Abstract
There have been two measurement methods for nasal nitric oxide (nNO): direct measurement of nNO and indirect measurement of NO metabolites or the NO synthase enzyme. The aim of this study is to investigate the difference in nNO between allergic rhinitis (AR) patients and the correlation of the direct measurement of nNO and indirect measurement of nNO metabolites, RANTES, and IL-5. Fifteen patients with AR, 15 patients with septal deviation as a control group were enrolled. We measured the nNO level using a chemiluminescence analyzer and the amount of NO metabolites, RANTES, and IL-5 in nasal lavage fluid using the ELISA method. Nasal NO levels were significantly higher in AR than the control (195.7 ± 39.6 vs 159.4 ± 25.8 ppb,
Keywords
Introduction
Nitric oxide (NO) is a highly reactive inorganic free radical present in high concentrations in normal nasal cavity and paranasal sinuses. 1 The physiological role of nasal NO (nNO) is to contribute to local host defense and modulate ciliary motility. It also serves as an aerokine in helping to regulate pulmonary function. 2
Nitric oxide is synthesized from L-arginine by the NO synthase (NOS) enzyme, which has three different isoforms: endothelial NOS (eNOS), neuronal NOS (nNOS), and inducible NOS (iNOS). 3 Endothelial NOS and nNOS are always expressed in constitutive form, but iNOS can increase by inflammation. The ciliary epithelial cells in paranasal sinuses mainly produce nNO continuously. Additionally, NO is synthesized by the iNOS of the nasal mucosa by inflammatory cells such as eosinophils and macrophages. 3 Inflammatory cells including eosinophils increase in the nasal cavity of allergic rhinitis (AR) patients.
The study of NO has been done in two ways. In the past, indirect methods such as the study of NOS enzymes and the metabolites of NO (nitrite and nitrate, nNOx-) have been used. Since then, many studies have been conducted on the direct measurement of NO in a gaseous state rather than an indirect method. Indirect methods have been used in research fields, and the direct measurement of NO has begun to be used in clinical fields. Clinically exhaled NO (eNO) measured orally is used in lower airway inflammatory diseases like asthma, and nNO measured nasally is used in the diagnosis of ciliary dyskinesia. More recently, the association between other sinonasal diseases such as AR, chronic rhinosinusitis (CRS), and nNO levels has been evaluated, but it is not yet used in clinical practice widely.4,5
The aim of this study is to investigate the difference in nNO between AR and control patients and correlation of the direct measurement of gaseous nNO and indirect measurement of NO metabolites, representative allergic cytokine (IL-5), and representative chemokine (RANTES) in nasal secretion of each group.
Patients and methods
This study was approved by the Institutional Review Board of Hanyang University Guri Hospital (IRB number: 2018-06-025) and informed consent was obtained from all participants before the study.
Subjects
To find out the level of nNO and its metabolites, IL-5, RANTES in AR patients, we enrolled 15 patients with AR as AR group and 15 patients without AR as a control group among 30 patients who was undergone septoplasty due to nasal septal deviation. The sample size of each group (n = 15) was calculated based on outcome variable of nNO that was estimated by means of the standard deviation of continuous endpoint and two independent groups(alpha = 0.05, power = 80%).
All participants from each group were included according to history-taking, a multiple antigen simultaneous test (MAST) or allergy skin test, nasal endoscopy, and a paranasal sinus CT scan.
Patient exclusion criteria were as follows: (1) younger than 15year’s old; (2) history of asthma; (3) patients who had undergone previous nasal operations; (4) patients who had taken medication for nasal symptoms within the previous 3 weeks; (5) patients who had rhinosinusitis on nasal endoscopy or paranasal sinus CT scan.
Clinical parameters
All participants underwent MAST or allergy skin test, measurements of total Ig E level and eosinophil count with peripheral blood tests including the erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP), and olfactory function tests with the Korean Version of the Sniffin’Sticks threshold tests II (KVSS II). We scored olfactory function as follows: normosmia = 1, hyposmia = 2, anosmia = 3.
