Abstract
Pigs are the most promising models for the study of wound healing and hypertrophic scarring because they are anatomically and physiologically similar to human beings. The Red Duroc pig and Mini Bama pig are two swine models that have attracted a lot of attention. The aim of the present study was to examine and compare the scarring process in a red Duroc pig and a Mini Bama pig, providing knowledge for researchers and clinicians to enable them to choose the most suitable pig model for studies.
Lay Summary
Pigs, with an anatomical and physiological skin architecture similar to that of the human, have been studied extensively as models for wound healing and scarring. However, the red Duroc pig, as a frequently used animal model for human wound/scar-related research, has the shortcoming of rapid growth in size and weight, which gives rise to difficulty in its handling and brings about enormous cost. The Mini Bama pig grows much slower and can be a potential replacement. This study compares wound healing and subsequent scarring between the Duroc and Mini Bama pigs with a time span of 35 weeks. Other than clinical appearance at week 35, when the Duroc scars showed an obvious hyperpigmentation while the Mini Bama scars showed slight hypopigmentation, the trends of change in shape, histological appearance (epidermis thickness) and collagen deposition in the tissues are similar in both types of pigs.
Introduction
Hypertrophic scars, formed by aberrant dermal wound healing, often lead to cosmetic and functional problems. 1 Existing therapies are insufficient to cure this disease due to the incomplete understanding of its underlying mechanism.2,3 Furthermore, these methods, usually complex and multi-staged, are ineffective with potential side effects. 4 To understand the progress of scar formation so as to develop an effective anti-scar therapy, preclinical animal models are of great importance.
Pigs are the most promising models for the study of wound healing and hypertrophic scarring because they are anatomically and physiologically similar to human beings. 5 They have a relatively thick epidermis, dermal papillae and dense elastic fibres in the dermis. The biochemical structure of porcine collagen is also similar to that of human collagen. A handful of studies have used porcine models to evaluate the effects of myriad treatments and devices, including surgical and enzymatic debridement agents, negative pressure devices, silver dressings, collagen gel dressings, sprayed cell suspensions and dermal substitutes. 6
Currently, the red Duroc pig, a heavy and cumbersome animal, is mostly used as a swine model for hypertrophic scarring.7–9 However, the rapid growth of their body size and weight during the experiment has created problems in both the experimental procedure and the analysis of the results. 10 Naturally, the Mini Bama pig, characterised by its small size, slow growth, good genetic stability and minor individual differences, has been proposed as a replacement for the red Duroc pig. 11 Studies have also shown that skin stratification, thickness and structure of the Mini Bama pig are similar to those of human skin. Recently, Deng et al. 11 created a full-thickness dermal burn injury on the Guangxi Bama minipig that resulted in hypertrophic scars. Using this model, they confirmed the anti-scar effect of Shikonin, an active component extracted from Radix Arnebiae that has been studied to induce apoptosis and reduce the production of collagen in hypertrophic scar-derived fibroblasts. 12
While it is exciting to see the potential of the Mini Bama pig as an alternative pig model, there is a lack of understanding about the similarity and difference of the hypertrophic scarring process between the red Duroc pig and the Mini Bama pig. This knowledge is necessary for researchers and clinicians to be able to choose the most suitable pig model for their studies. Therefore, the present study answers this critical question by a direct comparison of the scarring process in two pig models.
Eight nearly full-thickness defects were first made on the backs of the pigs and the wounds were left to heal naturally over the following 35 weeks. We utilised three-dimensional (3D) imaging and histology to examine both the appearance of the wounds/scars and the underlying tissue during this process. The wound area in both types of pigs shrank during the healing process and the shape of the wounds changed from a regular square to a spindle shape. The whole healing process took nine months. Haematoxylin and eosin (H&E) staining, Masson Trichrome staining and immunohistochemistry collagen I staining were used to study the wound/scar tissue. We observed an increase in the thickness of the epidermis from week 8 to week 35 post-surgically and an increase in the levels of collagen and fibre sizes in the dermis for both the Duroc and Mini Bama pigs, indicating the formation of the scar tissue.
Material and methods
Animal
The animal experiment was carried out in compliance with the rules of the Animal Care Committee of SG Med International Pte Ltd (SGG, IACUC-2018032). The female Red Duroc pig was 12 weeks old and had a body weight of 30.50 kg. The female Guangxin Mini Bama pig was the same age and had a body weight of 10.20 kg. The animals were acclimatised for seven days with 12 h light/dark cycles and fed with lab porcine grower diet and water ad lib.
