Abstract
Improvement in survival of systemic lupus erythematosus has been brought about with new advancement in treatment. However, glucocorticoids remain the sole cornerstone and as patients live longer, there is a need to address long-term complications brought by long-term glucocorticoid use such as osteoporosis. In this review, glucocorticoid-induced osteoporosis in systemic lupus erythematosus will be extensively discussed. This would include prevalence of osteoporosis in systemic lupus erythematosus patients, the difficulties in measuring fracture risk and pitfalls in using conventional methods such as bone mineral density. In addition, the mechanism of actions of glucocorticoids and evidence for glucocorticoids in the treatment of specific systemic lupus erythematosus manifestations would be explored and we also discussed specific pathophysiological mechanisms in the development of glucocorticoid-induced osteoporosis in systemic lupus erythematosus. We also reviewed the latest guidelines in the treatment of glucocorticoid-induced osteoporosis and the evidence for various osteoporosis medications. Finally, we recommend an approach in monitoring bone health and the treatment of osteoporosis specifically in systemic lupus erythematosus patients.
Keywords
Introduction
Systemic lupus erythematosus (SLE) is an autoimmune disease that follows a variable and unpredictable course. SLE can potentially affect multiple organ systems, leading to severe consequences. In the past, uncontrolled inflammatory process of the disease has led to high mortality, but in recent decades, mortality has improved with 10-year survival rates exceeding 90%.1,2
The improvement of overall survival in patients with SLE has partly been brought about with the use of an expanding armamentarium of immunosuppressive drugs, including glucocorticoids (GCs), cyclophosphamide, mycophenolate and recently, new biologics such as rituximab and belimumab.3–5 GCs, however, continue to be extensively used in SLE and remain the sole cornerstone of treatment. Patients with SLE are often exposed to high-dose GC for induction therapy and are subject to long-term low to moderate GC dose for maintenance of remission. As such, there is a need to address the long-term complications of GC use. In this review, glucocorticoid-induced osteoporosis (GIOP) will be extensively discussed, spanning from its prevalence, mechanism, impact on the health of SLE patients and potential strategies to mitigate the impact of GIOP on the well-being of patients with SLE.
Prevalence of GIOP in SLE patients
Diagnosis of osteoporosis is defined in the World Health Organization (WHO) guidelines. 6 Dual energy absorptiometry (DXA) is used to measure bone mineral density (BMD) and a T-score (the number of standard deviations the measured BMD differs with reference to a young healthy adult population) of −2.5 or less is considered to be osteoporotic. 6 Using this definition, the prevalence of osteopenia and osteoporosis differs widely in SLE patients in different geographical regions. In a British cross-sectional study of the 242 SLE patients, Yee et al. 7 showed that the prevalence of osteoporosis and osteopenia were 10.3% and 40.5%, respectively. However, in a Chinese study of 152 SLE women, prevalence was shown to be 21.7% and 59.9%, respectively. 8 while in a Canadian study of 81 postmenopausal SLE women, they were 12.3% and 42.1%, respectively. 9
There are a number of concerns with the use of T-scores in SLE patients in clinical settings. First, the WHO defined osteoporosis in postmenopausal Caucasian women based on the T-scores. 6 SLE is a disease that chiefly affects women of childbearing age. The applicability of T-scores in defining osteopenia and osteoporosis thus might not be ideal. As such, the use of Z-score has been proposed for defining secondary osteoporosis. The Z-score is the number of standard deviations the measured BMD differs from age, gender and ethnicity matched population. The Canadian Clinical Practice Guidelines defined pre-menopausal women and men below 50 years old as having BMD ‘below expected range for age’ if the Z-score is less than −2. 10 The American College of Rheumatology (ACR) for GIOP has recently proposed to use the Z-score for adults younger than 40 years old for predicting risk of fractures, with a threshold of less than −3 defined as moderate risk. 11
However, in a retrospective analysis over 247,000 individual Z-scores and patient records, McKiernan et al. 12 showed that although low Z-scores are associated with a higher prevalence of secondary osteoporosis, there was no clear inflection point. Hence, a useful clinical diagnostic threshold could not be defined. Second, fragility fractures can occur even in patients with normal BMD. In the same British cross-sectional study by Yee et al., 7 of the 22 SLE patients with fragility fractures, only seven (31.8%) had T-scores in the osteoporotic range. Similarly, in a cohort study of 152 SLE patients whom all had spinal radiographs, 31 were found to have morphometric vertebral fractures, of which nine (29.0%) were patients with normal BMD. 8