Measurement of exhaled and nasal nitric oxide
We measured eNO and nNO level using a chemiluminescence analysis method using the Sievers NO analyzer, NOA 280i (GE Analytical Instruments, Boulder, CO, USA) according to the American Thoracic Society/European Respiratory Society recommendations. 6 To measure the level of nNO, velopharyngeal closure was achieved through inhalation to total lung capacity, and oral exhalation maintaining an expiratory pressure of >10 cm H2O separated the nasal cavity from the lower airway. Then, air was aspirated at 700 mL/min by a suction pump, and nNO samples were taken at 200 mL/min midstream of the aspirated airflow. Because there is a difference in the size of the nasal cavity on the left and right in nasal septal deviation patients, we measured nNO on both the left and right sides, and there was no difference between the left and right sides. All the measured values were determined by averaging two samples that had a <10% difference. The level of eNO was measured by velopharyngeal closure, and the plateau was chosen when the expiratory flow rate reached 50 mL/s.
Method of nasal lavage
Nasal lavage fluids were obtained by using 0.9% NaCl, prewarmed to 37°C and modified Naclerio’s classic nasal lavage technique.7,8 Each patient remained in a supine position during general anesthesia. A 16 F Foley catheter was inserted through the nasal cavity and ballooning was done in order not to lose the instilled fluid though the nasopharynx. Posterior loss was limited by holding the Foley catheter tightly to the nostril direction during the period of nasal lavage retention. The volume instilled within each nostril was 3.0 ml, and the fluid was left within the nasal cavity for 10 s. Then, the fluid was suctioned out using a Juhn tym-tap collector (Medtronic, Product No.419010, USA) to collect the nasal lavage fluid. The fluid collected from the nasal cavity was transferred into three Eppendorf tubes resistant to -70°C and sent to a research laboratory.
Measurement of NO metabolites, RANTES and IL-5
The amount of NO metabolites, RANTES, and IL-5 in the nasal lavage fluid was measured by the ELISA method using a Human Nitrite/Nitrate colorimetric assay kit (Sigma-Aldrich, Product No. 23479; Merck, Darmstadt, Germany), a human RANTES kit (LSBio, Product No. LS-F2537, Seattle, USA), a Human IL-5 kit (Youngin Frontier, Product No. LF-EK0259; Seoul, Korea). The measurements were done according to the manual of each kit. All the nasal lavage samples were tested diluted to 1:4 in 1X Phosphate-buffered saline. After loading the plate with samples (100 µL) and regents of each kit was added to the wells. The plates were incubated at 37°C for 30 min and absorbance was measured at 540 nm using a standard plate reader. Fresh standard solutions were included in each experiment. We duplicated each test.
Statistical analysis
All the data were expressed as mean ± standard deviation. The Mann-Whitney
Results
Clinical parameters of subjects
We recruited 30 subjects, which comprised the control (n = 15), AR (n = 15). There were no statistically significant differences in gender, age, or Body mass index (BMI) between the control, and AR (Tables 1). The AR group showed significantly increased serum IgE, a higher positive rate of Dermatophagoides pteronyssinus (DP), and Dermatophagoides farina (DF) on the MAST or allergy skin test compared to the control (Table 1).
Comparison between control group and allergic rhinitis group.

BMI: body mass index; DP: Dermatophagoides pteronyssinus; DF: Dermatophagoides farinae; ESR: erythrocyte sedimentation rate; CRP: C-reactive protein; LM score: Lund-Mackay score; eNO: exhaled nitric oxide; nNO: nasal nitric oxide; KVSS II score: Korean version of Sniffin’ Sticks Test II score (Mann-Whitney