Wound creation for scarring
General anaesthesia was administered with 5 mL xylazine (100 mg/mL), dose = 1 mL/18 kg BW. The hair on the back of the animals was clipped and the skin was scrubbed with soap, followed by alcohol and povidone-iodine. Eight nearly full-thickness defects (7 × 7 cm) were made with four defects at each side of the posterior midline with a distance of at least 7 cm from each other (Scheme 1). The wounds naturally granulated and re-epithelialised for 35 weeks. In order to monitor the development of the scars, biopsies were done before the surgery and at day 10, week 4, week 8, week 23, week 24, week 25, week 27, week 31 and week 35 after the surgery for both pigs. The animals were sacrificed at week 35 after the biopsies were done.

Wound distribution scheme. Eight wounds with nearly full-thickness defects were made on the dorsal skin with a dimension of 7 × 7 cm.
Biopsy
The biopsied tissues were punched out by disposable and sterilised punch with a diameter of 5 mm. All biopsied tissues were fixed in neutral buffered formalin and then embedded in paraffin.
H&E staining
Sections of 4 µm in thickness were cut vertically to the skin surfaces and stained with H&E staining. Briefly, the sections were deparaffinised and re-hydrated and subsequently stained with H&E. Dehydration and mounting were done last to preserve the sample for imaging.
Masson Trichrome staining
The paraffin embedded samples were sectioned with a thickness of 10 µm using microtome (RM2255, Leica). The sections were then deparaffinised and hydrated for Masson Trichrome staining (ab150686, Abcam). Briefly, the slides were incubated in preheated Bouin’s Fluid for 60 min under a temperature of 60 °C and followed by a 10-min cooling under room temperature. The slides were rinsed in water and stained in haematoxylin, Biebrich Scarlet/Acid Fuchsin solution and Aniline Blue. Finally, the slides were dehydrated and mounted for imaging.
Immunohistochemistry staining of collagen I
The paraffin-embedded samples were sectioned with a thickness of 10 µm using microtome (RM2255, Leica). The sections were then deparaffinised and hydrated for immunohistochemistry staining. The samples were blocked using 3% BSA for 1 h at room temperature. Antigen retrieval was performed by incubation with pepsin (0.5% in 5mM HCl) for 20 min at 37 °C. Samples were incubated with the primary antibody (ab34710, Abcam) (1/500) for 1 h at room temperature. A Goat Anti-Rabbit Alexa Fluor®488 (ab150077, Abcam, 1/1000) was used as the secondary antibody.
Setting of 3D imaging and analysis
In order to obtain an accurate quantitative measurement of the scar, 3D photographs were taken every week throughout the experiment by Vectra H1 (Canfield). The 3D images were taken according to the VECTRA H1 user guide. Briefly, the pigs were in a resting state and the camera was held directly above the area of wound to be captured. Two green lights were emitted from the camera when it was on. The images were captured when two green dots converged to a single point by adjusting the distance from the camera to the subject. The absolute areas of the wounds/scars were measured using the software associated with the camera.
Epithelial thickness measurement
The measurement of the epithelial thickness histologically has been done by ImageJ as mentioned in the literature.13–16 To better avoid subjectivity and increase accuracy, ImageJ was used to drop a line perpendicular from the surface of the skin to the basement membrane to measure the distance. This was repeated six times on each skin section at six different spots.
Results
Creation of wounds on red Duroc and mini Bama pigs
General anaesthesia was administrated to bring the red Duroc and Mini Bama pigs into a resting state. Arms and legs were settled by tying them to the operating table as a safety procedure to reduce the movement of the pigs in case of sudden revival from the anaesthetic. Four squares of 7 × 7 cm at each side of the posterior midline were drawn by the marker and labelled with L1-L4 and R1-R4 as references for the exact area to be excised (Figure 1a). Eight near-full thickness defects were made by surgically removing the skin in the labelled area by scalpel (Figure 1b). The pigs were transferred to separated holding rooms after surgery and taken care of to make sure there were no infections on the wounds. All pigs were revived after surgery with clotted blood on the wounds and no infections were observed on the wounds.