In addition, SLE patients carry a higher risk of fragility fractures compared to the normal population. In a UK-based population-control study, the relative risk of fracture among SLE patients was found to be 22% higher than that of healthy individuals, and the risk increased with the duration of the disease, with it being double if the disease duration was 10 years or longer. 13 Hence, assessment that incorporates BMD to estimate fracture risk like the popular Fracture Risk Assessment Tool (FRAX) may underestimate risks in SLE patients. 14 This is likely due to SLE patients requiring prolonged and high doses of GCs throughout their lives and while FRAX can adjust for GC use, this is only limited to a prednisolone-equivalent dose of 2.5–7.5 mg/day. 15
Mechanism of action of GCs
GC produces an immunosuppressive response via two different pathways, the genomic pathway and the non-genomic pathway.16–18 In the genomic pathway, the highly lipophilic GC molecules pass through the cell membrane and bind to cytosolic GC receptors. The GC receptor complexes translocate into the nucleus and modulate gene expression in the cellular nucleus, resulting in inhibition of intracellular expression of pro-inflammatory molecules. The process, however, has a lag time of at least 30–60 min for changes in gene expression and protein translation to take place. 17
The non-genomic pathway, on the other hand, acts more rapidly than the genomic pathway. After binding to cytosolic GC receptors, the resultant complexes activate transcription-independent mechanisms via intracellular proteins that result in inhibition of inflammatory cytokines like arachidonic acid. They can also act on cellular and mitochondrial membranes and suppress adenosine triphosphate (ATP) production, leading to alleviation of the downstream pro-inflammatory cascade.16,18
The use of GCs in SLE
Treatment with GC for SLE has been described since the 1950s in case reports and case series.19–21 To date, no controlled trial has proven the efficacy of GC per se because trials testing the efficacy and safely of GC without combination of an immunosuppressant are ethically impossible to perform. In clinical practice, dosage of GC is often arbitrarily stratified into:22,23
Low dose: ⩽7.5 mg prednisolone equivalent a day;
Medium dose: >7.5 to ⩽30 mg prednisolone equivalent a day;
High dose: >30 to ⩽100 mg prednisolone equivalent a day;
Very high dose: >100 mg prednisolone equivalent a day;
Pulse: >250 mg prednisolone equivalent a day, for a few days.
High doses and pulse therapy are often used to treat SLE patients with severe organ manifestations in order to saturate the effects of the genomic pathway and take advantage of the non-genomic pathway. 23 In practice, high-dose prednisolone is often given at 1 mg/kg/day and pulse therapy is given at 250–1000 mg of intravenous (IV) methylprednisolone for 3 days, although such doses are largely based on expert recommendations. Robust evidence supporting the use of pulse methylprednisolone remains scarce and consists mainly of small clinical trials performed in the 1980s;24–26 however, clinical consensus for the use of high-dose GC for treating severe SLE has been nearly universal throughout the century and remains prominent in current guidelines.27–30
Proliferative lupus nephritis is one of the most serious manifestations of SLE and if left untreated, progression to end-stage renal failure is a near certainty. Hence, use of high-dose GC remains highly prominent and is recommended in both the ACR and European League Against Rheumatism (EULAR) guidelines.28,29 Neuropsychiatric systemic lupus erythematosus (NPSLE) is another serious manifestation of SLE and current treatment recommendation also consists of pulse therapy with cyclophosphamide.27,31,32
Pathogenesis of GIOP in SLE
Endogenous GC at physiologic concentrations may have a role in promoting osteogenesis. 33 Exogenous GC, on the other hand, induces bone loss predominantly by both increasing bone resorption and reducing bone formation.34,35 With long-term GC therapy, impaired bone formation usually becomes the predominant skeletal effect. 36 This leads to a pattern of bone loss that is most pronounced in the first few months of GC use, followed by slower but steady bone loss with chronic use. Trabecular bone loss predominates initially, with most significant changes in the lumbar spine, but over time also affects cortical bone at other sites such as the femoral neck. 37
The precise mechanisms of how GC affect bone are not completely understood but a number of theories have been proposed. 38 GC decreases intestinal calcium absorption and increases renal calcium loss by inhibiting renal tubular calcium reabsorption. GC also suppresses testosterone, oestrogens, as well as insulin-like growth factor 1, hormones crucial for skeletal growth. 39 In addition, Steroid-induced myopathy leads to less loading of bone, resulting in increased bone loss, and is also associated with increased falls with consequent fragility fractures.
