Abstract
Introduction
The key transformed T cell transcription factor recombination activation gene protein 2 (RAG2) is regulated during inflammation to allow for the acquisition of effector T cells functions. The present study was designed to investigate whether stress signals elicited by leonurine (LEO) could lead to the degradation of RAG2 through v-akt murine thymoma viral oncogene homolog (AKT) signaling in lung fibrosis.
Methods
A total of 120 female mice were randomly divided into five groups (Group I–V): Normal group, bleomycin (BLM), BLM+LEO 50 mg/kg/d, BLM+LEO 100 mg/kg/d, and BLM+LEO 50 mg/kg/d+LY294002. Hematoxylin-eosin, Masson’s, and terminal deoxynucleotidyl transferase dUTP nick-end labeling staining were performed to observe the pathomorphological changes. The expression of CD3+, TGF-β, RAG2, and Bcl proteins was examined by immunodetection, while that of E-cadherin (ECAD), AKT, TGF-β1, alpha-actin-2, Bax, and RAG2 was detected by Western blot analysis.
Results
The level of T lymphocytes was reduced sharply in LEO-treated mice as compared to the other groups. The AKT signal was greatly inhibited in the BLM group and activated with LEO treatment on day 14. In addition, RAG2 was attenuated by LEO on day 14 and day 28. LY294002 could reverse the expression of AKT and RAG2 on day 28. Remarkably, the low dose of LEO has a greater protective efficacy as compared to the high-dose LEO group in terms of pulmonary fibrosis, T cell inactivation, and apoptosis in alveolar cells.
Conclusion
The results of the present study suggested that LEO has a protective effect on lung fibrosis with possible mechanisms of attenuating apoptosis and inflammation via the upregulation of the AKT signal in transformed T cells by suppressing the expression and activity of RAG2.
Introduction
Idiopathic pulmonary fibrosis (IPF) is a common form of interstitial lung disease characterized by alveolar remodeling, respiratory failure, and death, often within 5 years of diagnosis.1,2 Idiopathic pulmonary fibrosis pathogenesis encompasses dysregulated repair responses, fibroblast proliferation, and extracellular matrix deposition.3,4 Although the underlying causes of the disease remain unknown, genetic and experimental evidence support that alveolar epithelial cells apoptosis (abnormal homeostasis) may be intrinsic to IPF disease pathogenesis. 5
The diversity of immune cell wound repair, lung fibrosis, and T helper (Th)2 high inflammation has been recognized as a vital driver for the development of lung fibrosis.6,7 The adaptive immune system plays a crucial role in the defense against damages and the function of highly specific antigen receptors expressed on B and T lymphocytes. In developing lymphocytes, the activity and expression of the recombination activation gene protein 1 (RAG1) and RAG2 endonuclease complex are tightly regulated to ensure ordered recombination of the immunoglobulin genes and avoid genomic instability. 8 Furthermore, lung remodeling and fibrosis are shown to have a correlation with the activation of the v-akt murine thymoma viral oncogene homolog (AKT) signal, which has detrimental effects on cell activation, survival,9,10 and T cell transformation.11–13 It has also been proved that the expression of the key transformed T cell transcription factor RAG2 and T cell differentiation could be influenced by the AKT signal. 14
Leonurine (LEO, 4-guanidino-n-butyl syringate) is the main plant alkaloid present in Chinese motherwort (also called Yi-Mu-Cao in the traditional Chinese medicine) for the treatment of dysmenorrhea, menoxenia, and gynecological disorders in women for hundreds of years. 15 A number of recent studies have demonstrated the protective effects of LEO on ischemia injury models. Although LEO has been widely studied in many medicinal fields, the role in immunoregulation and the exact mechanism on lung fibrosis is yet to be elucidated. The present study was designed to investigate whether stress signals elicited by LEO therapy could lead to negative modulating activity of T cells by suppressing RAG2 through AKT signal in pulmonary fibrosis.
Materials and methods
Experimental pulmonary fibrosis models and the grouping
Leonurine was from MedChemexpress (no. HY-N0741, New Jersey, USA) and the purity was 99.62%. Before use, LEO was added into normal saline to prepare a solution with 0.1 mg/mL concentration. LY294002 (Selleck, Houston, USA) is a specific inhibitor of phosphatidylinositol 3-kinase (PI3K)/AKT.
