Abstract
Cognitive dysfunction is one of the five psychiatric syndromes recognised by the American College of Rheumatologists neuropsychiatric lupus nomenclature committee. Cognitive dysfunction affects 20%–80% of systemic lupus erythematosus patients with and without overt central nervous system involvement. Specific cognitive dysfunction associated with systemic lupus erythematosus includes impairment in attention, memory and visuospatial process. Besides disease activity and vascular risk factors, the neuropathology behind cognitive dysfunction in systemic lupus erythematosus is a result of dysfunction involving the immune cells, cytokines, chemokines and autoantibodies (Abs), as well as volume reduction in the white matter and grey matter. In this review, we will discuss the challenges of using traditional neuropsychological assessment to assess cognitive function in clinical practice, the limitations of conventional neuroimaging methods, the controversies of using corticosteroids, other pharmacological treatments and non-pharmacological interventions to treat cognitive dysfunction associated with systemic lupus erythematosus.
Keywords
Introduction
Systemic lupus erythematosus (SLE) is a systemic autoimmune disorder which is characterised by hyperactive B and T cells, production of high level of autoantibodies (Abs), deposition of immune complexes and damages in multiple organ systems.1,2 Neuropsychiatric SLE (NPSLE) is caused by a combination of pathological processes including Abs, inflammatory markers and microvasculopathy. 3 Genetic polymorphisms in the pathways of immune complex clearance, such as the IgG Fc (Fcγ) receptor IIIa, Fcγ RIIIb and integrin alpha M (ITGAM) genotypes, are susceptibility genes for NPSLE. 4 NPSLE patients are more likely to have elevated serum levels of anti-cardiolipin (AcL), lupus anticoagulants (LA), antiphospholipid (APL), anti-ribosomal-P Abs and anti-neuronal Abs compared with SLE patients without neuropsychiatric (NP) manifestations. 4 Efthimiou and Blanco 5 highlighted the major challenge in diagnosing NPSLE is the lack of established biomarkers and diverse clinical features, a challenge shared by most psychiatric disorders. In a meta-analytical study, Mak et al. 6 highlighted the damages associated with NPSLE had led to a negative impact on the survival of SLE patients between 1950s and 2000s. As a result, early detection and management of NP symptoms may improve survival of SLE patients.
The diffuse central nervous system (CNS) disorders accepted by the American College of Rheumatologists (ACR) neuropsychiatric lupus nomenclature committee include acute confusional state, anxiety disorder, cognitive dysfunction, mood disorder and psychosis. 7 ‘Lupus brain fog’ is a common patient complaint which refers to forgetfulness and confusion associated with cognitive dysfunction. 8 Cognitive dysfunction is a common phenomenon in SLE patients without CNS involvement and leads to negative impacts on social functioning. 9 Cognitive dysfunction occurs in 20%–80% of patients with SLE. 10 The prevalence of cognitive dysfunction among patients with SLE is twice of the general population. 11 Impairment in executive function is the hallmark of cognitive dysfunction of SLE. It is important to note that SLE patients with and without CNS involvement display cognitive dysfunction, 12 but patients with NPSLE display more severe cognitive dysfunction compared to patients without overt CNS involvement. Psychomotor speed is the best factor to differentiate patients with SLE from healthy controls. Specific cognitive symptom such as inattention is associated with unemployment in patients with SLE. 13 Both cognitive dysfunction and depressive symptoms are common in SLE which incur high direct and indirect medical costs. 14 As a result, early recognition and adequate treatment of cognitive dysfunction remain one of the unmet medical needs in SLE. 15
Types of cognitive dysfunction associated with SLE
Attention
Attention is an intensive process in which information selection takes place. 16 SLE patients were found to have more clinically significant impairment in attention as compared to healthy controls. 13 SLE patients with CNS involvement performed significantly worse than those without CNS involvement and healthy controls in complex attention tasks which involve sustained, divided and selective attention to handle multiple stimuli. 17 The prevalence of inattention and slow processing speed in patients with SLE is around 20%. 12 Risk factors for inattention include cerebral involvement and high level of anxiety. The inattention is specifically associated with decreased prefrontal white matter integrity. 18 In a functional brain imaging study, patients with childhood-onset SLE and cognitive dysfunction demonstrated more brain activation during attention task as compared to their counterparts without cognitive dysfunction. 19 The activation is a compensatory mechanism which allows maintenance of attentional performance. The compensatory mechanism allowed childhood-onset SLE patients with cognitive dysfunction to perform equally well in the attention task compared with SLE patients without cognitive dysfunction. 19
Executive functioning
Executive function involves cognitive flexibility, decision making, abstract thinking, judgement and sequencing. Executive function (frontal lobe) tests include Stroop Colour-Word Test and Trail Making Test. 16 In an Asian study, Ho 4 reported that there were no significant differences between SLE patients without CNS involvement and healthy controls in the scores of Stroop Colour-Word Test and Trail Making Test. A recent meta-analysis found that deficits in the domains of cognitive fluency and visual reasoning were not significantly differentiated by the presence or absence of overt CNS involvement. 20 This finding supports the notion of compromise in executive function in SLE with or without overt NP symptoms.