Animal model studies have shown that high-dose GC resulted in diminished bone formation and turnover. This was determined by histomorphometric analysis of tetracycline-labelled vertebrae. Ex vivo bone marrow cell cultures have also shown impaired osteoblastogenesis and osteoclastogenesis. There was also threefold increase of apoptosis in osteoblasts in the vertebrae and apoptosis in 28% of the osteocytes in metaphyseal cortical bone, demonstrating that GC-induced bone disease arises from changes in the numbers of bone cells. 40
Further studies have also showed that GC directly induces apoptosis of osteoblasts and osteocytes. Increased cell death of osteocytes directly reduces bone strength that is independent of bone volume loss. Loss of osteocytes also disrupts the osteocyte–canalicular network, which is responsible for signal transmission necessary for stimulating new bone formation at areas of damaged bone. 41 Liu et al. demonstrated that GC directly induced apoptosis of osteoblasts by activating caspase-3. 42 Other studies also showed that oral prednisolone as low as 2.5 mg daily could suppress serum osteocalcin, a marker of bone formation. 43
GC can also aggravate bone loss by reducing apoptosis of osteoclasts. GC prolongs lifespan of osteoclasts by upregulating receptor activator for nuclear factor kappa-B ligand (RANKL) and suppressing osteoprotegerin (OPG). 44 This leads to enhanced osteoclast function and increased bone resorption. Hence, markers of bone resorption are often increased in GC-treated patients.
Recent advances in understanding of the Wnt signalling pathway have highlighted its importance in bone metabolism. In short, activation of the canonical Wnt/β-catenin pathway enhances mesenchymal stem cells along the osteoblastic lineage while suppressing their commitment to adipogenic and chrondrogenic lineages. 45 The pathway also increases OPG secretion, thereby inhibiting osteoclastic activity. 46 GC however shifts differentiation the mesenchymal stem cells towards the adipocyte lineage, leading to reduced osteoblastogenesis. 47 Moreover, GC also directly increases Dickkopf expression, an antagonist that suppresses Wnt binding to its receptor. 48
It has been well established that sex hormones play a key role in bone health. Over the decades, many studies on postmenopausal women have proven the link between oestrogen deficiency to increased bone fragility. Oestrogen acts on osteoclasts to downregulate bone remodelling by first inducing apoptosis of osteocyte. 49 It also decreases bone resorption by increasing OPG while downregulating RANKL production by osteoblastic cells. 50 Recent studies have also shown that serum oestrogen level is inversely associated with sclerostin, a potent inhibitor of the Wnt pathway. 51 Testosterone, on the other hand, enhances osteoblastic activity and therefore increases bone formation. Studies have shown that androgens enhance osteoblastic proliferation and inhibit apoptosis of osteoblasts. 52 GC however directly suppresses the hypothalamic–pituitary–adrenal–gonadal axis, reducing production of gonadotropin-releasing hormone and therefore decreases sex hormone production at the gonads. 53
Moreover, SLE patients have altered bone geometry compared to the normal population. In an American study comparing hip geometry between SLE patients and normal controls, there were reduced section modulus and cross-sectional areas with increased buckling ratio in SLE patients. 54 This has been postulated to be due to chronic inflammation itself and while some animal studies support this, 55 there is also evidence that chronic GC use can directly disrupt bone geometry and microarchitecture. In a study using quantitative computed tomography on SLE patients with low disease activity (mean SLEDAI 2.6) but with chronic GC usage (mean duration of 9.4 years, mean cumulative dose of 18.6 g), there were lower volumetric BMD, reduction in cortical thickness and increased porosity compared to normal controls. 56
In summary, alterations in bone turnover can contribute to microarchitectural change and result in reduced bone quality with a rapid increase in fracture risk. 34 This may explain why patients on GC have higher fracture risk compared to GC-naïve controls with the same BMD. 57
Other potential contributors of osteoporosis in SLE
The development of osteoporosis is not limited to GC therapy itself and SLE alone can affect bone quality. 58 The systemic inflammation of SLE induces bone loss by increasing osteoclastic bone resorption and by reducing osteoblastic bone formation.
