Abstract
Encephalitis is an acute inflammatory process of the brain parenchyma, which is often caused by viral infection. It is an vital cause of acute symptomatic seizures and subsequent epilepsy. The incidence of unprovoked and recurrent seizures after previous infections of the central nervous system is high and accounts for 1%~5% of the cases of epilepsy. Viral encephalitis (VE) is directly caused by viral infection. The occurrence of seizures after VE is associated with poor prognosis. In survivors of VE, among other neurological sequelae, the risk of developing epilepsy is increased 10-fold. The risk of severe neurological sequelae after VE is particularly high in very young children. Studies on seizure occurrence, possible underlying mechanisms, clinical characteristics, and clinical treatment (especially surgical treatment) of VE have yielded only limited detailed data. We reviewed the most recent literature on the clinical features and surgical treatment of post-VE epilepsy.
Keywords
1 Introduction
The term “encephalitis” refers to a broad range of pathophysiologic processes that result from inflammation of the brain parenchyma, most often caused by a viral infection [1 –3]. Several risk factors for the development of chronic epilepsy after cerebral infection have been identified in certain subgroups of patients with epilepsy after cerebral infectionencouraging. However, literature describing primarily the clinical characteristics and surgical treatment of epilepsy after viral encephalitis (VE) is limited [4 –7]. VE is an acute inflammatory process of the brain parenchyma that is directly caused by viral infection [8 –10]. It is generally an acute monophasic disease in which late relapses are uncommon [11]. Clinical manifestations of VE are nonspecific, but patients with postencephalitic epilepsy usually develop intractable and multifocal seizures [2].
The burden of refractory epilepsy associated with VE is well known and multifaceted; in addition to neuronal structural injury and atrophy over time, affected patients have difficulty navigating physical obstacles, are at an increased risk of mortality, and may experience adverse effects of the long-term use of antiepileptic drugs [12, 13]. Prospective studies are required to document the risk of late unprovoked seizures and post-VE epilepsy, as well as to determine the clinical characteristics, course, treatment, and outcome of postencephalitic epilepsy. The epilepsy associated with VE is often drug-resistant, and several cases of post-VE epilepsy are not surgically evaluated because of the difficulties in precisely localizing seizures; therefore, many patients do not receive surgical treatment [14]. However, in certain subgroups of patients with epilepsy, the surgical outcomes after VE have been encouraging; nonetheless, some surgical outcomes have been generally unsatisfactory [15, 16]. For these reasons, we reviewed the literature regarding VE epilepsy in association with VE, primarily focusing on the clinical features of the disease and on the outcomes of surgical treatment. We hope to draw attention to this condition and provide guidance for future treatment.
2 Diagnosis, epidemiology, and patho-genesis of epilepsy after viral encephalitis
2.1 Diagnosis and epidemiology
Specific viral diagnosis can be confirmed either by the demonstration of viral nucleic acid or antibody in cerebrospinal fluid or by isolation of the virus from cerebrospinal fluid or brain tissue [17, 18]. Serological tests are simpler to perform and widely available for viral diagnosis. During active infection, antibody titers against the virus are elevated in the serum or cerebrospinal fluid; the demonstration of a fourfold rise reliably confirms a diagnosis of acute VE [19]. Even with best efforts, however, clinically suspected VE remains undiagnosed in 30%~60% of affected patients [4].
Because of the limitations in viral diagnostic tests, the specific cause of VE is unknown. There is presently no consensus definition of posten-cephalitic epilepsy [20]. According to most researchers, the ongoing requirement for antie-pileptic drugs is indicative of postencephalitic epilepsy, with variable time thresholds [21, 22]; one specific time threshold is 24 months or longer after acute encephalitis [23].