Memory
Memory comprises encoding, registration, storage and retrieval of information. 16 Memory deficits in patients with SLE include impairments in immediate and delayed recall of verbal and non-verbal information.17,21 Working memory is short-term memory that temporarily holds information for processing. 16 SLE patients without CNS involvement trended towards greater impairment on working memory than healthy controls. 22 The n-back task is a continuous performance task that is used to assess working memory. The task consists of indicating when the current stimulus matches the stimulus from n steps earlier in the sequence. As the value of n increases, the working memory load gets higher. Under high working memory load conditions (such as 2-back), patients with SLE had lower accuracy scores and slower reaction time. 23 During working memory tasks, patients with childhood-onset SLE and cognitive dysfunction exhibited less brain activation than their counterparts without cognitive dysfunction in functional brain imaging. 19
Visuospatial processing
The Rey–Osterrieth Complex Figure Test (RCFT) assesses five domains of neuropsychological functioning: visuospatial recall memory, visuospatial recognition memory, response bias, processing speed and visuospatial constructional ability. Glanz et al. 24 reported that SLE patients with low disease activity were impaired in immediate visual spatial memory compared to healthy controls. Monastero et al. 21 found that patients with SLE showed more visuo-constructional skill impairment than healthy controls. Furthermore, NPSLE patients demonstrated poorer short-term visuospatial memory performance than SLE patients without CNS involvement. The significant impairment in visuo-constructional abilities is related to comorbid depression. In an Asian study, Ho 4 reported that SLE patients with high levels of anxiety and depression demonstrated significantly lower total copy scores of RCFT than SLE patients with low levels of anxiety and depression. This finding indicates low processing speed and visuospatial constructional ability are associated with high levels of anxiety and depression in patients with SLE. Poor performance in the RCFT indicates lesions in the parieto-occipital and frontal lobes in patients with SLE. 25 During visuospatial memory tasks, patients with childhood-onset SLE and cognitive dysfunction demonstrated less brain activation than their counterparts without cognitive dysfunction in functional brain imaging. 19
Clinical assessment for cognitive dysfunction in patients with SLE
Traditional neuropsychological assessments
The ACR recommends assessing cognitive function in SLE patients by sophisticated neuropsychological battery administered by a trained psychologist. Simple bedside cognitive screening test such as the Mini-Mental State Examination (MMSE) and Montreal Cognitive Assessment (MOCA) were shown to be unhelpful in assessing NP manifestations in patients with SLE. 10 A recent study found that 48% of SLE patients suffered from cognitive impairment based on the MOCA. 26 Poole et al. 27 found that cognitive deficits were better assessed by performance-based tests of disability rather than a self-report assessment. Breitbach et al. 28 recommended the measurement of cognitive function in early stage of SLE is important to provide a baseline reference for disease monitoring.