Increased serum levels of tumour necrosis factor (TNF) 59 and oxidized low-density lipoprotein (LDL) have been described in patients with active SLE. 60 Oxidized lipids activate T cells, increasing production of RANKL and TNF. Both RANKL and TNF stimulate osteoclast differentiation and activity. 59
Some studies have shown that SLE patients have reduced serum levels of osteocalcin, a marker for bone formation61,62 and osteocalcin is negatively associated with SLE disease activity. 62 Another study also demonstrated an association between high rate of disease flares and increased bone loss in SLE. 63 Interestingly, anti-Ro antibody positivity and absence of Anti-Sm antibodies were shown to be associated with reduced bone mass. 64 However, such observation may be confounded by the fact that anti-Ro positive SLE patients are often advised to avoid sun exposure. 58
Besides GC, other therapies for SLE may contribute to osteoporosis. Cyclophosphamide use was shown to be an independent risk factor for femoral neck fracture in SLE in a study from Hong Kong, 65 and this can be explained by the gonadal suppressive effect of cyclophosphamide.66,67 In addition, animal models have demonstrated that cyclophosphamide may directly influence the Wnt /β-catenin pathway, thereby inhibiting osteoblastogenesis. 68 Another study from Taiwan showed increased serum fibroblast growth factor-23 and decreased bone turnover in SLE patients treated with cyclosporin and GC but not GC alone. 69 A cohort study from Italy reported use of anticoagulants and antiepileptic drugs as independent risk factors for symptomatic fractures in patients with SLE. 70
Circulating vitamin D levels have been shown to be reduced in patients with SLE.71,72 This may be contributed by photosensitivity, increased use of sunscreen, reduced sunlight exposure, and physical inactivity and chronic arthritis. Lupus nephritis can also lead to renal failure which results in reduced 1,25-(OH)-D levels. Finally, SLE patients are also subject to the same risk factors for osteoporosis that affect the general population, including age, postmenopausal status and low body mass index (BMI).58,73
Management and monitoring of osteoporosis in patients with SLE
Osteoporosis develops in patients with SLE via complex mechanisms as elucidated earlier. Hence, the management of bone loss in these patients requires a multi-pronged approach which includes (1) evaluation of SLE management that impacts bone health; (2) modification of lifestyle; (3) review of medications associated with the development of osteoporosis; (4) addressing the secondary causes of bone loss; (5) management of low bone density and (6) mitigation of fall risk in patients with SLE. Figure 1 illustrates a suggested approach to the management of osteoporosis and the reduction of fracture risk in patients with SLE.
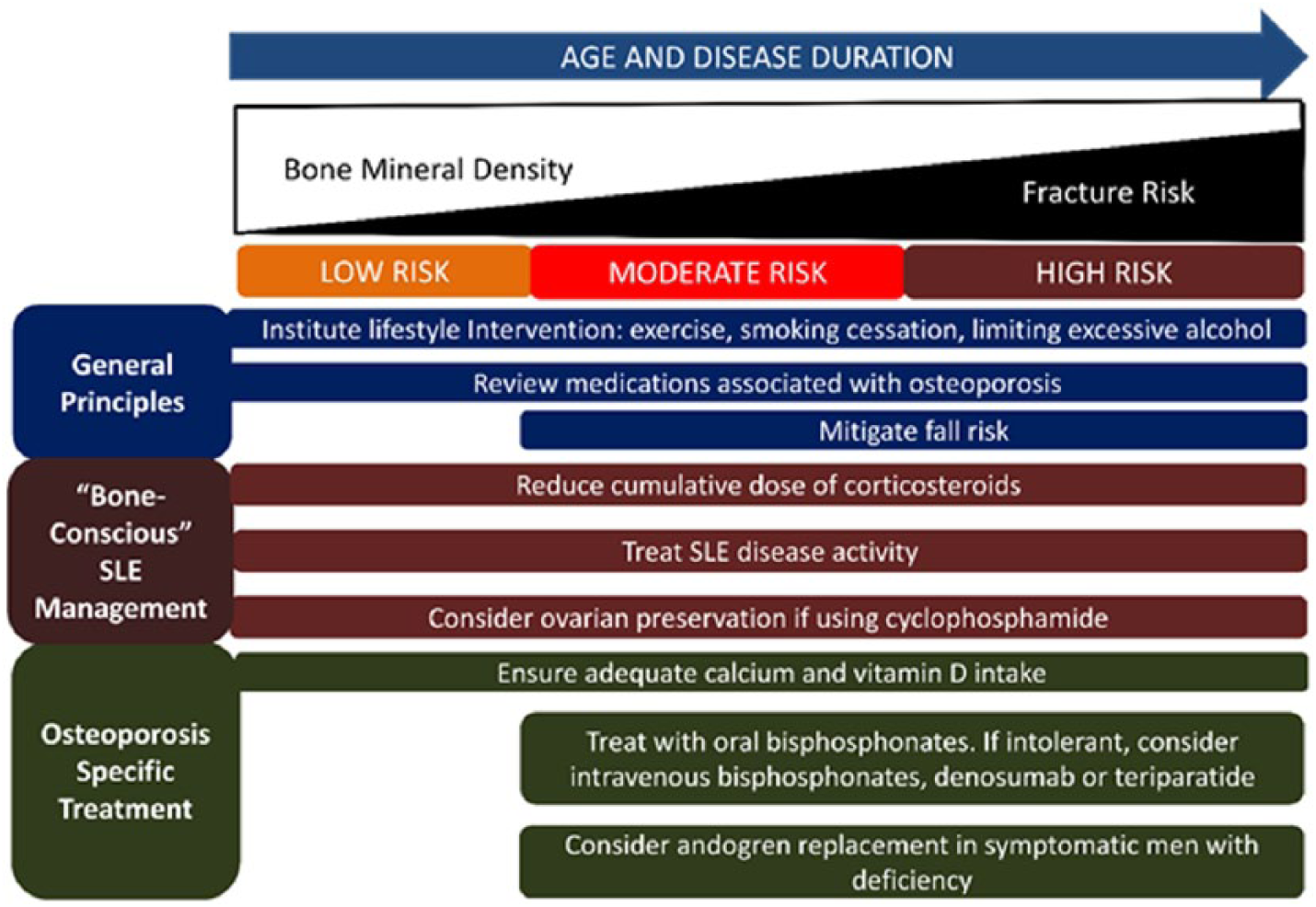
Approach to managing osteoporosis and fracture risk in patients with SLE.