10
This study was approved (under no. (2018)036) by the medical ethics committee of the First Affiliated Hospital of Fujian Medical University (Fuzhou, Fujian, China). During the experiments, the animals were treated in accordance with the Basel Declaration. All procedures strictly followed the National Institutes of Health guidelines for the care and use of laboratory animals. Kunming mice were derived from Swiss mice. 120 healthy female Kunming mice (6–8 weeks old, 18–22g) were purchased from Shanghai SLAC Laboratory Animal Co.,Ltd., China, (license no: SCXK [hu] 2012-0002). Before and after surgery, 120 female mice were raised in comfortable cages at 22–25°C with adequate food and water. Anesthesia for all procedures consisted of an induction with isoflurane inhalation (3.0%) followed by maintenance of general anesthesia using isoflurane inhalation (2.0%). Pulmonary fibrosis was induced by intratracheal instillation of bleomycin (BLM).
10
Before the operation, 120 qualified laboratory mice were randomly and equally divided into five groups as follows: Group I: Normal group as control. Group II: BLM group: intratracheal injection of BLM (3.5 mg/kg) Group III: BLM+L-LEO group: intratracheal injection of BLM 3.5 mg/kg and preconditioned daily with an intraperitoneal injection of 50 mg/kg LEO. Group IV: BLM+H-LEO group: intratracheal injection of BLM 3.5 mg/kg and preconditioned daily with an intraperitoneal injection of 100 mg/kg LEO. Group V: BLM+L-LEO+LY294002 group: intratracheal injection of BLM 3.5 mg/kg and preconditioned daily with an intraperitoneal injection of 50 mg/kg LEO and 25 µg/g/d LY294002.
At 7, 14, and 28 days after modeling, eight mice from each group were sacrificed by anesthesia for harvesting pulmonary tissue. We euthanized the mice by using fast CO2 CRR (Chamber Replacement Rates) and assured that the mice had no breathing (no thoracic movement) after exposure to high concentrations of CO2. The animal experiment was approved by the Experimentation Ethics Committee on Animal Rights Protection of Fujian Medical University Animal Experiment Center (Fujian, China).
TdT-mediated dUTP-FITC nick-end labeling (TUNEL) assay
The TUNEL assay was performed using the ApopTag Peroxidase In Situ Apoptosis Detection Kit (One Step TUNEL Apoptosis Assay Kit, Beyotime, Haimen, China) to assess programmed cell death.
Immunohistochemical studies
Paraffin-embedded lung tissue sections were deparaffinized in xylene and rehydrated through graded ethanol washes. Antigen retrieval was performed by heating tissue sections for 30 min in Tris-EDTA buffer (10 mmol/L Tris, pH 9.0, 1 mmol/LEDTA). The following primary antibodies were used: anti-CD3+ (ready-to-use, Maixin, Fuzhou, China), anti-TGFβ1 (1:200, Bioworld, Minnesota, USA), and BCL (1:100, Proteintech, Chicago, USA) at 4°C overnight. Subsequently, the sections were incubated with horseradish peroxidase-conjugated secondary antibodies (Dako, Denmark). For negative control, the primary antibody was replaced with species-matched isotype control, and the samples were flushed with PBS, stained with diaminobenzidine (Biotend, Shanghai, China), and counterstained with hematoxylin. Finally, the sections were examined by light microscopy (DWLB2, Leica, Germany). Collagen staining of lung fibrosis lesions was classified into three categories according its intensity by Masson’s trichrome staining as compared to the background staining (strongly positive, weakly positive, and negative).