The number of viruses that can cause encephalitis and subsequent chronic epilepsy is huge. The following viruses have been identified: herpes simplex virus (HSV) in 39%, varicella zoster in 23%, West Nile virus in 19%, Epstein–Barr virus in 6%, human immunodeficiency virus in 3%, and other viruses in 10% [4, 24, 25]. Viruses that cause encephalitis can be divided in two groups on the basis of their epidemiological behavior: those that cause sporadic encephalitis and those that are responsible for epidemics of encephalitis. The incidence of late unprovoked seizures in patients with different types of VE has not been systematically evaluated. HSV encephalitis is the most common sporadic encephalitis [26]; the estimated annual incidence is 1 per 250,000 to 1 per 500,000. One-third of HSV cases occur in children and adolescents, and more than 90% are caused by HSV type 1 [27]. Hospital-based studies have revealed that late unprovoked seizures occur in 40%~65% of patients after an episode of HSV encephalitis [28]. The majority of VE epidemics are caused by arboviruses [29].
2.2 Epilepsy after viral encephalitis: epilep-togenic substrates
Growing evidence in the literature suggests that inflammation plays a role in post-VE epilepto-genesis [30 –32]. Precipitating insults such as viral infections and autoimmune disorders result in the activation of brain parenchymal cells, endothelial cells of the blood-brain barrier, and leukocytes [33]. These cells produce and release inflammatory mediators in the brain. Inflammatory mediators can precipitate neurotransmitter release and activate glutamate receptors, which lead to increased neuronal excitability and exacerbates inflammation; thus, a vicious cycle contributes to the development of epilepsy [34, 35].
Viruses producing encephalitis have certain distinctive features that render them pathogenic in the brain parenchyma. These viruses are essentially neurotropic: they have the ability to invade, infect, and subsequently replicate within the human nervous system [36]. Studies on hippocampal cultures revealed that HSV infection causes a long-lasting reduction in the depolarizing membrane potential, which results in a hyperex-citable neuronal state. Lesions within the limbic circuit and hyperexcitability in the hippocampal CA3 region have been identified as potentially correlated, both morphologically and functionally, with the development of seizures in animal models of HSV encephalitis [37, 38]. Both these findings suggest that the hippocampus is uniquely susceptible. In addition, the necrotizing nature of HSV encephalitis, the extent of leukocyte infiltration, and the degree of cortical involvement may all play a role in the occurrence of seizures.
3 Clinical features of epilepsy after viral encephalitis
3.1 Semiotics
The predominant seizure patterns are unilateral temporal (19%), bilateral-temporal (20%), and extratemporal/multifocal or generalized (52%). Reportedly, 36 patients (86%) reported an aura; limbic-mesial-temporal auras were the most common (64%), followed by neocortical (auditory, vertiginous) and extratemporal auras (visual, somatosensory) in 12% and 21%, respectively [2, 39]. In children, another study showed that post-VE epilepsy often consists of infantile spasms and that the main risk factors for the occurrence of spasms in this context are the early age at the occurrence of encephalitis and the involvement of the insula and mesial-temporal structures [40]. Cognitive abilities in such children are often very poor, and the spasms adversely affect the quality of life. We retrospectively studied 10 patients who suffered from post-VE epilepsy using stereo-electroencephalography (SEEG), complemented by the epileptogenicity index, a quantitative and supplementary tool used to investigate the SEEG features associated with surgical outcomes. All patients had a high frequency(at least 3 times per week) of seizures, manifested primarily as nausea, facial rubefaction, cardiac frequency changes associated with partial tonic-clonic seizures, and other neurovegetative symptoms.
Because epilepsy after HSV is more common than epilepsy caused by other viral agents, peculiarities of this virus that may be relevant to the development of chronic epilepsies have been discussed in more detail [15]. Temporal lobe epilepsy after HSV encephalitis is rarely a pure temporal lobe epilepsy; far more often, it is a “temporal plus” epilepsy [41]. Rates of morbidity and mortality from HSV encephalitis are high partly because of its tropism for epileptic regions, which cause seizures that are difficult to treat, including status epilepticus [42]. The rate of mortality from status epilepticus is as high as 59%, and the rate of associated morbidity, which includes cognitive and memory deficits, is high in survivors [42]. Patients who have seizures or neurological deterioration must be monitored with electroencephalography (EEG). Recurrent or intractable seizures lead to raised intracranial pressure, increased metabolic activity, acidosis, and vasodilatation, which, in turn, further elevates intracranial pressure [43, 44]. Some reports have confirmed that decompressive craniectomy and temporal lobectomy is a potential life-saving measure for the treatment of medically refractory cases of HSV encephalitis [45, 46]. HSV encephalitis and encephalitis caused by other viruses lead to statistically similar rates of post-VE epilepsy.