Aspects of SLE management that impact bone health
Management of osteoporosis in patients with SLE must begin with mitigating the risk of BMD deterioration during the treatment of SLE. Because even low doses of GC can increase fracture risk, clinicians must aggressively taper the dose of GC when disease is under control. 74 Whether treating SLE disease activity to target would improve long-term outcomes in bone health needs to be addressed in future studies.
The use of cyclophosphamide is associated with premature menopause in up to 50% of patients with SLE and accounts for the accelerated bone loss in these patients. 75 Gonadotropin-releasing hormone analogues have been shown to prevent premature ovarian failure in patients with SLE receiving cyclophosphamide76,77 and should hence theoretically prevent BMD deterioration associated with ovarian failure. However, whether ovarian preservation with these methods truly protects against bone loss in clinical practice needs to be further studied. 78
Lifestyle modification and medication review
Physicians must also encourage patients to participate in regular aerobic, weight-bearing or resistance exercise to protect against BMD deterioration.79,80 Patients who maintain their body weight in the healthy range of BMI may circumvent the increased fracture risk associated with extreme BMI on both ends.8,81 Advice on smoking cessation and limitation of alcohol intake to 2 or less units a day is also standard practice. 82 Where possible, medications associated with osteoporosis and fractures should be reviewed for their indications and potential substitutes. These medications include unfractionated heparin, proton pump inhibitors, thiazolidinediones, selective serotonin-reuptake inhibitors and calcineurin inhibitors. 83 While a definite relationship between these agents and osteoporosis and fractures has not been fully established, careful evaluation of their risks and benefits is warranted. Adequate intake of calcium of at least 1000 mg/day and vitamin D of at least 600IU/day is also recommended. 11
Managing patients with SLE at moderate to high risk for fractures
The recently published 2017 ACR Guidelines on the Prevention and Treatment of Glucocorticoid-Induced Osteoporosis recommended anti-resorptive treatment for patients on long-term GC if deemed moderate or high risk. Here, high risk is defined as the presence of major osteoporotic fractures in adults, a BMD of ⩽−2.5 in postmenopausal women or men aged ⩾50 years old, or a FRAX 10-year probability of osteoporotic fracture of ⩾20% or a 10-year probability of hip fracture of ⩾3%. Moderate risk is defined as a FRAX 10-year probability of major osteoporotic fracture of 10%–19% and a probability of hip fracture of >1% but <3% in adults ⩾40 years of age. In adults <40 years, moderate risk is defined a hip or spine Z-score of <−3 or rapid bone loss of ⩾10% in hip or spine over 1 year and continuing GCs of ⩾7.5 mg daily for ⩾6 months. In patients receiving corticosteroids >7.5 mg daily, the FRAX score must be adjusted by multiplying the absolute 10-year major osteoporotic fracture risk by 1.15 and the hip fracture risk by 1.2. 84 It is important to note that the definition of ‘high risk’ and FRAX intervention thresholds may differ in different countries. For example, in Japan and China, the intervention threshold for 10-year probability of major osteoporotic fracture is 15% and 4%, respectively. 85
Oral bisphosphonates have been recommended as first-line treatment in this guideline. The use of bisphosphonates in GIOP has been supported by a large body of evidence, including a recently updated Cochrane systematic review which has concluded that it reduces vertebral fracture risk but not non-vertebral fracture risk. 86 In particular, the use of alendronate in patients with rheumatic diseases with GIOP has been supported by a meta-analysis of nine studies. 87 Where there are contraindications to oral bisphosphonates, IV bisphosphonates, denosumab or teriparatide can be considered. These agents are second line as they can be significantly costlier than oral bisphosphonates. 11 The use of oral and IV bisphosphonates, denosumab and teriparatide in SLE patients with osteoporosis was mainly extrapolated from studies in women with postmenopausal osteoporosis or patients with GIOP. 88 Only a few studies specifically investigated patients with rheumatic diseases or SLE.