Excision of nearly full-thickness skin on the Mini Bama and Duroc pigs. (a) Pig labelled with a marker to indicate the wound area on the day of operation. (b) The wounds were created by excision of the nearly full-thickness skin.
The appearance of the wound was monitored until the necrotic scab completely fell off and the wound was healed (Figure 2). In week 1, the granulation tissue and scab were formed on both the Duroc and Mini Bama pigs. In week 2, a full and thick scab was formed on both types of pigs. The scab was observed to fall off completely in week 8 for the red Duroc pig and in week 12 for the Mini Bama pig. The presence of a tight scab on a part of the wound was considered as not completely healed. 17 We observed that the red Duroc pig healed faster than the Mini Bama pig regarding the falling-off rate of the scab. Hair loss, contraction and hyper/hypopigmentation on wound tissues were observed in both the Duroc and Mini Bama pigs. There was no hair growth on scar tissues for both pigs. Scar tissue changed its appearance from a square shape to a four-pointed star shape from week 8 to week 35, which is due to the contraction of the tissue in the remodelling phase of wound healing when myofibroblasts execute contraction activities and drag the lesion boundaries toward the centre. 18 In week 23, melanin was observed on the wound area on the red Duroc pig. However, the skin tone was slightly lighter than the surrounding normal skin on the Mini Bama pig.

Hypertrophic scar formation after the full-thickness excision of the skin on (a) the Duroc pig and (b) the Mini Bama pig at 35 weeks.
The absolute areas of the wounds/scars were measured on 3D images captured by VECTRA-H1 camera (Figure 3). The camera-associated software (Face sculptor, VECTRA analysis module) allowed accurate measurement of the selected area. Before the scab fell off, the wound/scar area was defined by the scab covering the area. When the scab started to fall off or completely fell off, the edge between the normal tissue and wound area could be easily distinguished by comparing the skin tone as well as the contraction mark. Overall, the wound area kept shrinking during this healing process. Contraction happened at the proliferation stage where myofibroblast accumulated at the wound borders to execute contractive activities and dragged the borders towards the centre. 18 There was an enlargement (15.5% for the red Duroc pig and 32.6% for the Mini Bama pig at the end of week 35 [27.5 cm2 and 24.8 cm2, respectively] compared to week 12 [23.8 cm2 and 18.7 cm2, respectively]) of the wound area for both the red Duroc and Mini Bama pigs, which is anastomotic to the remodelling phase of the wound healing process where the skin is attempting to recover its normal tissue structure.

Absolute wound area versus time quantified through 3D imaging.
The normal skin tissue of the pig consisted of the stratum corneum, epidermis and dermis (Figure 4a). The average thickness of the epidermis layers was 94.6 µm and 70.7 µm for the red Duroc and Mini Bama pigs, respectively (Figure 4b). Skin tissues in both types of pigs showed well-developed rete ridges. Previously, we observed that the red Duroc pig healed faster than the Mini Bama pig regarding the falling-off rate of the scab (Figure 2). H&E staining (Figure 4a) of the wound skin sections confirmed that phenomenon. At week 4, no distinct epidermis layers were formed in either the Duroc or the Mini Bama pigs showing a slower rate of re-epithelialisation than the wounds created by scalding burn for which the re-epithelialisation occurs 7–14 days after wounding. 19 The differences in wound size as well as the cause of the wounds could be the reasons that affect the exact timeline of wound healing. In week 8, the Duroc wound tissue was fully developed with the stratum corneum, epidermis and dermis. There was no significant difference of the epidermis thickness between the normal skin tissue and the scar tissue (Figure 4b). For the Mini Bama pig, however, no obvious stratum corneum and epidermis were formed at the same time (Figure 4a) and thus the measurements of the epidermis thickness were not done for the Mini Bama pig in week 8 (Figure 4b). During weeks 23–35, both the red Duroc and Mini Bama pigs showed an increased thickness of the epidermis (Figure 4b), which correlates to the increasing scar abnormality. 20

(a) H&E staining of normal skin and wound tissues at 35 weeks (scale bar: 100 µm). (b) Quantification of the thickness of epidermis on Mini-Bama normal tissue (week 0), Mini-Bama wound/scar tissue (weeks 4, 8, 23, 27, 31 and 35), Duroc normal tissue (week 0) and Duroc wound/scar tissue (weeks 8, 23, 27, 31 and 35) (n = 18, three skin sections per group, six spots analysed for each skin section).
