Abstract
Introduction
Traditionally, materials used for internal fixation in orthopedics are metals such as titanium alloy and stainless steel. The elastic modulus of these metals is greater than that of human bone, so they can cause stress shielding effect which can decrease bone strength and delay bone healing. These implants can also undergo corrosion or abrasion and will release toxic ions or particles into the patient’s body, which may result in chronic inflammation and bone dissolution. 1 In addition, the metal materials used for bone fracture fixation are permanent and often require removal by an additional surgical procedure, which results in unnecessary morbidity;1-5 therefore, the development of resorbable metal materials to solve these problems has become a research focus.6-8
Magnesium and its alloys, which are easily corroded in solutions, especially in the presence of chloride ions, have become promising degradable biomaterials. Edward made pure magnesium wire into ligature to ligate blood vessels.
9
Payr proposed to use pure magnesium as orthopedic material in 1900.
2
Lambotte
10
treated lower limb fractures with a pure magnesium plate in 1907. Early research found that magnesium was safe and could promote bone tissue healing, but it was degraded too quickly in the body, fractures did not have time to heal, and internal fixation subsequently failed. As a result, magnesium and its alloys were replaced by stainless steel materials with better mechanical properties. With breakthroughs in smelting technology and casting processes, the purification and alloying of magnesium are greatly improving. This has significantly improved the corrosion resistance and mechanical properties of magnesium and its alloys, so magnesium and its alloys have again attracted attention as biodegradable medical materials in recent years.11-15 Previous studies have confirmed that magnesium alloys have good biocompatibility, may promote osteocyte growth, and induce production of osteoblasts and osteocytes.16-20 However, the osteogenesis and degradation behavior of magnesium alloy plate
Materials and methods
Materials
Magnesium alloy and titanium alloy were fabricated into plates and screws for internal fixation by Shanghai Puwei Medical Instrument Co., Ltd. (Figure 1). Photographs of (a) the magnesium alloy plate and (b) titanium alloy plate.
Chemical composition of the magnesium alloy plate.

The plates were 32 mm long, 5 mm wide, and 1.5 mm thick. There were four holes in the plates, the diameter of holes was 2 mm and the distance between the holes was 5 mm. The screws were 7 mm long and 2 mm in diameter. Plates and screws were sterilized and sealed after sterilization with ethylene oxide.
A total of 36 clean healthy adult New Zealand white rabbits weighing 2.0 kg–3.0 kg, an averagely 2.5 ± 0.3 kg, were supplied by the Shanghai Jiesijie Lab. Animal Co., Ltd., China (license No.: SCXK (hu) 2010-0026). 18 were male and 18 were female. Protocols were performed in accordance with Guidance Suggestions for the Care and Use of Laboratory Animals, issued by the Ministry of Science and Technology of the People’s Republic of China (2006-09-30). 21
Animal experiment
The 36 healthy adult New Zealand rabbits were randomly divided into two groups: the magnesium alloy plate was the experimental group and the titanium alloy bone plate was the control group, each with 18 rabbits. The New Zealand rabbits were raised in separate cages, and running water plus standard feed.
The rabbits were intramuscularly anesthetized with xylazine (4 mg/kg, Jilin Huamu Animal Health Products Co., Ltd., China) and ketamine (80 mg/kg, Fujian Gutian Medicine Co., Ltd., China). Prior to surgery, iodophors were utilized to sterilize the skin three times. Hole sheets were paved. A 4 cm longitudinal incision was made in the medial side of proximal right tibia. Under sterile conditions, the subcutaneous tissue was incised and the muscles were separated to expose the proximal tibia. Then, a fracture of proximal tibia was created using an electric saw, and the holes were created using an electric drill at both ends of the fracture. Finally, the respective implant, either magnesium alloy plate or titanium alloy plate, was fixed onto the proximal tibia to stabilize the fracture in each group (Figure 2). After saline irrigation, the incision was sutured layer by layer. The rabbits were able to move freely in their cages following recovery, with free access to food and water. Postoperatively, the animals received intramuscular injection of penicillin (400,000 U/day) for 3 days. Rabbit diet, activity, inflammatory reaction, and wound condition were recorded and clinical examination of the tibia was performed daily postoperatively. Surgical procedure of the animal experiments. (a) Magnesium alloy plate implanted into one tibia of a rabbit; (b) Titanium alloy plate implanted into one tibia of a rabbit.