More recent studies have found that denosumab is efficacious in GIOP by improving BMD, although they lack fracture outcomes. 89 Some studies have found denosumab to be more efficacious than bisphosphonates in GIOP.90,91 In particular, in a 24-month randomized controlled trial involving nearly 800 patients receiving GCs, denosumab was shown to be both non-inferior and superior compared to risedronate in improving BMD. 92 Interestingly, it has been suggested that denosumab may be more efficacious in patients with GIOP who have not received prior treatment with bisphosphonates. 93 One drawback, however, is the risk of fracture recurrence if denosumab is discontinued. In a review of 24 women with vertebral fractures after denosumab was stopped, factures occurred between 8 and 16 months after last denosumab dose and patients had more fractures if they received a longer duration of denosumab before stopping. 94
Also, most studies used BMD as a surrogate end-point for fracture risk, and only a handful of studies directly addressed fracture risk reduction. The fact that better BMD improvement does not necessarily translate to greater reduction in fracture risk was highlighted in the same trial comparing denosumab with risedronate, whereby although denosumab was better at improving BMD, fracture rates were similar between them. 92
Teriparatide has been shown to be superior to alendronate in individuals with GIOP. A randomized controlled trial published in 2007 showed that teriparatide conferred an absolute risk reduction of 5.5% for vertebral fracture in 18 months compared to alendronate. Although hip fracture rates were similar, hip BMD improved significantly more in the teriparatide group. 95 The 18-month extension of the same trial again showed a 6% absolute risk reduction in vertebral fracture over this period with similar hip fracture rates. 96
Importantly, as a result of the young age of onset and increased longevity of patients with SLE, they are likely to be exposed to a longer duration of anti-osteoporosis therapy than postmenopausal women. 97 Therefore, the risks of long-term therapy such as atypical femoral neck fractures and osteonecrosis of the jaw, particularly in GC-treated patients, must be weighed against their benefits. 98 However, it must be noted that the risks of these adverse events are rare, and that the risk–benefit profile of bisphosphonate therapy in these patients still remain favourable. Bisphosphonates are retained in bones for extended duration and are able to cross the placenta. Although several small prospective studies have not demonstrated any teratogenic effect of bisphosphonates, clinicians must exercise caution in prescribing bisphosphonates in pre-menopausal women.99,100 Based on bone turnover marker studies, some authors recommended a washout period of 5 years for alendronate and zoledronate, and 2 years for ethidronate and risedronate before conception.99,101 A small body of case reports and case series suggest that bisphosphonate exposure before or during pregnancy does not lead to adverse foetal outcomes.100,102
Raloxifene should be avoided in patients with SLE who have thrombotic tendency or history of vascular thrombosis. Notably, the benefits of raloxifene in GIOP are not fully elucidated. 11 While a small randomized controlled trial of 62 patients with SLE has shown that raloxifene improved lumbar spine BMD without causing disease flares, this study excluded patients with hypercoagulability or anti-phospholipid syndrome. 103 Denosumab has been shown to be an effective agent in GIOP but it may theoretically increase the risk of infections in patients who are heavily immunosuppressed. Indeed, the ACR Guidelines for GIOP states that there is a lack of safety data on the use of denosumab in patients who are immunosuppressed. 11 Strontium ranelate and calcitonin are options for treatment of postmenopausal osteoporosis. However, there have been insufficient evidence for the use of these agents in the treatment of GIOP. 104 Moreover, due to increased cardiovascular risk associated with strontium use, it should only be used when other agents are considered unsuitable. 105 Calcitonin alone may not be efficacious in treating osteoporosis and is mainly being utilized for its analgesic effect in patients with compression fractures. 106
Recently, the ARCH trial demonstrated that an 1-year treatment of romosozumab, an anti-sclerostin antibody, followed by alendronate conferred a 5.7% absolute risk reduction of vertebral fractures over 24 months in patients with postmenopausal osteoporosis compared to patients who received alendronate alone for 24 months. 107 However, there was an imbalance of cardiovascular and cerebrovascular events in the romosozumab group and this agent is not currently available in many countries. In patients with severe osteoporosis, the combination of denosumab and teriparatide has also been shown to improve femoral neck and vertebral BMD more than either agents alone. 108 However, the utility and long-term safety of these approaches remain to be investigated in patients with GIOP. Table 1 compares the advantages and disadvantages of each class of medications for patients with SLE.109–125
Comparison of anti-osteoporotic agents.