The ACR-SLE neuropsychological battery is composed of the Wechsler Abbreviated Scale of Intelligence (WASI), Stroop Colour-Word Interference Test, Trail Making Test, California Verbal Learning Test (CVLT) and RCFT. Kozora et al. 29 established the reliability and validity of the ACR-SLE neuropsychological battery and found that 7 of 12 measures from the ACR-SLE neuropsychological battery were lower in patients with SLE compared to controls. In a sample of Caucasian patients with SLE and healthy women, Peretti et al. 30 found that patients with SLE had significantly greater cognitive impairment on the WASI (Visual Back-span and Digit Symbol Substitution), Trail Making Test A and B, and Stroop Colour-Word Interference Test compared to controls. In this study, elevated antinuclear antibody (ANA) significantly correlated with impairment in the WASI visual span, WASI visual back-span and cancellation task, whereas elevated anti-double-stranded DNA antibody correlated with impairment in the Trail Making Test A and WASI auditive back-span. In a sample of Portuguese patients, patients with NPSLE were significantly impaired in the Auditory Verbal Learning Test and Trail Making Test – Part A compared to SLE patients without overt CNS involvement and healthy controls. 31 Table 1 summarises the neuropsychological battery used to assess SLE patients in studies before and after the development of ACR-SLE neuropsychological battery.4,9,17,21,22,29–44
Summary of neuropsychological battery used to assess SLE patients in studies before and after the development of ACR-SLE neuropsychological battery.
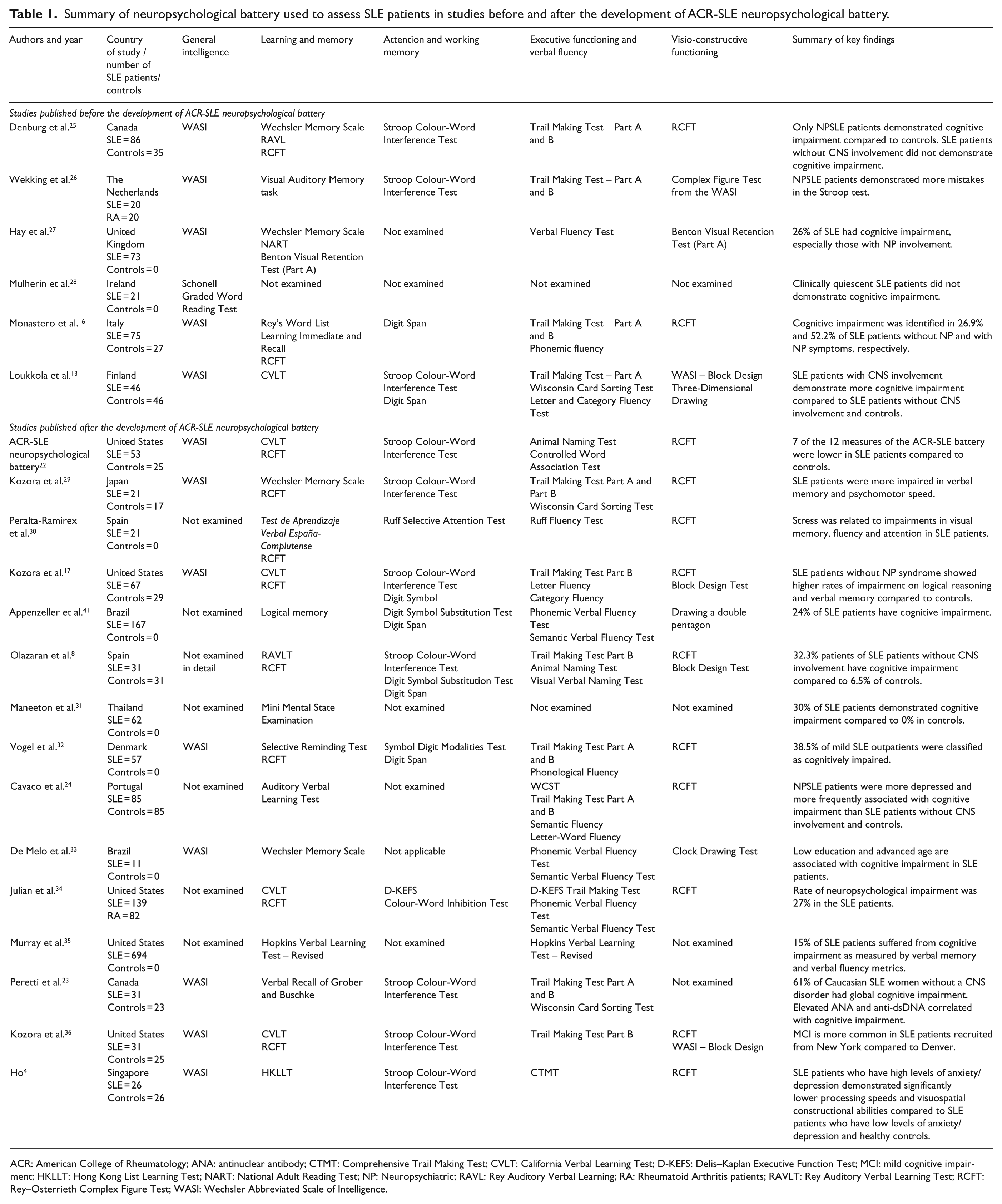
ACR: American College of Rheumatology; ANA: antinuclear antibody; CTMT: Comprehensive Trail Making Test; CVLT: California Verbal Learning Test; D-KEFS: Delis–Kaplan Executive Function Test; MCI: mild cognitive impairment; HKLLT: Hong Kong List Learning Test; NART: National Adult Reading Test; NP: Neuropsychiatric; RAVL: Rey Auditory Verbal Learning; RA: Rheumatoid Arthritis patients; RAVLT: Rey Auditory Verbal Learning Test; RCFT: Rey–Osterrieth Complex Figure Test; WASI: Wechsler Abbreviated Scale of Intelligence.