3.2 Electroencephalography and neuroimaging
Some, but not all, patients develop periodic lateralizing epileptiform discharges at a rate of 2~3 Hz originating from the temporal lobe (Fig. 1). These discharges are not specific for HSV encephalitis, but their presence supports the diagnosis [48]. Trinka [2] studied 42 patients with epilepsy secondary to VE and reported that the interictal abnormalities were restricted to the temporal lobe in 21 (50%), unilateral in 7 cases. The abnormalities were extratemporal only in 1 patient and multifocal or generalized in 14 (33%). In this study, focal slowing was present in 21 (50%) patients; among those patients, the brain regions affected were the temporal lobe in 13 (61.9%), an entire hemisphere in 4 (19.0%), the occipital lobe in 3 (14.3%), and the frontal lobe in 1 (4.8%). An electrographic seizure originated in one or both temporal lobes in 12 patients, unilaterally in 5 of them; in 2 patients, the onset was extratemporal, and in 15, the onset was multifocal or generalized.
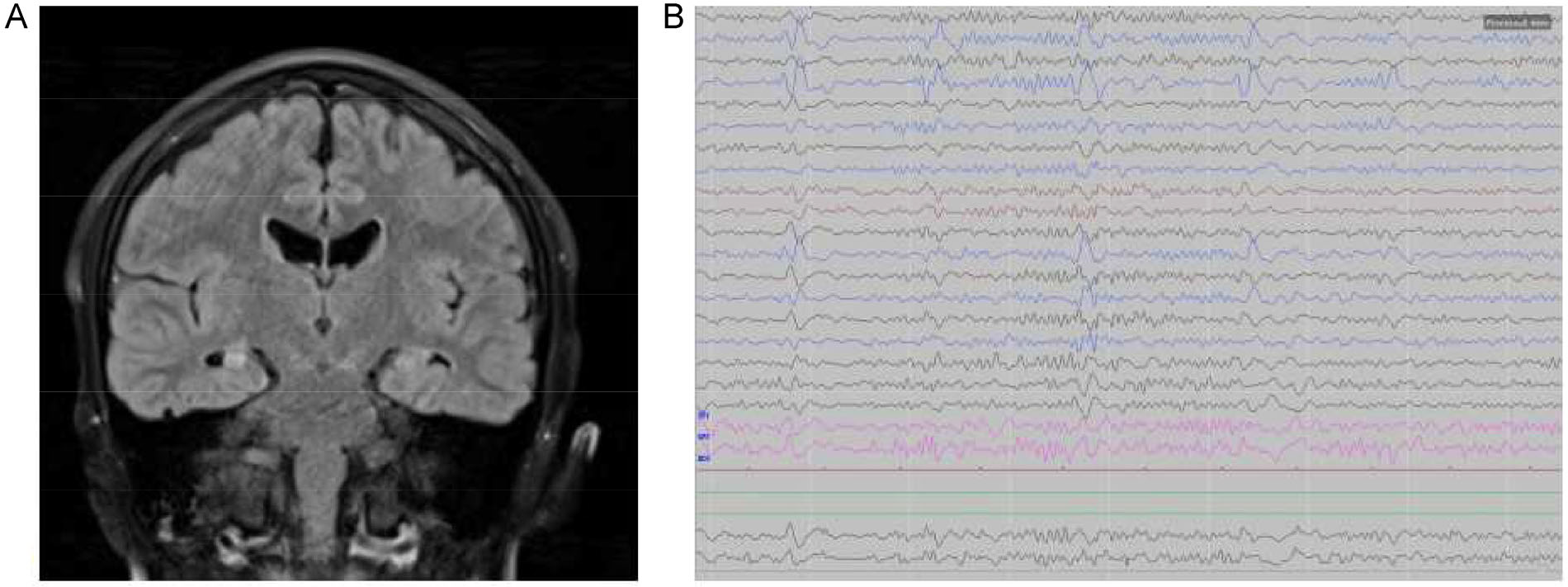
Typical MRI and EEG findings in acute HSE. (A) Coronal FLAIR MRI showing bilateral hippocampal sclerosis. (B) Periodic lateralized epileptiform discharges (PLEDs) over the right temporal lobe.