Western blot analysis
The right lungs were frozen in liquid nitrogen for 15 min and preserved at −80°C. A total amount of 100 mg of right lungs was homogenized in 1 mL of prechilled RIPA lysis solution till pieces. The protein sample was separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis, transferred to a polyvinylidene difluoride membrane, blocked with 5% non-fat milk powder for 1 h, and probed with primary antibodies [1:1000 for TGFβ1 (Bioworld, Minnesota, USA), AKT (CST, Boston, USA), and RAG2 (Abcam, Cambridge, UK), 1:4000 for E-cadherin (ECAD; Proteintech, Chicago, USA) and alpha-actin-2 (ACTA2; Proteintech, Chicago, USA), 1:5000 for BCL2-associated X protein (BAX; Proteintech, Chicago, USA) and β-actinantibodies] for 2 h and secondary antibody (1: 50,000 anti-rabbit IgG) for 1 h. The immunoreactive bands were visualized using the ECL-PLUS chemiluminescence system. Enhanced chemiluminescence signals were detected using a Typhoon 9400 scanner (GE Healthcare, Bethesda, MD, USA).
Statistical analysis
Statistical analysis was performed using the Statistic Package for Social Science 18.0 software package (Statistic Package for Social Science Inc., Chicago, IL, United States). One-way analysis of variance was used for comparison among more than two groups, and Tukey’s post-hoc test was used for the two-by-two comparison of the groups. A level of
Results
Histomorphological observation of animal models
Compared to the control group, the pathological changes in the lungs of the BLM group indicated acute alveolitis between 7 and 14 days. The peak of alveolitis was on day 7 after the BLM injection. The stage of pulmonary fibrosis started from the 14th day after modeling, and typical changes in diffuse pulmonary fibrosis were observed on day 28 in the BLM group. Subsequently, at day 28 day after modeling, hematoxylin-eosin (H&E) and Masson’s staining showed that the pulmonary tissue structure was destroyed, while a part of pulmonary alveoli was fused with the infiltrating inflammatory cells and masses of broad-band–shaped or lamellar-shaped collagen fibers, indicating diffuse pulmonary fibrosis. Compared to the BLM group on day 28, the degree of airsacculitis and fibrosis was obviously lighter in the LEO treatment groups, and LY294002 reversed the effects of LEO (Figure 1). Histological features of lung fibrosis on 28-day-old mice. Different degrees of pulmonary fibrosis were shown by H&E and Masson staining. Typical changes of diffuse pulmonary fibrosis were observed on day 28 in the BLM group lesions, while the lower degree of fibrosis was observed in the LEO treatment groups. LY294002 reversed the effects of LEO. (H&E and Masson staining, original magnification, ×400). The BLM group showed a significant number of apoptosis-positive cells, with a decrease in the BLM+L-LEO and BLM+H-LEO groups by TUNEL staining (TUNEL staining, magnification, ×200). Also, compared to the control group, the expression of CD3+, TGFβ1, and RAG2 was increased in the BLM group, while that of BCL was decreased. Compared to the BLM group, the levels of CD3+ and TGF-β1 were distinctly lower in the BLM+L-LEO and BLM+H-LEO groups, while that of BCL was higher in the BLM+L-LEO group. Compared to the H-LEO group, no obvious changes were detected in CD3+ and TGFβ1 expression in Group V (the BLM+L-LEO+LY294002 group), while there is a lower expression of BCL and a higher expression of RAG2 (immunohistochemistry, magnification, ×400). The horizontal axis represents integral optical density (IOD), and the horizontal axis represents the groups. RAG2, recombination activation gene protein 2; BLM, bleomycin; LEO, leonurine.
Results of TUNEL assay
Few TUNEL-positive cells were detected in the control group. Conversely, the BLM group showed a significant number of apoptosis-positive cells with a decrease in the L-LEO and H-LEO groups (Figure 1).
Immunohistochemistry results
CD3+, TGF-β1, RAG2, and BCL were primarily expressed in the nucleus and cytoplasm of alveolar epithelial cells, as assessed by immunohistochemistry. In the control group, no obvious changes were detected in CD3+, TGF-β1, RAG2, and BCL expression in mouse pulmonary tissue. Compared to the control group, the expression of CD3+, TGF-β1, and RAG2 was increased in the BLM group, while that of BCL was decreased. Compared to the BLM group, the levels of CD3+ and TGF-β1 were distinctly lower in the BLM+L-LEO and BLM+H-LEO groups, while that of BCL was higher in the BLM+L-LEO group. Compared to the H-LEO group, no obvious changes were detected in CD3+ and TGF-β1 expression in Group V (the BLM+L-LEO+LY294002 group), while there is a lower expression of BCL and a higher expression of RAG2 (Figure 1).