The European League Against Rheumatism (EULAR) task force published a set of evidence-based recommendations, addressing diagnostic and therapeutic requirements for NPSLE. 45 The EULAR recommended that SLE patients with severe cognitive impairment should undergo neuropsychological tests administered by neuropsychologists. Pamfil et al. 46 performed a chart-based review in two European centres and found that only 27.8% of SLE patients with severe cognitive impairment underwent formal neuropsychological assessment. Small number of SLE patients could receive neuropsychological assessment because such assessment is very time-consuming which often results in long waiting time. Neuropsychological assessment has other limitations including standardisation for English-speaking patients only, inability to detect reaction time and large practice effects on repeated testings. 47 It poses a challenge to administer traditional neuropsychological assessment to non-English-speaking patients with SLE.
Computerised neuropsychological assessment
To overcome the language barrier and practice effect, the Automated Neuropsychological Assessment Metrics (ANAM) was validated to assess cognitive function of patients with SLE. 47 The ANAM is a computerised programme which assesses simple reaction time, continuous performance (vigilance/sustained attention), code substitution (visual scanning and learning) with immediate and delayed memory (non-verbal memory), simultaneous spatial processing (visual perception and mental rotation), Sternberg test (sustained attention/working memory), digit span (working memory/span of attention) and matching to sample test (visuospatial perception and working memory). 47 The paediatric version of ANAM was validated in children suffering from childhood-onset SLE. 48 The ANAM is a time- and cost-efficient tool for screening and monitoring cognitive functioning in patients with SLE. 49 Efforts must be made on validating new assessment tools since the ACR battery is time-consuming.
Neuroimaging
Magnetic resonance imaging (MRI) is a non-invasive investigation to assess patients with SLE. Besides structural MRI, other MRI techniques including the fluid-attenuated inversion recovery (FLAIR) scan can detect lesions with hyperintensities. 50 The diffusion-weighted imaging (DWI) is able to detect white matter lesions. 50 Functional MRI (fMRI) scan specifically records blood-oxygen-level-dependent (BOLD) signal which is a measure of neuronal metabolism and proxy measure of neuronal activity. 51
During the working memory task, NPSLE patients showed greater frontoparietal activation than healthy controls. 52 During the executive function task, an increase in the contralateral cerebellar-frontal activity was found to compensate for the compromised cortico-basal ganglia-thalamic-cortical circuit in SLE patients in order to maintain their cognitive test performance as comparable with healthy controls. 53 During the sustained and divided attention task, patients with SLE presented with significantly expanded areas of activation in the frontal-parietal lobes, and these activated areas exhibited significantly higher functional connectivity strength in SLE patients during the resting state. 54 The above functional imaging studies showed that SLE patients without overt CNS involvement recruited additional pathways to execute goal-directed tasks to compensate for their reduced strategic planning skill despite clinically sufficient control in SLE disease activity. 55
Underlying pathology and other factors causing cognitive dysfunction in patients with SLE
Immunological factors
The neuropathology behind cognitive dysfunction in SLE is a result of dysfunction involving immune cells, cytokines, chemokines and Abs in important neuroanatomical structures. 8 In lupus-prone MRL-lpr mice, leukocyte infiltration, intrathecal autoantibody and pro-inflammatory cytokine synthesis lead to progressive neurodegeneration.56,57 Peripheral interleukin (IL)-6 level has an inverse correlation with memory performance in patients with SLE. 58 SLE is a disease which predominantly affects women and oestrogen may play in role in its pathogenesis. Cunningham et al. reported that oestrogen receptor alpha deficiency provided significant protection against cognitive dysfunction in mice. The oestrogen receptor alpha deficiency suppresses pro-inflammatory actions of microglia in autoimmune disease. 59
