Abstract
Catastrophic antiphospholipid syndrome (CAPS) is a lethal disease that occurs suddenly and progresses to multi-organ failure. We present a case of CAPS successfully treated with the rituximab biosimilar CT-P10. A 38-year-old man was referred with a sustained fever and unexplained elevated creatinine levels. Cardiac arrest by ventricular fibrillation occurred upon arrival at the hospital. We diagnosed probable CAPS because of coronary thrombus, renal impairment, suspected diffuse alveolar hemorrhage, and positive anticardiolipin antibody immunoglobulin G. We performed percutaneous coronary intervention for the cardiac arrest, and treated him with extracorporeal membrane oxygenation, mechanical ventilation, and continuous renal replacement therapy. When CAPS was diagnosed, we administered CT-P10 after administering high-dose glucocorticoid. Our case suggests that the use of a rituximab biosimilar is economically efficient in the treatment of CAPS, as in other rheumatic diseases. The patient was cured without recurrence at the 2-year follow-up.
Keywords
Introduction
Catastrophic antiphospholipid syndrome (CAPS) is characterized by multi-organ failure with thrombotic microangiopathy or systemic inflammatory response syndrome (SIRS) associated with cytokine release. About 37% of the patients diagnosed with CAPS do not survive. 1 In the long term, 66% remain thrombosis-free and 17% have APS-related manifestations. 2 There are only a few case reports of relapse. 3 Because of the lower relapse rate and good long-term prognosis, the diagnosis and initial treatment are important. The treatment of CAPS is based on glucocorticoid, plasma exchange, or intravenous immunoglobulin (IVIG). 1
The humanized anti-CD20 monoclonal antibody rituximab has been used increasingly to treat severe autoimmune disease. Rituximab, an anti-CD20 monoantibody, has good efficacy and safety in the treatment of CAPS. 4 Recently, a rituximab biosimilar (CT-P10, Truxima®) has been approved and prescribed in South Korea and Europe. Here, we report a CAPS patient with coronary thrombosis, renal impairment, and diffuse alveolar hemorrhage whose renal function recovered fully after treatment with rituximab biosimilar.
Case report
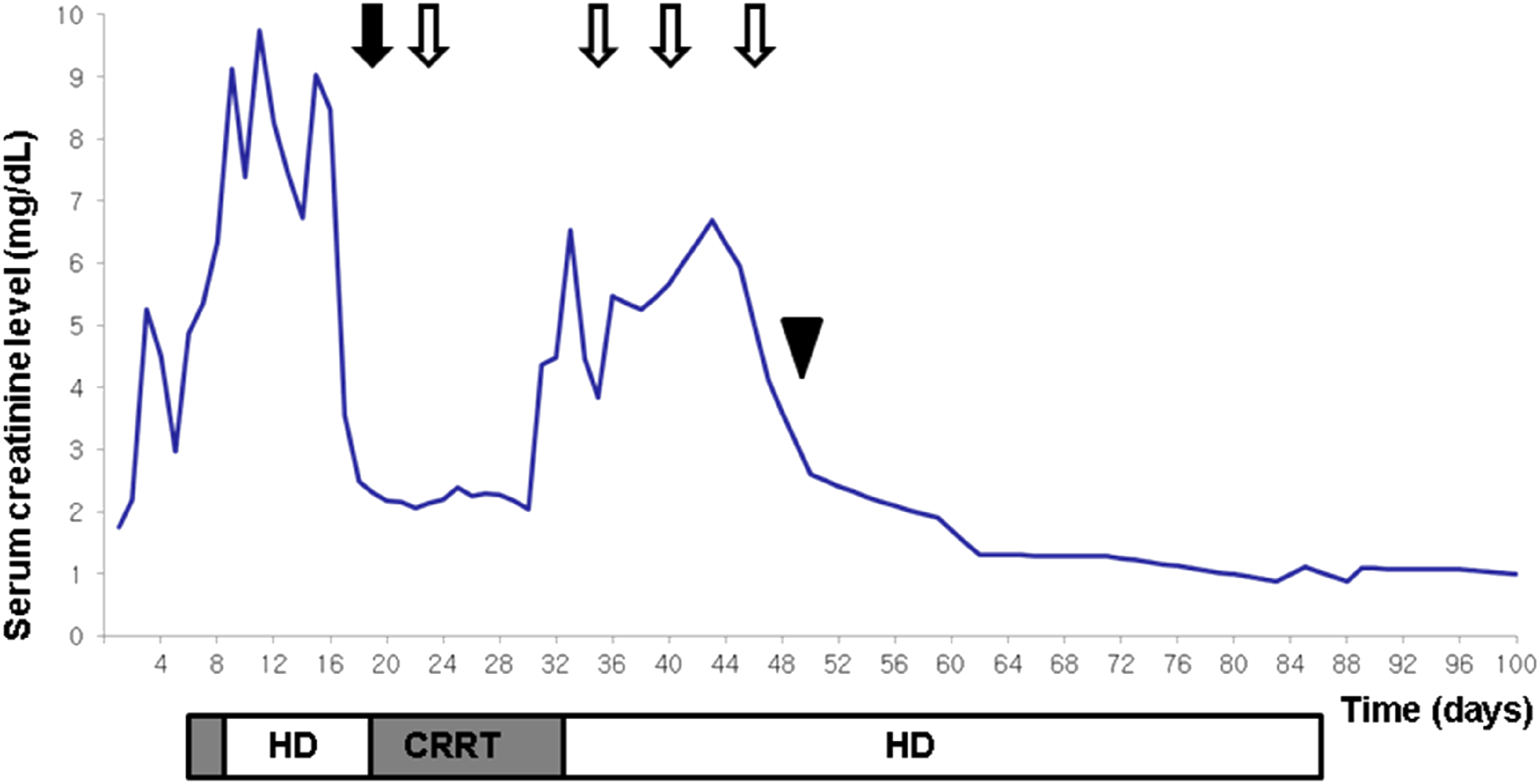
A 38-year-old man was referred to us with a sustained fever and elevated creatinine levels. He had no relevant medical or family history. On admission, he had a cardiac arrest with ventricular fibrillation. Percutaneous coronary intervention and extracorporeal membrane oxygenation (ECMO) were performed. Thrombi were identified in the distal right coronary artery and distal lateral circumflex at percutaneous coronary intervention. Two days after admission, his creatinine level increased from 1.53 to 5.25 (normal 0.5–1.2) mg/dL and the urine output decreased to less than 5 mL/h. Simultaneously, he had a fever of 38°C while on continuous renal replacement therapy (CRRT). After the CRRT was converted to hemodialysis (HD), he was extubated. On day 4, acute respiratory distress syndrome occurred. Chest computed tomography (CT) showed rapid