The virus may gain access to the olfactory bulbs through the nose and may subsequently spread via the olfactory pathway to the orbitofrontal and mesial-temporal lobes [49] (Fig. 1). The virus lies dormant in the anterior and middle cranial fossae, but from this location, it has easy access to the frontotemporal cortices [50]. The distribution of acute changes on magnetic resonance imaging (MRI) matches the distribution of cortical scars, subcortical white matter changes, and atrophic changes in patients with drug-resistant epilepsy after HSV encephalitis [23]. Reportedly, unilateral changes were seen in the temporal lobes in 88% of patients, in the insular cortex in 70%, in the frontal lobes in 68%, and in the thalamus in 28%, whereas bilateral changes were observed in almost 50% of the patients [15]. The HSV has an affinity for the basal frontal and mesial-temporal (limbic) cortices and spares most of the remaining cerebral cortex, the deep gray matter nuclei, and the white matter. The typical MRI abnormality in HSV encephalitis is the presence of asymmetric hyperintensity on fluid-attenuated inversion recovery imaging that corresponds to edematous changes in the gray matter of the mesial-temporal lobes, inferior frontal lobes, and insula [51, 52]. These changes occur because of the necrotizing properties of the viruses, and post-VE epilepsy is bound to arise from areas of MRI-visible cortical damage.
4 Surgical treatment of epilepsy after viral encephalitis
VE is treated primarily with acyclovir, and the treatment of seizures in VE is similar to that of any acquired focal or secondarily generalized epilepsy. For acute VE, epileptic activity must be controlled adequately. In many cases, however, post-VE epilepsy is refractory to antiepileptic drugs, and combination therapy or neurosurgery is therefore necessary to attempt to control seizures [8]. Late provoked epilepsy is often refractory to treatment with antiepileptic drugs; Chen et al. [29] reported that in 10 of 11 children with epilepsy after HSV encephalitis, treatment for 3 years or more yielded poor outcomes. Thus epilepsy surgery needs to be considered early in the course. Of the patients undergoing surgery for drug-resistant epilepsy, those with post-VE epilepsy have been only a small minority in epilepsy surgery series. Nevertheless, some patients do benefit from surgery. In a retrospective study of 42 patients who suffered from epilepsy secondary to VE, 24 patients were successfully treated with surgery [2].
4.1 Surgical method
Potential procedures for post-VE epilepsy include anterior temporal lobe resection, neocortical resections, frontal resections, anterior callosotomy, and combinations of these methods [53, 54]. An L-shaped incision was made to expose the frontotemporal and parietal regions [55]. Bick reported a unique case in which anterior temporal lobectomy was successfully used in treating refractory status epilepticus in patients with HSV encephalitis. The temporal cortex was resected 4.5-cm back from the temporal tip according to standard procedure for anterior temporallobectomy (ATL) to minimize risk to language and visual fields [56] (Fig. 2). Surgical resection for epilepsy, including lobectomy or lesionectomy, were beneficial for the resolution of findings on EEG and for cognitive function [57–58].