Among all Abs, anti-N-methyl-D-aspartate receptor (NMDAR) and anti-ribosomal-P Abs contribute to cognitive dysfunction in patients suffering from SLE. 60 Anti-NMDAR and anti-ribosomal-P Abs recognise neuronal surface antigens 60 that are distributed in neuroanatomical areas involved in memory, cognition and emotion.61,62 APL Abs play a pathogenic role in cognitive dysfunction by causing microvascular thrombosis. 63
Matrix metalloproteinases (MMPs) are a family of zinc- and calcium-dependent endoproteinases that mediate degradation and remodelling of the extracellular matrix proteins. 64 In the immune system, MMP-9 is secreted by neutrophils. 65 MMP-9 can degrade components of basal lamina 66 and disrupt blood–brain barrier. 64 Such disruption allows anti-NMDAR Abs to cross the blood–brain barrier and influence cognitive function. 66 NPSLE patients with cognitive dysfunction have significantly higher concentrations of serum MMP-9 than SLE patients with normal cognitive function. 67
Damages in neuroanatomical structures
The presence of cerebral atrophy increases the risk of cognitive dysfunction in patients with SLE. 68 Patients with SLE have more microstructural white matter damage than general population. 69 Cognitive dysfunction in patients with SLE is associated with damages to white matter tracts including corpus callosum 12 and grey matter. 70 The association between cognitive dysfunction and white matter and grey volume loss was found in patients with childhood-onset SLE. 71
White matter is subcortical and white matter tract contains nerve fibres. Patients with SLE demonstrated lower white matter volume (WMV) than healthy controls in the superior longitudinal fasciculus, cingulum cingulate gyrus and inferior fronto-occipital fasciculus. 72 In diffusion tensor images (DTI), decreased fractional anisotropy in the superior white matter pathways were significantly associated impairment in executive function in patients with NPSLE. 73 Cognitive dysfunction and mood disorder in SLE were associated with lower white matter magnetization transfer ratio histogram peak heights in magnetization transfer imaging (MTI). 74 Poor working memory performance was correlated with higher left frontal white matter choline-to-creatinine (Ch/Cr) ratio in magnetic resonance spectroscopy (MRS), signifying damage in the left frontal microstructural white matter. 75 The change in WMV was associated with SLE disease duration. 71 Treatment with immunosuppressant, especially pulse corticosteroids, could reduce the loss of WMV in adult patients with SLE.71,76
Grey matter is found on the cortical surface of the brain (cortical) and contains the cell bodies of neurons. Patients with SLE were found to have lower grey matter volume (GMV) than healthy controls in the middle cingulate cortex, middle frontal gyrus and right supplementary motor area. 72 Patients with SLE were found to be impaired in the immediate visual spatial memory compared to healthy controls. 24 Impairment of visuo-constructional performance was associated with GMV loss in the orbitofrontal, dorsolateral and prefrontal cortex, as well as anterior cingulate cortex. 71 In a longitudinal study, reduction in SLE disease activity was accompanied by region-specific GMV improvement in the prefrontal regions. 72
Damages in specific neuroanatomical structures are responsible for cognitive dysfunction in SLE. The cornu ammonis 1 (CA1) is a key area within the hippocampus responsible for learning and novelty detection by comparing stored information from the dentate gyrus and new information from the entorhinal cortex.16,77 Patients with SLE showed reduced volume of CA1 compared to healthy controls and small CA1 volume was associated with cognitive dysfunction. 78 Amygdala is continuous with the tail of caudate nucleus and produces fear response and emotion. 16 Watson et al. 12 highlighted the relationship between anti-NMDAR Abs and amygdala dysfunction in animal model of lupus. The negative impact on attention and memory is related to negative emotion experienced by patients with SLE.