progression of diffuse bilateral parenchymal opacities from day 3 to 8 (Figures 1(a) and (b)). Despite appropriate antibiotic therapy, a fever of 38.6°C reappeared. Repeated blood cultures showed no bacterial growth and bronchoscopy suggested alveolar hemorrhage. Laboratory tests showed a white blood cell (WBC) count of 42,000 (normal 4000–10,000)/μL, hemoglobin of 12.8 (normal 12–16) g/dL, and platelet count of 835,000 (normal 130,000–450,000)/mm3. The erythrocyte sedimentation rate (ESR) was 120 (normal 0–30) mm/h, and C-reactive protein (CRP) was elevated at 25.35 (normal 0.0–0.5) mg/dL. The lactate dehydrogenase 773 (normal 0–250) U/L, aspartate transaminase 52 (normal 0–40) U/L, alanine transaminase 1 (normal 0–41) U/L, blood urea nitrogen 95.3 (normal 6–20) mg/dL, and creatinine (6.78 mg/dL) were checked. Antinuclear antibody was positive at a titer of 1:160. Anti-dsDNA, antiphospholipid, anti-Scl-70, anti-Ro/La antibodies, myeloperoxidase (MPO), PR3 anti-neutrophil cytoplasmic autoantibody, Lupus anticoagulant, anti-β2 glycoprotein I immunoglobulin G (IgG), immunoglobulin M (IgM), and anticardiolipin (aCL) antibody IgM (3.7 mL) were all negative. However, aCL antibody IgG was elevated at 30.1 (normal <20) U/mL. When diffuse alveolar hemorrhage developed, the hemoglobin dropped to 8.0 g/dL and the microangiopathic hemolytic anemia (MAHA) index was positive. Based on the sudden development of cardiac thrombus, renal impairment, suspected diffuse alveolar hemorrhage, MAHA, and positive aCL antibody IgG, we diagnosed probable CAPS and administered high-dose methylprednisolone (1000 mg/day) pulse therapy for three consecutive days. After the glucocorticoid therapy, the pulmonary hemorrhage improved slightly (Figure 1(c)), but the renal impairment persisted and HD was continued. We decided to administer CT-P10 (375 mg/m2) four times a week. After four doses, the ESR and CRP had decreased and the lung manifestations gradually improved (Figures 1(a)–(g)). Simultaneously, the creatinine level decreased and urination started. After 1 month of CT-P10, his renal impairment had recovered fully and we could stop the HD (Figure 2). His creatinine levels have remained normal for 2 years, without recurrence. The patient provided informed consent for publication of the clinical process and images. (a, b) Diffuse bilateral parenchymal opacity rapidly progressed from day 3 to day 8 on computed tomography (CT). (c) After glucocorticoid pulse therapy, chest CT showed that the diffuse bilateral parenchymal opacifications had improved with residual disease. (d) A chest X-ray after the first dose of CT-P10 shows left perihilar infiltration and right cardiophrenic angle haziness. (e) A chest X-ray after the second dose shows decreased left perihilar infiltration. (f) After the third dose, the right cardiophrenic angle haziness was decreased. (g) After the fourth dose, the lung parenchyma was clear bilaterally. Overall treatment course of CAPS. After glucocorticoid pulse therapy (black arrow), four doses of CT-P10 (white arrows) were given. Urination restarted at day 48 (black arrowhead).