In the remodelling phase of the wound healing response, abnormal deposition of extracellular matrix (ECM), especially abnormal collagen deposition and reorganisation, accounts for one of the main factors that contributes to abnormal scarring. 21 There are larger collagen contents and fibre size in scar tissues compared to normal tissue. 22 To investigate the collagen contents in the scar/wound tissues and compare those to normal tissues, Trichrome staining was done on both Duroc and Mini Bama skin tissues (Figure S1). At week 8, collagen was visible throughout the sections and showed a disordered pattern for both the red Duroc and Mini Bama pigs. At week 23, collagen was distributed evenly in the tissue of the red Duroc pig, while the collagen fibres form more obvious packed and parallel bundles in the tissue of the Mini Bama pig. At week 35, there was a significant increase in collagen contents in both the red Duroc and Mini Bama pigs compared to normal tissue. The quantification of the collagen in the dermis of the skin tissue for both the red Duroc and Mini Bama pigs was done using ImageJ in normal tissues and scar/wound tissue in weeks 8, 23 and 35. There was an upward trend in the fractions of collagen in the dermis of both the red Duroc and Mini Bama pigs (Figure 5a). Together with the Trichrome staining images, excessive dermal fibrosis is identified in both the red Duroc and Mini Bama pigs, showing the clinical evidence of hypertrophic scarring. 21 Although scar tissue shares the same ECM components as normal skin tissue, the ratios of different collagen are different. For instance, the level of collagen I is elevated in dermal scar tissue. 23 To investigate the collagen I content in the scar/wound tissues, immunohistochemistry staining was performed (Figure S2). Both the red Duroc and the Mini Bama showed an upward trend of collagen I contents from week 8 to week 35. At week 35, the percentage increase of collagen I content was 48.9% compared to normal tissue for red Duroc and 22.7% for Mini Bama (Figure 5b). The increase in collagen I content in both the red Duroc and the Mini Bama indicates the formation of scar tissue.
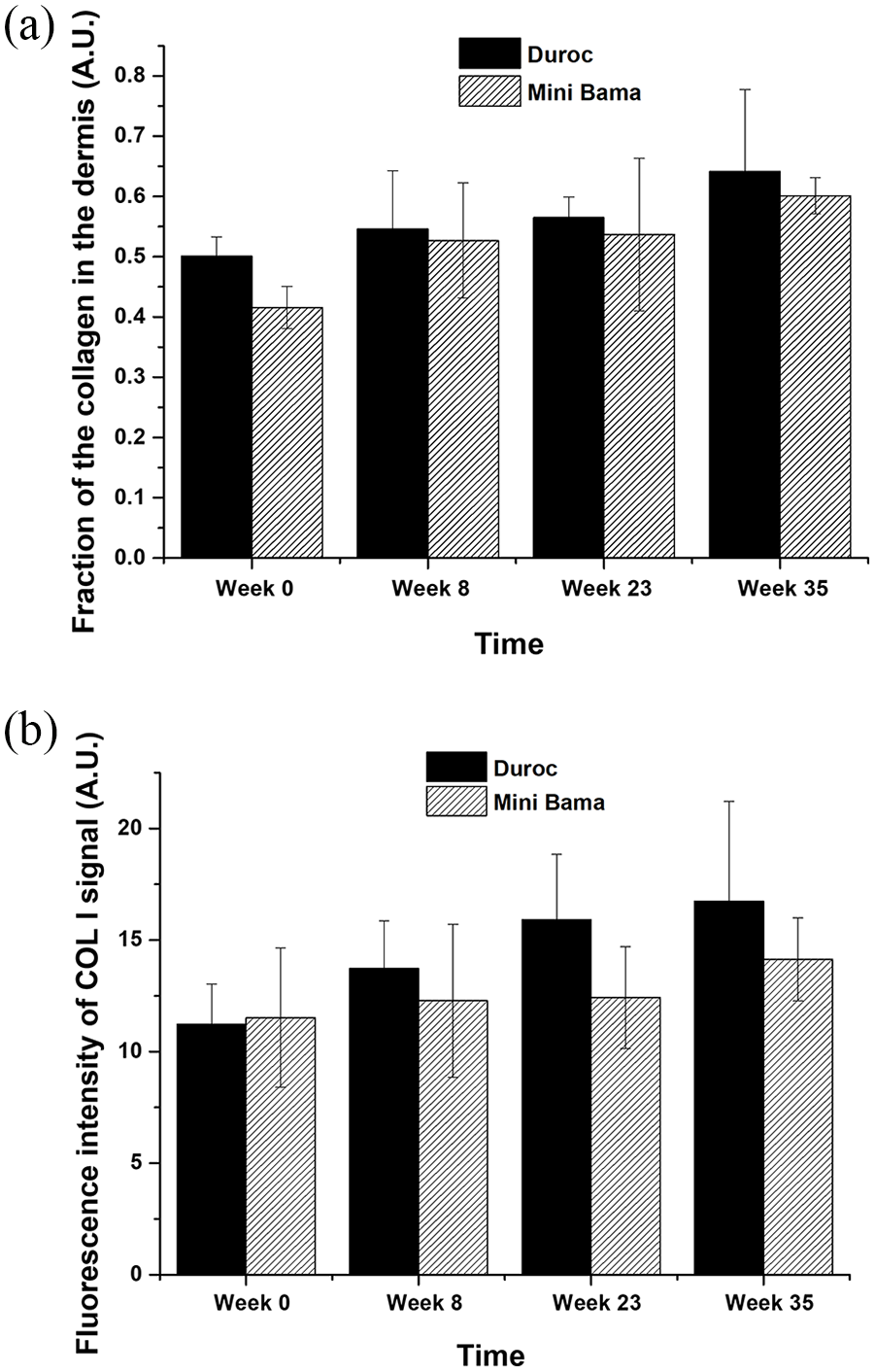
(a) Fraction of the collagen contents in the dermis of the red Duroc and the Mini Bama for normal tissue (week 0) and scar/wound tissue at weeks 8, 23 and 35 (n = 3). (b) Quantification of the Col I signal in the dermis of the red Duroc and the Mini Bama for normal tissue (week 0) and scar/wound tissue at weeks 8, 23 and 35 (n = 3).
Discussion and conclusion