Clinical factors
Clinical factors play a key role in the aetiology of cognitive dysfunction in SLE. Hay et al. 34 studied 73 patients with SLE and reported that cognitive dysfunction was found in 26% of SLE patients and was associated with clinical evidence of CNS involvement. Besides CNS involvement, researchers also found other clinical factors associated with cognitive dysfunction in SLE patients, including hypertension and stroke. 42 In SLE patients without overt CNS impairment, the severity of cognitive dysfunction was associated with high disease activity of SLE 79 and low exercise capacity. 80 Long duration of SLE, previous history of major cerebral involvement and presence of multiple medical complications contribute to higher rate of mild cognitive dysfunction in patients with SLE.43,81
Depression as a confounder between SLE and cognitive dysfunction
Depression is a confounder that influences both SLE and cognitive dysfunction. Depression is a syndrome comprising other symptoms including insomnia, early morning awakening and anhedonia. Sleep disturbance may also be a confounder to cognitive dysfunction. When comparing the cognitive function in SLE patients and healthy controls, SLE patients with high levels of anxiety/depression demonstrated significantly lower processing speeds and visuospatial constructional abilities compared to SLE patients with low levels of anxiety/depression and healthy controls. 82 Vogel et al. 39 reported that depression demonstrated a stronger association with subjective experiences of cognitive dysfunction compared to the actual cognitive functioning of SLE outpatients with low levels of disease activity. Monastero et al. 21 found that severity of depression was the only clinical factor that significantly predicted cognitive dysfunction in SLE patients with and without CNS involvement. Peralta-Ramirez et al. 37 found that daily stress was related to impairments in visual memory, fluency and attention in patients with SLE. Petri et al. 83 proposed that treatment of depression in SLE patients with moderate to severe depression may improve their cognitive functioning. For patients with SLE, depression and cognitive impairment may share the same underlying pathophysiological mechanisms because a common genetic vulnerability related to serotonin and inflammation between depression, cognitive impairment and vascular diseases were found.84,85 Furthermore, high circulating levels of pro-inflammatory cytokines such as IL-6 were associated with memory impairment in patients with SLE 58 and depression. 86 Nevertheless, the ANAM has the ability to predict neuropsychological functioning after controlling for severity of depressive symptoms. 49
Challenges in diagnosis and management
Challenges in neuroimaging and opportunities
The common MRI techniques including FLAIR, DWI and fMRI are mostly restricted to the research settings rather than clinical applications. In addition, MRI scan has several barriers. First, these techniques require the participants to be in a supine position with their head restrained and without any movement of the body. 87 Second, the scanning procedures are typically conducted by radiographers, with high costs incurred by each measurement. 88 In Asia, clinicians have used functional near infrared spectroscopy (fNIRS) which measures BOLD signals in assisting diagnosis, predicting clinical outcomes and treatment outcomes in NP illnesses.82,89 fNIRS may have potentials to assess functional changes in patients with SLE and further research is required.
Challenges in pharmacological treatment and opportunities
The EULAR recommended that the management of cognitive dysfunction should include managing the SLE and non-SLE associated factors, as well as psycho-educational support. In terms of psychopharmacology, corticosteroids remain the mainstay of evidence-based treatment for SLE but it is associated with organ damages, cataracts and osteoporotic fractures. 15 The relationship between corticosteroid and cognitive dysfunction remains a controversial topic. SLE patients without past NP history and exposure to corticosteroids showed significant impairments compared to healthy controls on executive function, complex attention, immediate recall and psychomotor speed. 79 There are counter-evidences which demonstrated that cognitive dysfunction was not caused by corticosteroid. Previous study showed that SLE patients without corticosteroid use and overt CNS involvement had significantly higher prevalence of cognitive dysfunction than healthy controls. 79 SLE patients with and without corticosteroid use were found to have poorer decision-making capacity than healthy controls. 90 Similarly, cognitive outcomes in children with lupus nephritis were found to be better than children suffering from other glomerular chronic kidney diseases due to greater uses of corticosteroids and chemotherapeutic agents. The use of prednisolone was found to be independently associated with better executive function, leading to better health-related quality of life in children with lupus nephritis. 91
As depression is an important confounder contributing to cognitive dysfunction, 83 antidepressant treatment may improve cognitive function in patients with SLE. Furthermore, antidepressants reduce pro-inflammatory cytokines including IL-6. 92 Other novel treatments have been proposed to treat cognitive dysfunction associated with SLE. As pro-inflammatory cytokines are involved in cognitive dysfunction, anti-cytokine therapies may provide benefits through centrally medicated process. 8 Deposition of 16/16 Id antibodies were found in the brains of patients suffering from NPSLE which caused impairment in visual and spatial memory. 93 Anti-16/6 Id antibody was found to increase astrocyte number in the hippocampus of mice 94 and offers the potential as novel treatment for cognitive dysfunction associated with SLE.