Leonurine negatively modulates T cell activity by suppressing RAG2 via AKT signal, which causes lung fibrosis.
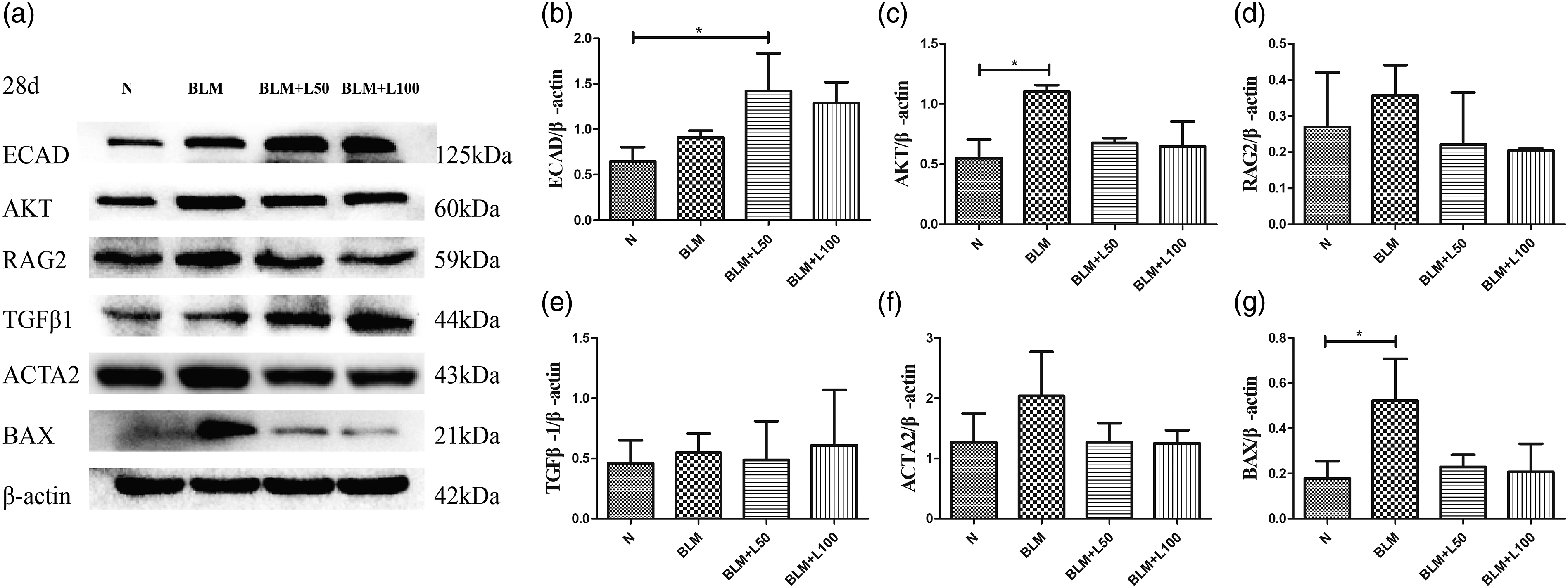
We investigated the expression of AKT, RAG2, TGF-β1, BAX, ECAD, and ACTA2 proteins in lung fibrosis. Although AKT was strongly expressed in the acute alveolitis phrase (Figure 2(a) and (c)), western blot showed that AKT signaling was greatly inhibited in the BLM group and activated with LEO treatment in group III–IV (LEO 50 mg/kg/d and 100 mg/kg/d, respectively) on day 14 (Figure 3(a) and (c)), and the AKT expression was increased compensably in the chronic stage of pulmonary fibrosis on day 28 in the BLM group (Figure 4(a) and (c)). In addition, RAG2 was attenuated by LEO on days 14 (Figure 3(a) and (d) and 28 (Figure 4(a) and (d)). With LEO treatment, the levels of pulmonary fibrosis and apoptosis were decreased by regulation of ECAD, TGF-β1, ACTA2, and BAX (Figure 2(a), (b), and (d)–(f), Figure 3(a), (b), and (e)–(g), Figure 4(a), (b), and (e)–(g)). Remarkably, the high dose of LEO did not have greater protective efficacy as compared to the low-dose LEO group. The optimal dose effect of LEO was attained in 50 mg/kg/d in pulmonary fibrosis. On the early-stage pulmonary fibrosis on day 14, the expression of AKT and RAG2 in the BLM+L-LEO+LY294002 group was lower than that in the BLM+L-LEO group. However, compared to the BLM+L-LEO group on late-stage pulmonary fibrosis on day 28, the AKT expression was lower and the expression of RAG2 was higher in the BLM+L-LEO+LY294002 group. In a word, the expression of AKT and RAG2 could be reversed by LY294002 in late-stage pulmonary fibrosis (Figure 5(a)–(e)). Expression of ECAD, AKT, TGF-β1, ACTA2, and BAX on 7-day-old mice. (a) The electrophoresis data of ECAD, AKT, TGF-β1, ACTA2, and BAX on 7-day-old mice obtained by Western blot. (b–f) Quantitative analysis of ECAD, AKT, TGFβ1, ACTA2, and BAX in each group. AKT signal showed a compensatory increase in the acute alveolitis phase, while LEO treatment reversed the expression of AKT on 7-day-old mice (Figure 2(a) and (c). The levels of TGFβ1 and BAX in the BLM group were significantly higher as those in the L-LEO and H-LEO groups (LEO 50 mg/kg/d and 100 mg/kg/d, respectively) (Figure 2(a), (d), and (f)), while ACTA2 was significantly increased in the BLM and H-LEO groups and decreased in the L-LEO groups (Figure 2(a) and (e)). The trend of ECAD was opposite to that of ACTA2 (Figure 2(a) and (b)). ∗ Expression of ECAD, AKT, RAG2, TGF-β1, ACTA2, and BAX on 14-day-old