X-ray examination
The fracture site in each group was X-rayed under fixed conditions at (60 kV, 5 mA, 0.8 s) 1, 2, 4, and 16 weeks postoperatively to ensure the correct position of the plates, to check callus formation around the plate, and to monitor the degradation behavior of the magnesium alloy plates
Pathological examination
Six rabbits from each group were euthanized by ketamine overdose at 4, 8, and 16 weeks postoperatively. The bone from the fracture region was obtained and fixed with 4% formalin for 24 h, decalcified with 5% nitric acid solution, dehydrated with gradient ethanol, permeabilized, embedded in paraffin, and sliced into 5 μm thick sections. The sections were stained using hematoxylin and eosin (HE) staining and the formation of callus was observed under a microscope.
Immunohistochemical examination
Immunohistochemistry (IHC) staining was performed by streptavidin-perosidase (SP) method. The slice was baked at 60°C for 20 min, and dewaxed with xylene and dehydrated with gradient alcohol; then 3% H2O2 was added and incubated in a 37°C incubator for 10 min, and rinsed with phosphate buffered saline (PBS) 3 times for 5 min. Placed into 0.01M citrate buffer (PH6.0, 95°C) for 20–25 min, then cooled naturally for more than 20 min, and rinsed 3 times with PBS for 5 min and serum was added to block. The primary antibody was added and put it in a 4°C refrigerator overnight, then rinsed with PBS for 3–5 min. The secondary antibody was added and incubated at 37°C for 30 min, and rinsed with PBS for 3–5 min. Then, horseradish peroxidase was added and incubated at 37°C for 30 min, and rinsed with PBS for 3–5 min. Finally, stained with DAB/H2O2 and counterstained with hematoxylin, then dehydrated, transparented, dryed, and mounted. The bone morphogenetic protein 2 (BMP-2) was detected under a microscope. The results of IHC staining were divided into four grades according to the depth of staining, that is, no staining meaning negative, light yellow meaning weak positive, brown yellow meaning positive, and brown black meaning strong positive.
Degradation behavior of magnesium alloy plates
The degradation behavior of magnesium alloy plates was observed using scanning electron microscope (SEM) and energy dispersive spectroscopy (EDS) system (Hitachi S-4800, Japan). At 4, 8, and 16 weeks postoperatively, plates were removed from rabbit tibias, rinsed with distilled water, and dried in the air, then the surface morphology was observed by SEM. The products on the surface of the magnesium alloy plats were detected by EDS. Last, the plates were cleaned by 200 g/L chromic acid to remove the corrosion products. The surface morphology was observed by SEM and the degradation rates were obtained according to the weight loss method.
Statistical analysis
The statistic software SPSS13.0 (SPSS, Inc., Chicago, IL, USA) was used to analyze the data. The difference between the groups was compared using one-way analysis of variance (ANOVA).
Results
General condition of experimental animals
Of the 36 rabbits obtained, a total of 34 were included in the final analysis. One rabbit, which fractured its surgical region in the magnesium alloy group and a rabbit that died of diarrhea in the titanium alloy plate group after the operation were not included. The remaining rabbits in each group were active and grew well, with normal diet and drinking. No infection, swelling, necrosis, inflammation, or fever occurred in the surgical site.
X-ray examination
Figure 3 shows an X-ray of the surgery area in the tibia of rabbits at 4, 8, and 16 weeks after surgery in each group. In the magnesium alloy plate group, the plates and screws were well positioned during bone healing. The fracture healed gradually and there was significant callus around the plate. In the titanium alloy plate groups, the plates and screws were well positioned during bone healing. However, only a small amount of callus was formed around the plate. X-ray photographs of the fracture region in the magnesium alloy plate group and titanium alloy plate group at 4, 8, and 16 weeks post-operation. (a–c) The magnesium alloy plate implanted into the New Zealand white rabbit tibias at 4, 8, and 16 weeks after surgery. (d–f) The titanium alloy plate implanted into the New Zealand white rabbit tibias at 4, 8, and 16 weeks after surgery.