Petri et al. 95 conducted a randomised, double-blind, placebo-controlled single centre 12-week trial in patients with SLE and reported that patients treated with, an NMDAR antagonist, memantine did not exhibit significant improvement in cognitive function compared with the placebo group. This finding should be interpreted with caution because one quarter of participants did not show objective evidence of cognitive impairment at baseline. Furthermore, the most commonly used cognitive enhancer, acetylcholinesterase inhibitors have not been assessed in any randomised controlled trial involving SLE patients with cognitive dysfunction. Further research is required to evaluate other cognitive enhancers.
Non-pharmacological treatment
Not all therapies on cognitive dysfunction need to be immunosuppressive. Since cognitive dysfunction in SLE patients is correlated with 6-minute walk test and lung function test, Kozora et al. 80 proposed that supervised training programme with regular and strenuous exercise may be a cost-effective approach to improve cognitive function of SLE patients. Other non-pharmacological interventions include cognitive behaviour therapy and cognitive rehabilitation.
Cognitive behaviour therapy
Patients with SLE often encounter difficulty coming to terms with illness perception and functional losses. They often develop negative ruminations which interfere with cognitive function. 4 Psychotherapy can help patients with SLE to cope with their negative emotions which indirectly improve their cognitive functioning. Patients with SLE should go through a series of behaviour therapy including stress management and relaxation exercises. Cognitive therapy can address cognitive errors and ruminations leading to negative perceptions of self and adjust their premorbid expectations. Behaviour therapy can help patients to develop a structured routine and regain control in the chronic course of SLE.
Cognitive rehabilitation
Cognitive rehabilitation consists of psychoeducation, use of memory aids, prioritisation, time optimisation and cognitive training exercises. Psychoeducation provides information to patients with SLE on the basic mechanisms of cognitive function and how SLE causes cognitive dysfunction in lay language. For memory aids, SLE patients with cognitive dysfunction are encouraged to use written reminders or smartphones to prompt them. Memory aids help patients to stay focused and maintain their activities of daily living. Visual cues can help patients to retrieve information from their memory. Due to cognitive dysfunction, patients with SLE need to prioritise their tasks of the day and focus on one task before moving on to the next. Time optimisation involves engaging cognitively challenging tasks in the early part of the day and more relaxed task towards the end of the day. Engagement in cognitive training exercises including Sudoku, chess, Risk and Mahjong (for Asian patients) will enhance their executive function and problem-solving skills.
Conclusion
Cognitive dysfunction is a common phenomenon in SLE patients with and without overt CNS involvement, leading to negative impacts on social functioning and quality of life. SLE patients with overt CNS involvement, the presence of depression, vascular risk factors and higher disease activity are at greater risk to develop cognitive dysfunction. Specific cognitive dysfunctions associated with SLE include impairment in attention, memory and visuospatial process. SLE patients may achieve similar cognitive test performance compared with healthy controls. Functional imaging studies found that patients with SLE, especially those without overt CNS involvement had compensatory mechanism including greater frontoparietal activation to maintain their cognitive test performance. Pro-inflammatory cytokines such as IL-6, Abs including anti-NMDAR, anti-ribosomal-P and APL Abs, and MMP-9 play a key role in the pathology behind cognitive dysfunction by damaging important neuroanatomical structures such as the hippocampus and amygdala. Imaging studies demonstrated that patients with SLE had volume loss in white matter and grey matter. Corticosteroid treatment may reverse volume loss in the white matter. The diagnosis of cognitive dysfunction of SLE faces several challenges. The traditional neuropsychological assessment recommended by ACR and EULAR is time-consuming and validated for English-speaking patients. Computerised neuropsychological assessment offers a cost-effective alternative. The conventional neuroimaging method using MRI is expensive and most of the recent discoveries in functional neuroimaging are limited to research settings. For pharmacological treatment, corticosteroids and antidepressant may improve cognitive dysfunction. Novel treatment including anti-cytokine therapies and cognitive enhancers require further study. Non-pharmacological interventions including cognitive behaviour therapy and cognitive rehabilitation are helpful to improve cognitive function in patients with SLE.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