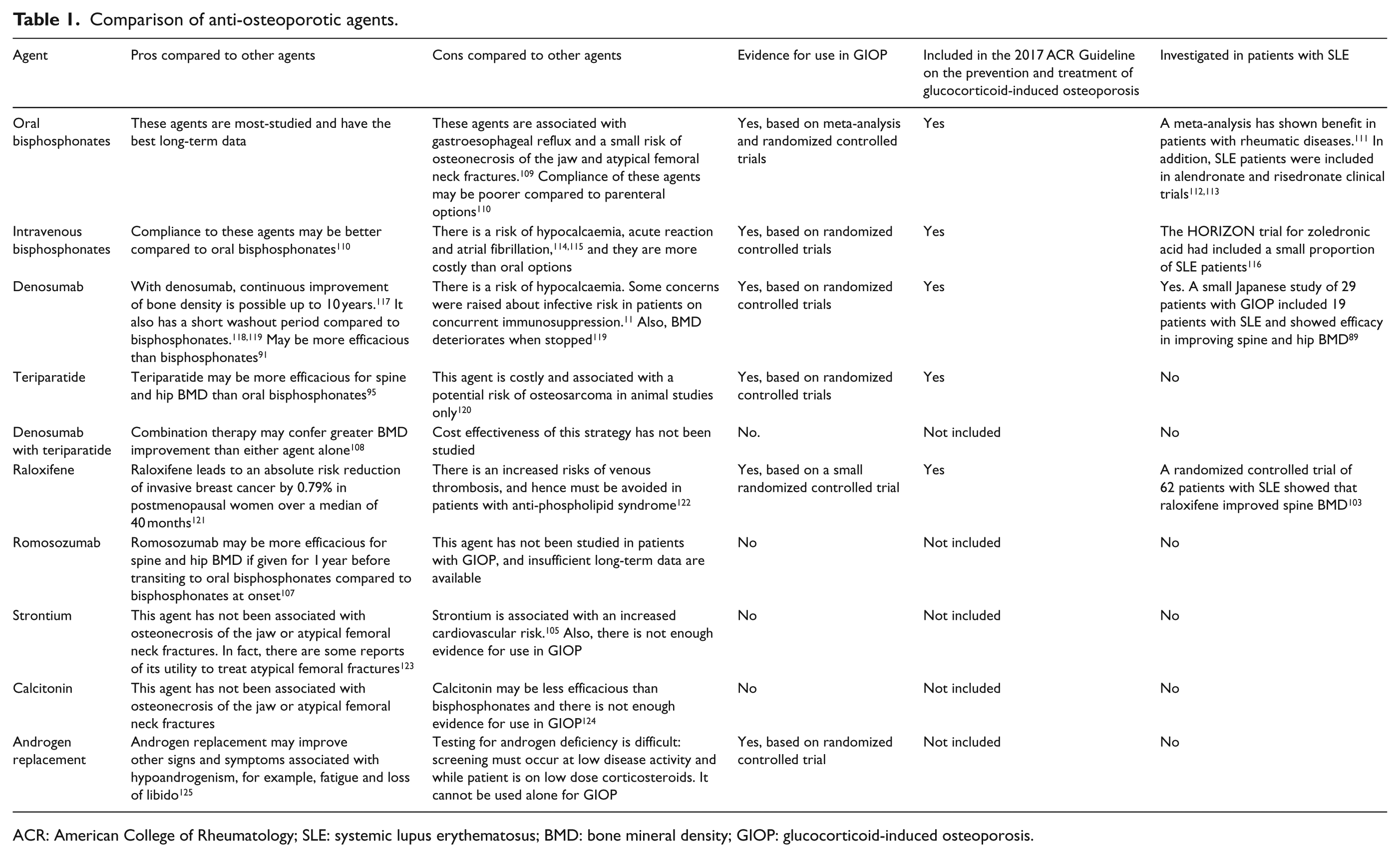
ACR: American College of Rheumatology; SLE: systemic lupus erythematosus; BMD: bone mineral density; GIOP: glucocorticoid-induced osteoporosis.