Discussion
The mechanism of APS is mainly explained by a two-hit model. The presence of antiphospholipid antibodies is called the “first hit.” When someone with these antibodies is triggered by a “second risk factor” such as pregnancy, infection, inflammation, medical procedure, or trauma, it inhibits natural anticoagulant and fibrinolytic systems, disrupts the endothelium through activation of complement, and initiates thromboses. 5 Some APS patients with widespread thrombotic disease with multi-organ failure are deemed to have CAPS, which is associated with complement activity and complement regulatory gene mutations. 6 Because CAPS is a rare, lethal thrombophilia, early diagnosis, and treatment are important. The diagnosis of definite CAPS is accompanied by antiphospholipid antibodies, with evidence of the involvement of three or more organs within 1 week and confirmation by the histopathology of small vessel occlusion. 7 In our patient, multiple thromboses and MAHA occurred within 1 week, and aCL antibody IgG was positive in the APS antibody panel, although a histological examination was not performed. He was diagnosed with probable CAPS because he did not have histological confirmation of the CAPS diagnostic criteria.
Recently, it has been shown that the B-cell subset expressing CD5 is associated with aCL antibodies, and B-cells produce many antibodies associated with the pathogenesis of APS. It is also known that SIRS is associated with the acute phase reactants (such as cytokines) in CAPS. B-cell ablation therapy in CAPS appears convincing, as it not only removes antibodies but also reduces the production and blood concentration of cytokines. For similar purposes, rituximab was administered to 17 APS patients; the aCL titer decreased and there was no recurrence of thrombosis. 8 In several case reports related to CAPS, the use of rituximab for B-cell ablation caused negative conversion of antiphospholipid antibodies, the normalization of serum creatinine, and improvement in acute respiratory distress syndrome and thrombocytopenia. In the CAPS registry, 15 (75%) of 20 patients treated with rituximab recovered. 7 Rymarz and Niemczyk reported a CAPS patient whom they initially treated with the classic regimen of anticoagulation, glucocorticoids, plasmapheresis, and intravenous IVIG; however, the renal impairment persisted and so they added rituximab, after which the serum creatinine improved. 9 Although no biopsy was performed in our case due to the cascading clinical progression, the clinical features strongly suggested probable CAPS and the patient had complete recovery of renal function and no relapse of thrombosis at the 2-year follow-up after rituximab administration. Recently, secondary antiphospholipid antibody syndrome has been reported with coronavirus disease 2019 (COVID-19). 10 In terms of reducing antiphospholipid antibodies and reducing cytokine release, B-cell ablation therapy should be a good treatment option for secondary antiphospholipid antibody syndrome associated with COVID-19.
Biosimilars such as CT-P10 are currently being developed for use in oncology, endocrinology, and rheumatology. CT-P10 is a rituximab biosimilar developed in 2015 and named Truxima®. Several published clinical trials of CT-P10 demonstrated its non-inferiority and safety. In rheumatoid arthritis and B-cell lymphoma, CT-P10 has equivalent biological activities, efficacy, and safety profile. In addition, the indications and dose used were the same as those for rituximab.11,12 A recent multicenter study of systemic sclerosis showed that CT-P10 caused only transient neutropenia and was similar to rituximab in efficacy and other safety profiles. 13 Biosimilars are biological products that contain the active substance of an already authorized original product. These biosimilar products contain the active substance of the original products, so they have the same effects as the original products. Moreover, they can reduce the cost of treatment, reducing the economic burden of a promising treatment for both the patient and healthcare professionals. 14 In some cases, the effects are similar, but there may be differences in the safety profile. 15 Although still under study, biosimilars may be an important treatment option, even in CAPS.
Conclusion
We successfully treated a case of probable CAPS with glucocorticoid pulse therapy followed by CT-P10. The rituximab biosimilar is a promising treatment for CAPS.
The English in this document has been checked by at least two professional editors, both native speakers of English (for a certificate, please see: http://www.textcheck.com/certificate/3o6ePG).
Footnotes
Author contributions
Changgon Kim, Hyun-Sook Kim: Conceptualization; Changgon Kim: Data curation; Changgon Kim: Writing-original draft; Changgon Kim, Hyun-Sook Kim: Writing-review and editing.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: This study was supported by Soonchunhyang University, Republic of Korea.
Ethics approval
Ethical approval to report this case was obtained from Institutional Reviewer Board (IRB) of Soonchunhyang University Hospital (IRB No.2018-12007).
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