mice. The electrophoresis results of ECAD, AKT, RAG2, TGF-β1, ACTA2, and BAX on the 14-day-old mice by Western blot. (b–g) Quantitative analysis of ECAD, AKT, RAG2, TGF-β1, ACTA2, and BAX in each group. In the early stage of pulmonary fibrosison day 14, the AKT signal was greatly inhibited in the BLM group and activated with LEO treatment in the L-LEO and H-LEO groups (LEO 50 mg/kg/d and 100 mg/kg/d, respectively) (Figure 3(a) and (c)). Moreover, the increasing degrees of AKT expression were not influenced by the dose of LEO (Figure 3(a) and (c)). RAG2 negatively modulated AKT the signal(Figure 3 (a) and (d)). The levels of RAG2, TGF-β1, ACTA2, and BAX in the BLM group were significantly higher as compared to the L-LEO and H-LEO group (Figure 3(a) and (d)–(g)), while ECAD was decreased in the BLM group and increased in the L-LEO and H-LEO groups (Figure 3(a) and (b)). ∗ Expression of ECAD, AKT, RAG2, TGF-β1, ACTA2, and BAX on 28-day-old mice. Electrophoresis results of ECAD, AKT, RAG2, TGFβ1, ACTA2, and BAX on 28-day-old mice by Western blot. (b–g) Quantitative analysis of ECAD, AKT, RAG2, TGFβ1, ACTA2, and BAX in each group. In the late stage of pulmonary fibrosis on 28-day-old mice, AKT signal has a compensatory increase, while LEO treatment reversed the AKT expression, and the decreasing degrees of AKT expression were not influenced by the dose of LEO (50 mg/kg/d or 100 mg/kg/d) (Figure 4(a) and (c)). The levels of RAG2, ACTA2, and BAX in were significantly higher in the BLM group as compared to the L-LEO and H-LEO groups (Figure 4(a), (d), (f), and (g)), while TGFβ/1 was increased in the BLM group and increased in L-LEO (Figure 4(a) and (e)). The trend of ECAD was opposite to that of ACTA2 (Figure 4(a) and (b)). ∗ Expression of AKT and RAG2 in the BLM+L50+LY294002 group. The electrophoresis data of AKT and RAG2 by Western blot. (b–e) Quantitative analysis of AKT and RAG2 in Group V (LEO 50 mg/kg/d as well as LY294002 5 µg/g/d). On the early-stage pulmonary fibrosis on day 14, the expression of AKT and RAG2 in the BLM+L-LEO+LY294002 group was lower than that in the BLM+L-LEO group. On late-stage pulmonary fibrosis on day 28, the AKT expression was lower and the expression of RAG2 was higher in the BLM+L-LEO+LY294002 group than the BLM+L-LEO group. In a word, the trend of AKT and RAG2 could be reversed by LY294002 in late-stage pulmonary fibrosis (Figure 5(a)–(e)). RAG2, recombination activation gene protein 2; BLM, bleomycin; LEO, leonurine; AKT, v-akt murine thymoma viral oncogene homolog.