Exhaled and nasal NO
There was no significant difference between the groups in eNO (Control 16.9 ± 15.1 ppb, AR 17.2 ± 16.2 ppb,
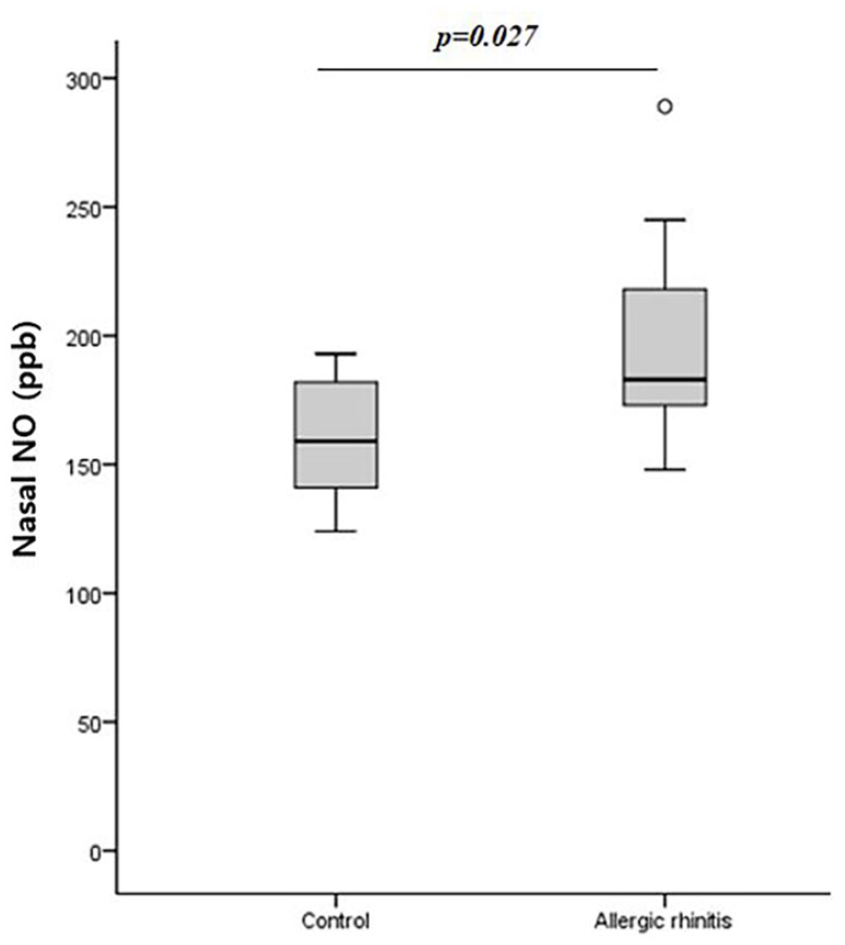
Nasal NO levels Nasal NO levels was significantly higher in allergic rhinitis group than control group.
NO metabolite, IL-5, RANTES in nasal lavage fluid
NO metabolite levels were higher in AR (1.47 ± 1.02 μg/mL) than the control (1.17 ± 0.64 μg/mL), but there was no significant difference between the groups (Figure 2). The RANTES levels (117.4 ± 159.2 vs 42.1 ± 28.3 pg/mL) and IL-5 levels (81.1 ± 273.8 vs 1.6 ± 3.8 pg/mL) were higher in AR, but there was no significant difference between groups (Table 1).

NO metabolites in nasal lavage fluid Nasal NO metabolites (Nitrite and nitrate) increased in the nasal secretion of patients with allergic but showed no statistically significant difference.
Correlation between nasal NO and clinical parameters
There was no significant correlation between nNO and NO metabolites, RANTES, IL-5, and clinical parameters except for the olfactory test score in AR. There was a negative correlation between nNO and the olfactory test score in AR (

Correlation between nasal NO and olfactory test score in allergic rhinitis There was a negative correlation between nasal NO and the olfactory test score in allergic rhinitis group.
Discussion
Nitric oxide is present in high concentrations in the upper respiratory tract. In the paranasal sinuses, NO is produced continuously without inflammatory stimuli by constitutive NOS, and its concentration is maintained at 20–25 ppm. The NO concentration measured in the upper airways including the nasal cavity is 400–900 and 20 ppb or less in the lower airway.1,2
The amount of nNO measured through the nasal cavity is determined by the patency of the ostiomeatal unit (OMU) of the sinus, which is related to the level of NO passing from the sinus and the amount of additional NO production of the nasal mucosa by the inflammatory stimuli. 9 Nasal NO can be decreased due to obstruction of the OMU induced by mucosal swelling of AR and chronic sinusitis (CRS) with nasal polyp, which inhibits release of nNO produced in the paranasal sinuses. On the other hand, nNO production can increase due to induction of iNOS in response to inflammatory stimuli like URI, AR in upper respiratory epithelial cells.2,4 Therefore, the concentration of nNO is high in AR, low in CRS and very low in primary ciliary dyskinesia and cystic fibrosis. 10 In our study, nNO was statistically significantly higher in AR than the control group, which is thought to be useful for the diagnostic or follow up tools of AR.
Nasal NO level has been measured by indirect or direct methods. NO is produced from arginine by NOS and converted to metabolites of NO (nitrite and nitrate, nNOx-). There have been two major indirect measurement methods of nNO levels: PCR or immunostaining in nasal mucosa and metabolites of NO in nasal secretion.11–14 Takeno et al. 12 showed that NO production increased in nasal epithelial cells from AR using RT-PCR and a fluorescence indicator. Kawamoto et al. 13 localized NO synthase in nasal mucosa with AR. Another method for indirect measurement is the study for metabolites of NO in nasal secretion. Nitric oxide is a free radical and quite reactive, so it may freely diffuse in a liquid environment and rapidly transforms into nitrite and nitrate, so nitrite and nitrate are good parameters of endogenous NO production.15,16 Imoto et al. 15 studied nitrite and nitrate in nasal lavage fluid in AR. They showed that NO metabolites reflected nasal symptoms after a single nasal allergen provocation.