The present study chose the young female red Duroc and Guangxin Bama Minipig (12 weeks) because young pigs are easier to handle in surgery and the cutaneous wound healing rate of the pig decreases with age. 24 The wounds were created by excising the near-full thickness skin because the deeper wound demonstrates more prominent hypertrophic scar symptoms. 17 Eight wounds were located on the backs of the pigs from head to tail, with each wound 7 × 7 cm in size. The rationale for designing large wound areas on the back of the porcine is twofold7,8: (1) the excision of a nearly full-thickness skin defect with a size of around 50 cm2 is a well-established model in hypertrophic scar study, which allows biopsies for continuous observation of histological changes during the course of wound healing; and (2) more skin defects created on one animal can greatly reduce the number of animals used, implying a higher ethical and economical value. After the wounding, both types of pigs healed naturally. However, the red Duroc pig healed faster. The scab was observed to fall off completely in week 8 for the red duroc pig, but only in week 12 for the Mini Bama pig (Figure 2). This was later confirmed through H&E staining (Figure 4). In addition, in week 35, the percentage increase of collagen contents was 48.9% compared to normal tissue for the red Duroc and 22.7% for the Mini Bama (Figure 5b).
In conclusion, other than the clinical appearance at week 35 showing an obvious hyperpigmentation in the red Duroc scars while the Mini Bama scars showed slight hypopigmentation, the trends of change in shape, histological appearance (epidermis thickness) and collagen deposition in the tissues are similar in the red Duroc and Mini Bama pigs. The advanced rate of healing in the red Duroc pig implies faster establishment of the hypertrophic scar while the slow growth rate of the Mini Bama pig makes the handling of the animal much easier.
Supplemental Material
Supplemental material for Comparison of hypertrophic scarring on a red Duroc pig and a Guangxi Mini Bama pig
Supplemental material, Supplemental_material for Comparison of hypertrophic scarring on a red Duroc pig and a Guangxi Mini Bama pig by Xiaoyu Ning, Kai Yang, Wei Shi and Chenjie Xu in Scars, Burns & Healing
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We acknowledge the funding support from Singapore Agency for Science, Technology and Research (A*STAR) Science and Engineering Research Council Additive Manufacturing for Biological Materials (AMBM) program (A18A8b0059), internal grant from City University of Hong Kong (#9610472), and Primary Research & Development Plan of Jiangsu Province of China (BE2016770).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