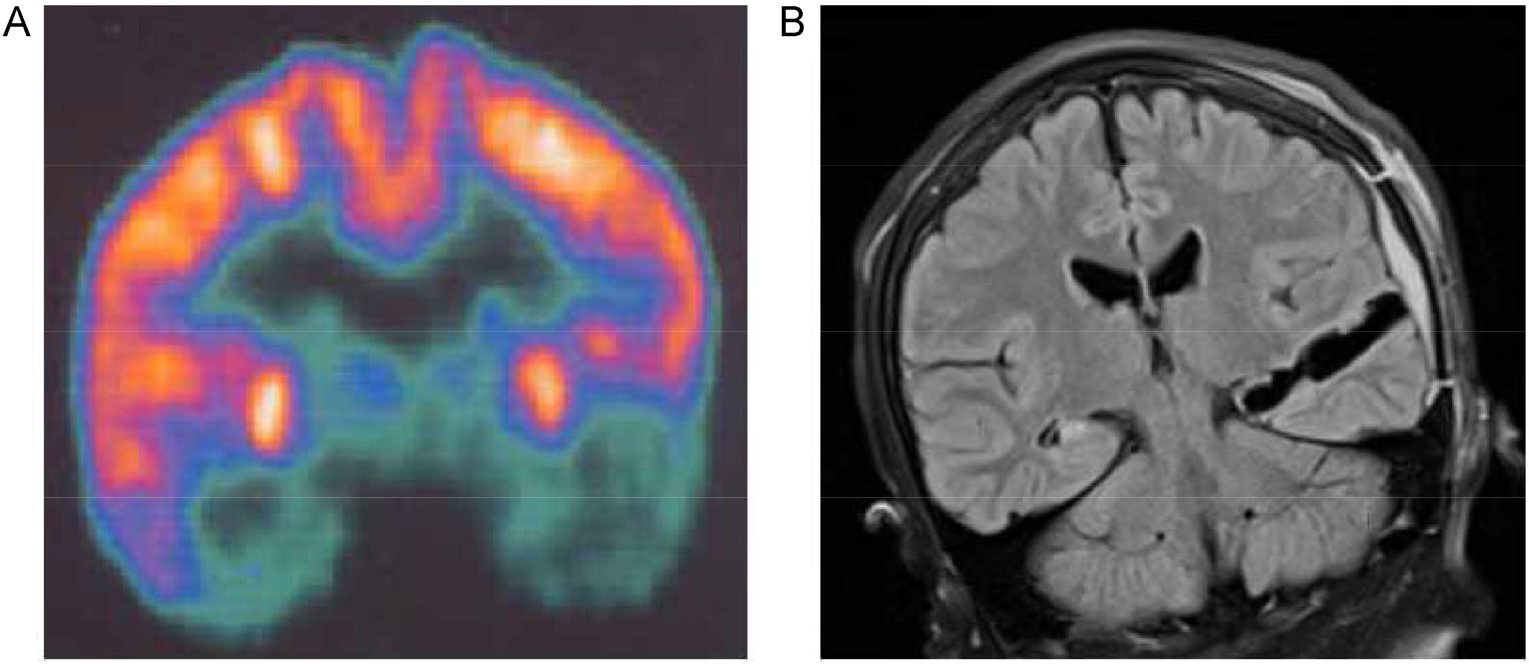
Preoperative positron emission tomography (PET) and postoperative MRI of patient who suffered epilepsy after VE. (A) Preoperative PET demonstrated left temporal lobe hypometabolism. (B) Postoperative coronal image showed left temporal and hippocampus resection.
Surgical procedures involving disconnection, such as hemispherectomy and corpus callosotomy, are effective in dearling with extensive unilateral or bilateral brain abnormalities acquired after VE [59]. Because the effects of VE on the brain are diffuse, surgery is difficult, and noninvasive investigations do not allow a clear definition of the epileptogenic zone (EZ). SEEG is mandatory in these cases, especially in the absence of a clear epileptogenic lesion or diffuse lesions [60]. SEEG is safe and useful in children and adults and is the best option for assessing deep-seated structures. A reasonably lateralized and localized origin of seizures is rarely identified by invasive techniques [61]. Detailed imaging studies using all available approaches may help identify a subgroup of individuals with unilateral temporal lobe epilepsy, which may be expected to respond favorably to surgery.