Pathological examination
The New Zealand white rabbits were sacrificed at 4, 8, and 16 weeks after the surgery and the fractured tibias of the rabbits were observed (Figure 4). Callus around the plates can be seen at 4 weeks post-implantation in the magnesium alloy plate group and titanium alloy plate group. However, the formation of callus was more abundant in the magnesium alloy plate group than that in the titanium alloy plate group at 4 weeks after operation. Photographs of the callus around the plates at 4 weeks post-implantation in (a) the magnesium alloy plate group and (b) titanium alloy plate group.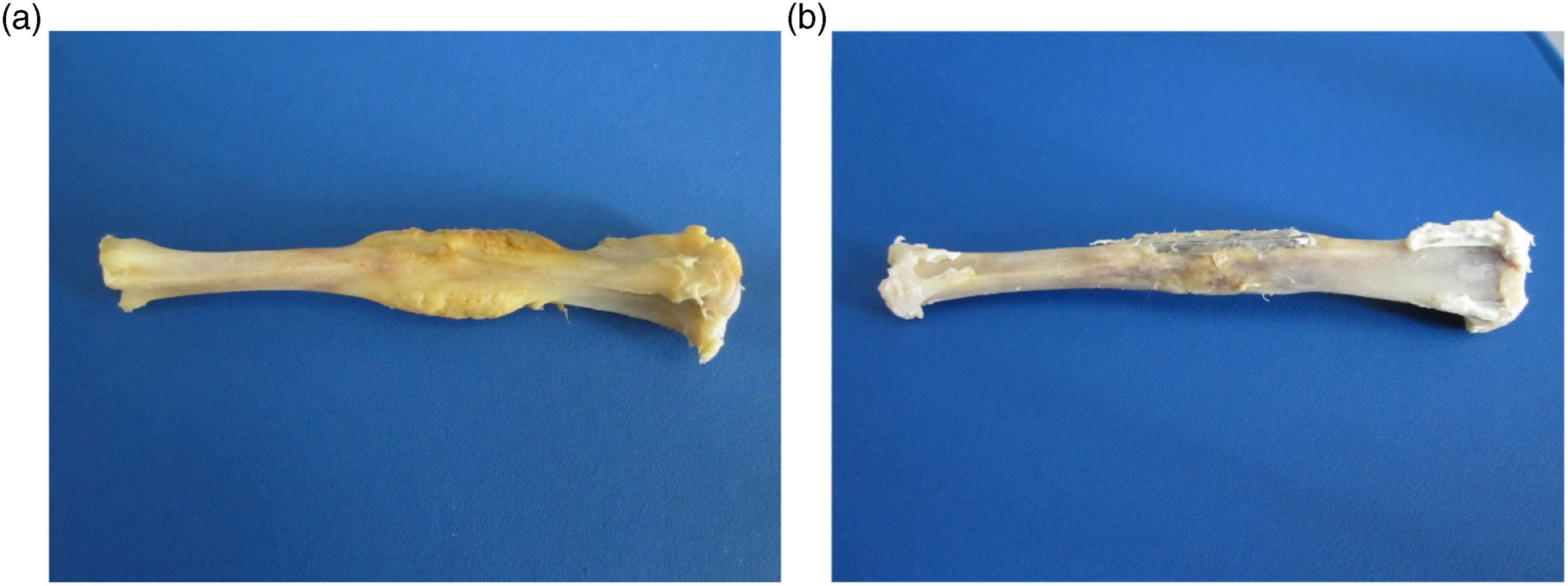
As shown in Figure 5, pathological sections were stained with hematoxylin and eosin. From pathological sections, callus around the plates can be seen at 4 weeks post-implantation in the magnesium alloy plate and titanium alloy plate groups. However, the formation of callus was more abundant in the magnesium alloy plate