Discussion
The present study was designed to emphasize alveolar epithelial cells apoptosis, the immunoregulatory effect of LEO therapy, and the possible immune mechanisms underlying lung fibrosis (Figure 6). TGF-β1 is one of the most important cytokines in the course of pulmonary fibrosis and plays significant roles during inflammation and fibrosis. The TGF-β1 expression in the BLM group was higher than that in the low-dose LEO treatment group (administration of LEO 50 mg/kg/d) at all times, especially on day 7. After low-dose LEO treatment, the degrees of pulmonary fibrosis and apoptosis were decreased via downregulation of TGF-β1 and BAX. Notably, LEO activated the AKT signal by suppressing RAG2 expression and T cell function. The low dose of LEO has a greater protective efficacy as compared to the high-dose LEO group (administration of LEO 100 mg/kg/d) in terms of pulmonary fibrosis, T cell inactivation, and apoptosis in alveolar cells. In Group V (LEO 50 mg/kg/d and LY294002 5 µg/g/d administration), LY294002 reversed the expression of AKT and RAG2 proteins regulated by the low-dose LEO treatment on day 28. Leonurine negatively modulates T cells activity by suppressing RAG2 in pulmonary fibrosis. Leonurine has a protective effect on lung fibrosis via attenuation of alveolar epithelial cells apoptosis, alveolitis, and fibroblasts proliferation. These phenomena are exerted by upregulating the AKT signal in T cells differentiation via suppressing the expression of RAG2. The arrows indicate positive regulation, while the symbol T indicates the inhibition of the target molecule(s). RAG2, recombination activation gene protein 2; LEO, leonurine; AKT, v-akt murine thymoma viral oncogene homolog.
The pathogenesis of IPF is complex and poorly understood. It involves dysregulated repair responses in the context of repetitive alveolar injury, which can lead to alveolar epithelial cells’ apoptosis and pulmonary fibrosis.6,16 AKT targets apoptosis-related proteins, such as the pro-apoptotic proteins BCL-2, caspase-3, and caspase-8. 17 AKT has been proved to be dysregulated in pulmonary fibrosis; also, it has been shown to play a dominant role in profound changes in the differentiation, morphological changes, migration, proliferation, and survival of epithelial cells.5,18 Notably, we observed the AKT signal was greatly inhibited in the BLM group and activated with LEO treatment in the initial stage of pulmonary fibrosis on day 14, following by a high level of alveolar epithelial cells apoptosis in the end stage of pulmonary fibrosis on day 28.
Immunological factors may be correlated to the mechanism of IPF.6,19 Interestingly, the current study showed that whether on day 14 or day 28, the expression of key transformed T cell transcription factor RAG2 in the BLM group was always obviously higher than that in the control group and obviously, increased T cells were detected in pulmonary fibrosis. The features of autoimmunity have been described for lung fibrosis and are presumed to contribute to fibrosis exacerbation since auto antibody-reductive therapies have been shown to improve the lung function in patients with lung fibrosis during acute exacerbation. 6 Significant induction and activation of Th2 cells and type 2 cytokines are observed in lung fibrosis.7,20 AKT was considered to be an important negative regulator in early T cell differentiation. 14 It was reported that the recombination of variable (V), diversity (D) and joining (J) gene segments of immunoglobulin and T-cell receptor (TCR) required RAG1 and RAG2, 21 and AKT could regulate RAG2 expression and T cell differentiation 14 , and anti-CD3 treatment could activate AKT signal.13,22,23 Similarly, the AKT signal plays a major role in host defense against infection by promoting T cell immunity. 24 Our study also proves that AKT regulated changes in RAG2 expression and constituted a signaling pathway for T cell differentiation processes in lung fibrosis.
