maintained, while the issues of rational selection and patient safety are addressed by the pharmacist. The Government Accountability Office (GAO) recently released a study that evaluated and compared the BTC availability and impact in 5 countries: Italy, Netherlands, Australia, United Kingdom, and United States. The study found that if the pharmacists had access to the patient’s clinical and laboratory information, they were willing to take the initiative to recommend these drugs. Without this clinical information, the benefits of BTC medications are not actualized. 45 While pharmacists are in favor of the shift to BTC drugs, the American Medical Association is against the move, seeing it not only as an infringement of its autonomy, but also as a potential risk to patient safety.46,47
In another regulatory change, the FDA has also issued new guidelines that enable distribution of information of off-label uses of approved drugs and devices to health care professionals, by using peer-reviewed literature. 48 Although the information may serve as an educational tool, there is potential for bias to be introduced due to the fact there is a tendency for pharmaceutical sponsors to avoid publishing null or unfavorable findings. 49 Concern exits as to whether this guidance is in line with the FDA mission of protecting the public health by ensuring the efficacy and safety of medication because it precludes the FDA review process for acquiring a new indication. There is a need to foster a more active role of the FDA in promoting evidence-based practice, requiring a fair and balanced presentation of evidence, and collecting and monitoring postmarketing data to quantify the harms or benefits of off-label use. 50
Conclusion
The absence of a US national drug policy (including an essential drug list), the presence of a few strong stakeholders with committed policy preferences, and the heterogeneous and fragmented character of the US health system all contribute to the lack of rational medication use policies and the ineffectiveness of those policies that do exist. The need to develop a more universal rational medication use policy in the US is evident if the costs of medication utilization in the US are to be contained. Table 2 lists our recommendations which could lead to increased rational medication use in the US.
Recommendations to Increase Rational Medication Use in US


In the US, measures are available for addressing the issues involved in developing rational drug policies within the private and public sectors, through consumer, health care, pharmaceutical industry, professional, and government entities. A strong, independent, and adequately funded FDA is a critical element in formulating and implementing a viable rational medication use policy in the US The FDA is also, in turn, affected by the conflicting interests of policy makers, payers, consumers, pharmaceutical industry, and health care professionals, and is limited by the funding available. It is also imperative that appropriate and necessary information is available for the determination of rational medication use. Research on comparative effectiveness of new agents to standard of care therapies or comparison of existing therapeutic modalities is necessary to determine which treatment is the best option. Also, research on cost effectiveness of alternative treatment regimens are necessary to determine the cost per value gained, especially for newer treatment modalities, such as monoclonal antibodies. Increased research into outcome measurements on medication use and disease management can provide insight on best practices. Furthermore, incorporation of outcome or quality measurements into reimbursement strategies will lead to further benefit in the overall rational use of medications.
We believe another strategy for promoting rational medication use policy is to enhance the role of pharmacists in the US health system. The introduction of a new “behind-the-counter” class of medications would decrease the potential for abuse or misuse that exists with pure OTC products because there would be a health care professional involved in the decision making process of choosing a medication, and may provide more rational choices for treatment, especially for the uninsured population, who may not have access to other health care providers. Pharmacists’ roles and responsibilities must be reassessed and enhanced to enable them to play the role for which they are trained that is, they must have access to the necessary medical information and be reimbursed for their pharmaceutical care activities. 45
The overriding objective of interest for all concerned must be to promote more rational use of medications in the US, leading to more efficient and more efficacious patient outcomes. Increasing and improving rational use of medication in the US is a worthy goal that is likely to see progress in the coming decade. This is, in good part, because of the commitment to education, ongoing improvement in medication management, and improved transparency in regulation.
Footnotes
Acknowledgements
Presented in part at the 136th American Public Health Association Annual Meeting & Exposition, San Diego, CA. October 25-29, 2008.
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