group than that in the titanium alloy plate group at 4 weeks after operation. Pathologic photographs of the callus around the plates at 4 weeks post-implantation in (a) the magnesium alloy plat group and (b) titanium alloy plate group (HE staining, ×100).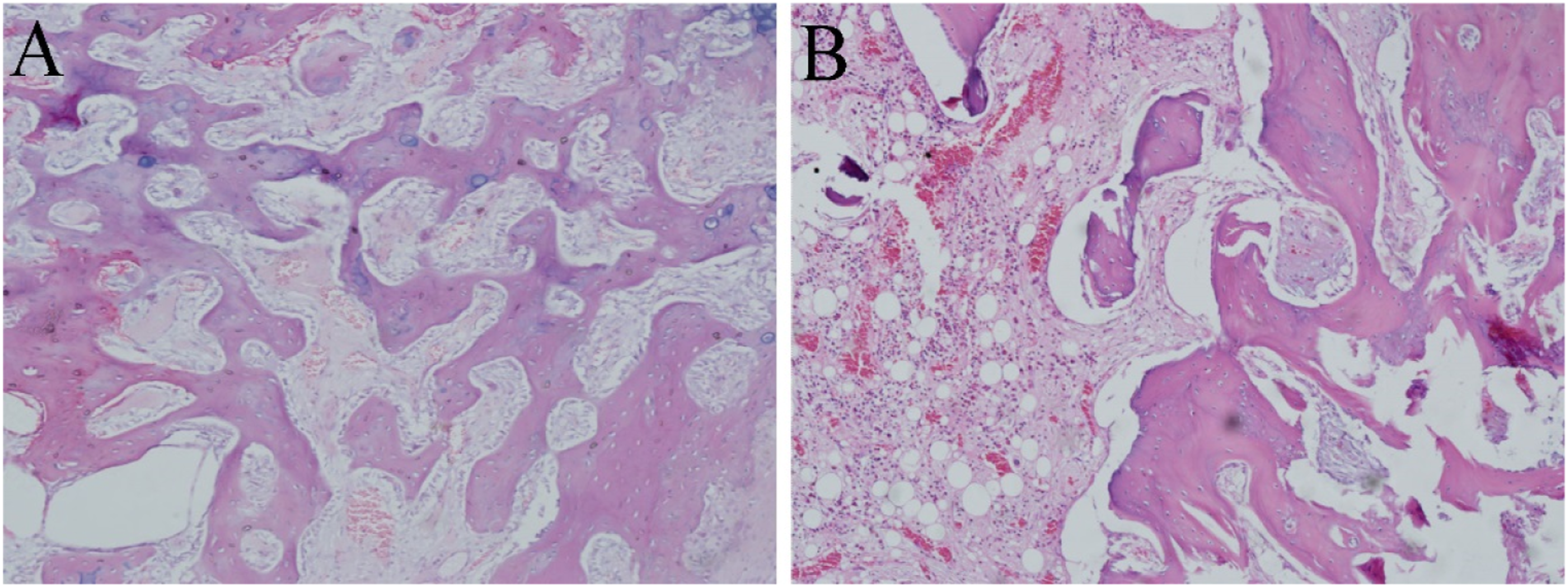
Immunohistochemical examination
Figure 6 shows the expression of BMP-2 stained with immunohistochemistry in the new bone tissue around the plates in each group. It can be seen the brown yellow staining, which indicates the positive expression of BMP-2 in the new bone tissue around the plates at 4 weeks after fracture in the magnesium alloy plate and titanium alloy plate groups. The expression of BMP-2 was more obvious in the magnesium alloy plate group than that in the titanium alloy plate group. The expressions of BMP-2 in the new bone tissue around the plates at 4 weeks post-implantation in (a) the magnesium alloy plate group and (b) titanium alloy plate group (×100).