Because patients with SLE also suffer degraded bone microarchitecture and trabecular quality in addition to reduced bone density, it is important to consider the effects of therapy on bone quality. Anti-resorptives, including bisphosphonates and denosumab, improve bone quality by inhibiting remodelling and reducing trabecular volume loss, therefore improving connectivity. Thus, these agents exert their anti-fracture effects via improvement of both bone density and quality. 126 Teriparatide, being an osteo-anabolic agent, has been shown to improve trabecular architecture in a bone biopsy study and a randomized controlled trial.127,128
It is also important to note that while both anti-resorptives and osteo-anabolic agents increase BMD and reduce fracture risk, the mechanisms of how these drugs achieve this are different. In a randomized controlled trial comparing teriparatide with zoledronic acid, measurements of bone mineralization density distribution (BMDD) were used to determine differences in mineralization kinetics. The authors demonstrated that teriparatide increased bone turnover rate with heterogenous mineralization while zoledronic acid slowed bone turnover rate but resulted in higher and more homogeneous mineralization of the bone matrix. 129
There is also evidence that combination therapy performs better than monotherapy. In a randomized controlled trial comparing denosumab, teriparatide and the combination of denosumab and teriparatide, combination therapy improved radial trabecular volumetric BMD more than either agent alone. 130
Hypotesteronism afflicts up to 60% of males with SLE, particularly as a result of exposure to cyclophosphamide or GC. 131 A small body of evidence suggests that testosterone replacement in men deficit of the hormone may modestly improve spine BMD, although it is unclear whether this leads to reduced fracture risk. 132 The 2018 Endocrine Society guidelines recommend ‘testosterone therapy in hypogonadal men to induce and maintain secondary sex characteristics and correct testosterone deficiency’. 125 In patients with SLE, serum androgen levels have been found to correlate with BMD. 133 Testing and treatment should also be considered in patients who exhibit signs and symptoms of hypoandogenism, such as erectile dysfunction and low libido. 125 As active SLE disease and GC may both interfere with the pituitary–gonadal axis, measurement of free and total testosterone levels is only meaningful when SLE disease activity is low and GC is tapered to a low dose. 125 Interestingly, in vitro studies demonstrated that testosterone inhibited B-cell activity and decreased anti-dsDNA production by peripheral blood mononuclear cells. 134 A small open-label study also demonstrated that dehydroepiandrosterone replacement in females is associated with lower lupus disease activity measured by SLEDAI. 135 However, testosterone replacement has several important contraindications which are not discussed in the scope of this review. Clinicians should therefore work with endocrinologists to discuss the risks and benefits of androgen replacement for the individual patient.
Monitoring bone health and fracture risk in patients with SLE
The 2017 American College of Rheumatology Guideline for the Prevention and Treatment of GIOP recommend that all patients older than 40 years should receive a fracture risk assessment ideally with an assessment of BMD upon GC initiation. 11 BMD testing within 6 months of initiation of GC is recommended for patients younger than 40 years with risk factors of osteoporosis. 11
Patients with SLE should be assessed for fracture risk annually. The FRAX tool is the most commonly used instrument for fracture assessment. 11 Nevertheless, clinicians must acknowledge the important limitations of using FRAX in patients with SLE, including the fact that (1) it has not been validated in patients who are receiving anti-osteoporotic treatment; (2) it does not take into account the dose of GC for fracture risk calculation; (3) it does not incorporate fall risk, which may be elevated in patients with SLE; (4) it may underestimate fracture risk in patients with SLE; (5) the fracture risk can only be imputed in patients above the age of 40 and (6) different FRAX intervention threshold exists for different countries.85,88,136 Whether imputing a trabecular bone score with the FRAX calculation improves fracture risk estimation for patients with SLE remains unclear. The Qfracture tool and Garvan risk assessment tools allow imputation of fall risk but they do not take into account the BMD and their use in patients with rheumatic diseases is limited.137,138
Conclusion and future perspectives
GCs remain an indispensable therapeutic agent in the treatment of SLE. Given at high doses, it allows control of acute flares with severe manifestations and used at low doses to achieve long-term disease control. Unfortunately, its use comes with cost of multiple side effects, and one of the most significant being GIOP. Hence, given the excellent survival of most patients, management of GIOP must be incorporated into routine care of SLE. Looking forward, ongoing development of newer immunosuppressive agents has led to more novel approaches against SLE. This will hopefully reduce our dependence on GCs and its associated complications in the future. On the other hand, treatment of osteoporosis has often been limited to the use of mainly anti-resorptive agents with few ‘bone-building’ agents. More data with the use of newer therapeutic options such as romosozumab for SLE patients remains awaited.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