Direct measurement methods of gaseous NO have been developed recently and have been tried in research and clinical fields.4,5,17–19 Chemiluminescence analysis and electrochemical analysis techniques are direct methods measuring nNO, but they require a special gas analyzer and equipment. The chemiluminescence analyzer used in our study is composed of a relatively large equipment and is advantageous for nNO measurement. The electrochemical analyzer is a small hand-held equipment, so it is used in clinical fields, but only in eNO measurement for asthma follow up. Unlike eNO, which is simple to measure and relatively reproducible, the level of nNO is highly influenced by outside air NO level and very sensitive to the aspiration flow-rate of air. Therefore, it is difficult to maintain reproducibility, so it is still only used for research purposes. 20
The purpose of our study was to determine whether metabolites of NO in nasal secretion, which has been used in the past, or direct measurement of gaseous NO are useful in AR. In our study, the direct method for nNO showed a significant difference in AR. However, the NO metabolites of nasal secretion did not show a significant difference and did not correlate with nNO. Therefore, direct measurement of nNO was more useful in AR than indirect measurement of metabolites of NO in nasal secretion.
Allergic rhinitis is inflammatory diseases of the nasal cavity and paranasal sinus. In addition to NO originating from sinuses naturally, NO can be elevated by increased iNOS of the nasal cavity and sinus due to inflammation of AR. Allergic rhinitis is an IgE-dependent inflammatory disease and is associated with eosinophil, which is a representative inflammatory cell. 21 In our study, for this reason, nNO increased in AR.
The representative cytokine of the Th2 cell, IL-5, and the representative chemokine of eosinophil, RANTES, were compared and examined for a direct association with nNO. Although IL-5 and RANTES could be increased in AR, it was not directly related to nNO, so measurement of cytokine and chemokine in nasal secretion is helpful for the evaluation of AR, but it would be difficult to use as an indirect method of evaluation of nNO. 22 Arnal et al. 18 showed that nNO production increased regardless of the presence or absence of symptoms in AR due to the persistent inflammatory response of the nasal mucosa. However, it has been reported that nNO levels can be reduced when symptoms are severe and long-lasting. If the mucosa of OMU is swollen, nNO from sinuses decreases, so nNO can be lowered in severe AR patients.4,23
In our study, there was a statistically significant negative correlation between nNO and olfactory function in AR. Increased levels of nNO mean that the allergic inflammatory reaction is severe, which means that the swelling of the mucosa in the nasal cavity and sinuses is severe. As the olfactory pathway is narrowed or obstructed, the olfactory function can be decreased. It can be estimated that the severity of AR is related to the nNO level. In several studies of the relationship between olfactory function and nNO in CRS, both nNO and olfactory function increased after CRS surgery. They concluded that the olfactory function was not directly related to the level of nNO too.24,25
Our study has some limitations. First, there was no statistically significant difference between NO metabolites, IL-5 and RANTES. This finding is necessary to confirm in larger sample sizes. Although NO metabolites were statistically insignificantly higher in AR than in controls and this might probably depend on the sample size. Second, the nasal lavage method we used to collect nasal secretion made it difficult to evaluate the very small amount of metabolites, cytokine, and chemokine. Therefore, a more precise collection method and development of an analysis method for nasal secretion is needed. Third, we were only able to compare one cytokine and one chemokine. However, it is necessary to compare more diverse cytokines and chemokines of Th1, Th2, and other T cells in future work.
The skin prick test is an invasive, time consuming, one-time diagnostic test to find out the specific allergen for allergic rhinitis. On the other hand, nasal NO measurement is a non-invasive, rapid test and it can be used as a monitoring for the severity and treatment response of allergic rhinitis by repeated tests. Recently, hand-held nasal NO measurement devices have been introduced and started to be used in clinical practice.
Conclusion
Nasal NO concentration increased in AR. However, nNO metabolites and IL-5, RANTES in nasal secretion did not show any correlation with nNO. Direct measurement of nNO could be a potentially useful biomarker of AR compared to indirect measurement of NO metabolites, cytokines, and chemokines in nasal secretion.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from Institutional Review Board of Hanyang University Guri Hospital (IRB number: 2018-06-025)
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from all subjects before the study.