Degradation behavior of magnesium alloy plates
The degradation behavior of magnesium alloy plate
According to the X-ray, the degradation behavior of the magnesium alloy plate deepened gradually
The magnesium alloy plates were removed from the rabbit tibias at 4, 8, and 16 weeks after the surgery. At visual inspection, the surface of the magnesium alloy plates had lost its original metallic luster and its edge was not distinct. After drying, the surface was coated with some loose gray-white materials. As shown in Figure 7(a), a layer of off-white degradation products under the SEM was visible on the surface of the magnesium alloy plates at 8 weeks after implantation in the magnesium alloy plate group. Energy dispersive spectroscopy analysis showed that the corrosion products formed on the surface of magnesium alloy plates were mainly composed of oxygen, carbon, nitrogen, magnesium, and phosphorus at 8 weeks after the surgery (Figure 7(b)). As shown in Figure 8, the surface was rough with irregular corrosion pits on the magnesium alloy plates when the plates were cleaned by chromic acid to remove the degradation products on the surface of the magnesium alloy plates. The size of the corrosion pits was small and the distribution was more uniform. (a)The surface of the magnesium alloy plate was rough with a layer of off-white degradation products at 8 weeks post-implantation; (b) Energy dispersive spectroscopy of the corrosion products formed on the surface of magnesium alloy plate. Corrosion morphologies of magnesium alloy plate after removing degradation products using chromic acid.

Corrosion rates of magnesium alloy plates at 4, 8, and 16 weeks after implantation are shown in Figure 9. The weight loss of magnesium alloy plates were 18.34%, 24.53% and 27.79% at 4, 8, and 16 weeks after implantation, respectively, suggesting resorption had occurred. The results showed that the degradation behavior of the magnesium alloy plates deepened gradually Degradation rate of magnesium alloy plate after 4, 8, and 16 weeks post-implantation.
Discussion
Metabolism of bone and repairing of bone injury are a dynamic equilibrium process between osteoblasts, and osteoclasts, activity. There are three key points in this process-mesenchymal stem cells differentiating into osteoblast precursor cells: synthesis of bone matrix and mineralization following differentiation and mature of osteoblasts, some osteoblasts returning to the osteoblast precursor cell bank or buried in bone as osteocytes and dying gradually, and the process involving the development of osteoblasts is regulated by many systemic and local regulators. The cytokine BMP-2 is the only one that can result in ectopic osteogenesis and induce bone formation. 22 Bone morphogenetic proteins, produced by osteoblasts, are a group of highly conserved functional proteins with similar structures and that are mainly bound to the bone matrix and rarely released outside the bone. There are currently more than 20 types of BMPs and all of them are members of the Transforming Growth Factor Beta ( TGF-β) family except BMP-1. They have a lot of biological functions, including mesenchymal cells to differentiate into cartilage and bone and promoting osteoblast maturation.23-25 Bone morphogenetic protein-2, with the strongest osteogenic activity, mainly recruits and differentiates mesenchymal cells. It has specific effect on differentiation of osteoblast and formation of bone. In the early stage of bone formation, BMP-2 not only recruits bone mesenchymal cells to the center of bone formation and differentiates bone mesenchymal cells into osteolineage cells but also reverses fibroblasts, myoblasts, and bone marrow stromal cells into osteolineage cells.26-29
Our previous studies have confirmed that magnesium alloys have good biocompatibility, may promote osteocyte growth, and may induce production of osteoblasts.16-18 Recently, there were few reports about the application of magnesium alloy plates
The results of local observation of callus in this study showed that callus around the plates can be seen at 4 weeks post-implantation in the magnesium alloy plate and titanium alloy plate groups. However, the formation of callus was more abundant in the magnesium alloy plate group than that in the titanium alloy plate group at 4 weeks after operation, and this was confirmed by the results of histopathological sections in this study. Immunohistochemical test results showed that the osteoblastic cytoplasm caused brown yellow staining in the new bone tissue around the implant. However, the osteoblast staining in the magnesium alloy plate group was more intense, proving that the magnesium alloy plate can promote the formation of local callus and osteogenesis
As well as exploring the repairing of fractures with magnesium alloy plates, its degradation behavior
Conclusions
In this study, the effects of magnesium alloy plates implanted into rabbit tibia on fracture healing and
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Scientific research project of colleges and universities in Gansu (2018B-013); Science and technology project of Chengguan District of Lanzhou (2018-1-2); Scientific research projects of central universities (31920200021); National Natural Science Foundation of China (81960398).