Leonurine, one of the active compounds from purified
Both low-dose and high-dose LEO treatment groups showed a synchronous increase for lung fibrosis. It was reported that treatment of LEO at 60 mg/kg per day could significantly reduce the infarct volume and alleviate the neurological impairment, indicating the lesser histological damage. 28 In our study, we explore the best therapeutic effect of drug concentration and also found the high dose of LEO did not have a better efficacy in our study. Instead, the low dose of LEO had a greater protective efficacy with respect to pulmonary fibrosis, T cell inactivation, and apoptosis in alveolar cells. In a word, the best dose effect of LEO was attained in 50 mg/kg/d pulmonary fibrosis mice. These results indicated that the immunoregulatory effect of LEO might be involved in the successful treatment of BLM-induced lung fibrosis at a low concentration of the drug.
However, the immunological mechanism underlying pulmonary fibrosis is yet unclear. TGF-β1 is a well-characterized pro-fibrotic growth factor during lung fibrosis. The immune mechanism of lung fibrosis with LEO treatment associated with low TGF-β1 levels remains unknown, especially with respect to the self-promotion and self-suppression effects of TGF-β1. The driving force underlying TGF-β1 production is a multifactor pathophysiological process, which will be investigated via in vitro experiments in the future. On the other hand, there may be other cytokines in the impact process of AKT-mediated RAG2 repression. Previous studies showed the AKT-mediated RAG2 repression is effectuated by direct inhibition of FOXO1 and by feeding into NF-κB–mediated FOXO1 regulation because simultaneous inhibition of AKT and NF-κB pathways increased the level of nuclear FOXO1 with a concomitant decrease in FOXO1 phosphorylation. 8 In addition, it was reported that the loss of FOXO1 could bind to the enhancer region of the RAG1/2 locus and provoke FOXO1 cleavage, downmodulating RAG1/2 expression and activity. 30 The correlation between FOXO1 and AKT-mediated RAG2 repression needs to be elucidated further in our study. An additional limitation of the present study was the lack of the calculation and justification of the sample size selected as only animal samples, not human specimens, were involved in our study.
Conclusions
The present study indicated that the AKT signal promoted the anti-apoptosis effect by suppressing the expression and activity of RAG2 and transforming T cells in lung fibrosis. LEO has a protective effect on lung fibrosis via attenuation of apoptosis and inflammation. These phenomena are exerted by upregulating the AKT signal in transformed T cells via suppressing the expression and activity of RAG2. In a word, the administration of LEO is a potential clinical therapy for pulmonary fibrosis.
Footnotes
Authors’ contributions
X.-T.L.,G.-P.C, F.-D.D, and B.-X.L. planned the project. H.-Y.Z. and F.G. conceived of and designed the study. X.-T.L., X.-H.Z., and F.-D.D. performed the sample collection. F.-F.M. and F.-D.D. performed immunohistochemistry. F.G., Y.-P.Z, and F.-D.D performed the expression analysis. F.G. and G.-P.C analyzed the data and drafted the manuscript. All authors reviewed the manuscript and approved the final version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (no.81800070 to Xiaoting Lv), Development Fund Program of Fujian Provincial Health and Family Planning Commission (no.2017-1-43 to Yongping Zhu), and Medical innovation in Fujian Province (no. 2019-CX-27 to Feng Gao).
Ethics approval and consent to participate
The authors sincerely thank the participants for their help and willingness to take part in this study. All experimental procedures in animal work were approved by the Ethical Committee of Fujian Medical University. This study was carried out in accordance with the recommendations of the Medical Ethics Committee of the First Affiliated Hospital of Fujian Medical University. The protocol was approved by the Medical Ethics Committee of the First Affiliated Hospital of Fujian Medical University [APPROVAL no. (2018)036]. The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Availability of data and materials
All data and materials generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