4.2 Surgical outcome
Because the effect of VE on the brain is diffuse, the surgical outcome was usually unsatisfactory [44]. In severely affected patients with a widespread functional deficit zone, the evidence of bilateral and diffuse disease, and extratemporal clinical features have the benefits of surgery that limited [62]. There is evidence, however, that certain patients have a higher chance for better outcome after epilepsy surgery. In the Montreal series [62], six of the nine patients with unilateral temporal seizure onset had favorable outcomes, and only one patient had a poor outcome. In another study of 42 patients who suffered from epilepsy secondary to VE, 24 patients were surgically treated; the postsurgical outcome was best in those who had unilateral temporal seizures and comparatively poor in those who had bilateral temporal seizures and extratemporal, multifocal, or generalized seizures [2].
Recently, we retrospectively studied surgical outcomes in patients with post-VE epilepsy [63]. In 10 surgically treated patients with epilepsy secondary to VE, we used SEEG, complemented by epileptogenicity index, to investigate the features associated with surgical outcomes (Fig. 3). We found that in the patients who achieved total freedom or significant improvement in seizures, the seizure onset was located either in the anteromesial temporal structures or in the focal gyrus, which is consistent with previous findings. The fact that temporal resections were successful in those patients confirms the apparent localization of maximal abnormality, but this result does not necessarily apply to other patients. The presence of extratemporal clinical features, coupled with the knowledge of the cause, means that the surgical decision should be made with caution.
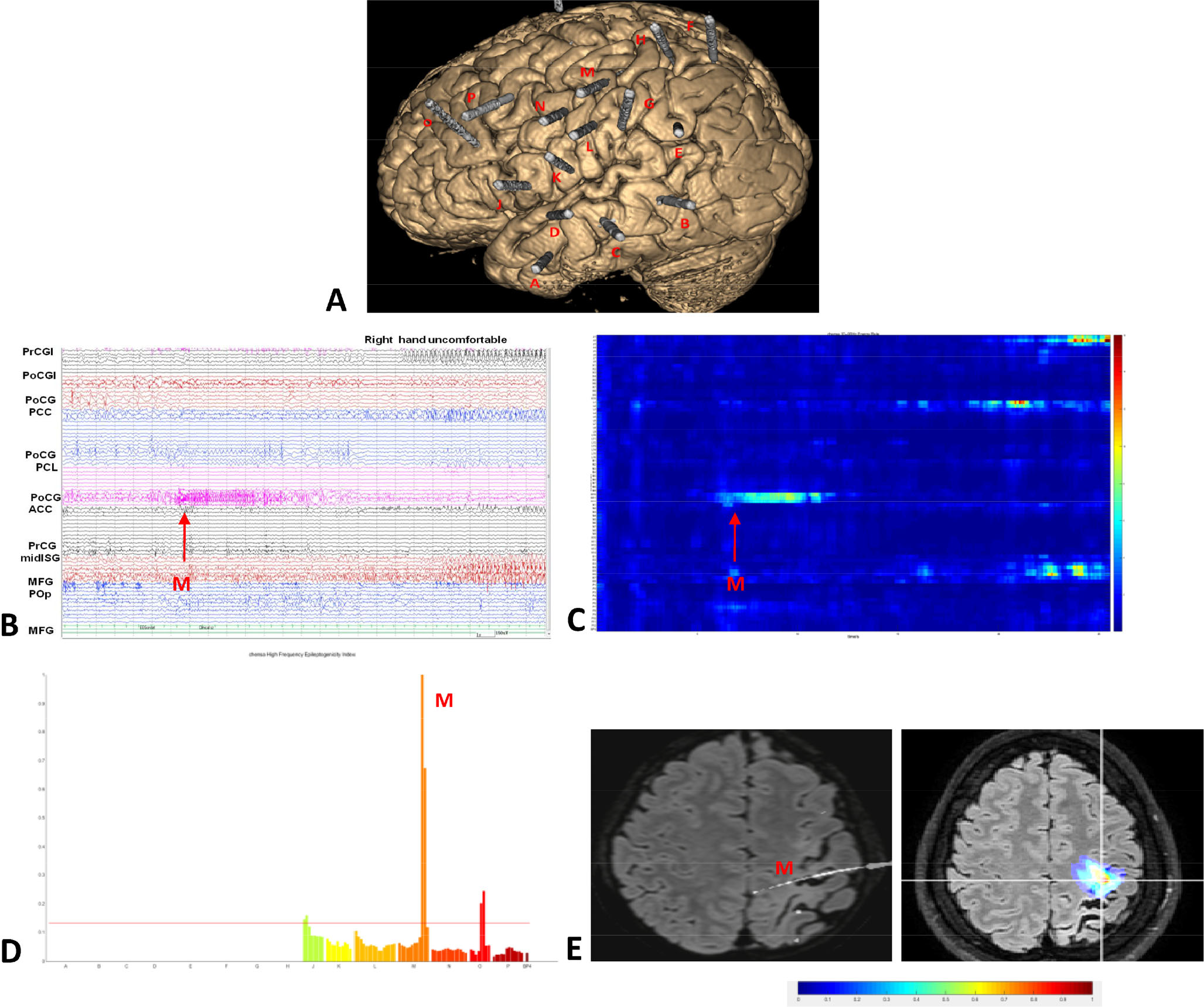
SEEG recordings in a patient who suffered epilepsy after viral encephalitis. (A) Locations of the SEEG electrodes (A to H and J to P) are shown in the three-dimensional MRI. The main area explored was the perisylvian region of the left hemisphere. (B) Seizures started with low-voltage fast-paced electrical activities in the postcentral gyrus (external leads of electrode M). ACC: anterior cingular cortex; MFG: middle frontal gyrus; midISG: middle short gyrus; PCC: posterior cingular cortex; PCL: paracentral lobule; PoCG: postcentral gyrus; PoCGI: postcental gyrus of insula; POp: parietal operculum; PrCG: precentral gyrus; PrCGI: precentral gyrus of insula. (C) Electrode contacts involved in seizure onset were captured by the epileptogenicity index analysis, and the seizure onset was in the same position (electrode M) as visually seen. (D) Epileptogenicity index values quoted in the chart, demonstrating maximal epileptogenicity in electrode M. (E) Superimposition of epileptogenicity index values on the axial planes shown on MRI. Seizure onset was confined to a single gyrus. Reproduced with permission from Ref [63], ©the authors, 2019.
In our study, 10 patients of intractable epilepsy after VE were performed surgery using SEEG, 5 patients reported a reduction in seizure frequency and severity, which improved their quality of life. 3 patients became completely seizure-free after surgery; 5 patients achieved Engel class II or III outcomes, and a worthwhile reduction of seizure frequency was achieved among these patients. For these patients, surgical treatment was palliative rather than curative. A presurgical evaluation is necessary in patients with drug-resistant post-VE epilepsy; SEEG makes the accurate localization possible, and surgery may be palliative or even curative.
4 Conclusions
In summary, VE is an acute central nervous system infection. In survivors of VE, the risk of developing epilepsy, both in the acute presentation and the long-term neurologic sequelae, is increased tenfold. This is clearly an area of great clinical importance requiring further investigation. Our primary aim was to review the most recent information regarding the clinical features of and the surgical treatment for post-VE epilepsy. Clinical manifestations of post-VE epilepsy vary, and the manifestations and imaging findings reflect primarily effects in the frontal and tem-poral lobes. In children, research has shown that post-VE epilepsy often consists of infantile spasms. Successful tailored surgical intervention may lead to a favorable outcome or may improve quality of life in patients with VE. Exploration with SEEG is mandatory in these cases, especially in the absence of a clear epileptogenic lesion or diffuse lesions.
Footnotes
Conflict of interests
All contributing authors have no conflict of interests.
