Abstract
It has been reported that the normal adults can suffer from an intracranial aneurysm (IA) that might present the risk of rupture and cause the subarachnoid hemorrhage. Peroxisome proliferator-activated receptor-γ (PPAR-γ) as a nuclear hormone receptor has been identified to involve in the progress of the formation and rupture of IAs. Quantitative reverse transcription polymerase chain reaction (qRT-PCR) was used to detect PPAR-γmRNA expression in the macrophages of the patients with IAs. The information including fasting blood glucose (FBG), interleukin-6 (IL-6), and systolic blood pressure (SBP) were collected. The aneurysm parameters of all the participants were obtained through the cerebral angiography. Establishing the receiver-operating characteristic curve (ROC curve) evaluated the clinical significances of PPAR-γmRNA for IAs rupture. In this study, we observed that the rupture of IAs was caused by the maximum height of aneurysm ⩾7 mm, the location of aneurysm in posterior communicating artery (PCOM) or anterior communicating artery (ACOM), and the increase of aneurysm size ratio (SR). The levels of SBP and IL-6 in the rupture group were higher than those in the unrupture group, and PPAR-γmRNA expression in the rupture group was also significantly reduced. In addition, heavy drinking was statistically significant between the ruptured and unruptured groups. There was no significant difference in serum FBG level between the two groups. The evidences of this study showed that PPAR-γmRNA was negatively correlated with SBP, SR, and IL-6 levels in rupture group, respectively. The AUC of PPAR-γmRNA in ROC curve was 0.867, indicating that the change of PPAR-γmRNA level had obvious effect on IAs rupture. The aim of this study was to evaluate the potential of PPAR-γ in macrophages to prevent IAs rupture.
Keywords
Introduction
Previous studies have reported that normal adults may suffer from intracranial aneurysms (IAs), its prevalence is not found to be higher and presents 2%–6%. 1 However, IAs have the risk of rupture. 2 Saccular IAs rupture is the most common cause of subarachnoid hemorrhage (SAH), and is the most common stroke type. It has a high incidence rate and a high mortality rate. 3 A report on 11 European and Chinese populations showed that its 28-days mortality rate was 42%. 4
Current studies have found that inflammation, 5 lifestyle such as heavy drinking, 6 blood pressure, 7 aneurysm location, 8 and aneurysm size 9 are associated with IA rupture. Although the mechanism of formation and rupture of IA has not been fully elucidated, the inflammation, and matrix remodeling are also the main culprit. 10 In a recent study by Chalouhi et al., 5 the inflammation mediated vascular remodeling and responded to aneurysm formation. The process of macrophages entering the IA wall induced inflammatory reaction and promoted the formation and development of aneurysms. Therefore, noninvasive assessment of the inflammatory state of aneurysm wall can guide the treatment of unruptured aneurysms by identifying reliable markers that increase the risk of aneurysm rupture. 11 There was an evidence that peroxisome growth activated receptor-γ (PPAR-γ) participated in a variety of pathophysiological processes through inflammatory response. 8 PPAR-γ has four subtypes: γ1, γ2, γ3, and γ4, which is involved in the control of adipocyte differentiation, lipid storage (adipogenesis) and macrophage differentiation. PPAR-γ1 is expressed throughout the body, including endothelial cells, vascular smooth muscle cells, cardiomyocytes, and macrophages, as well as foam cells in atherosclerotic lesions. 12 A new insight into the role of PPAR-γ in human macrophage activation has been provided. PPAR-γ appears an influence on the expansion of a discrete subset of M2c-like anti-inflammatory macrophages. 13 Therefore, the purpose of this study was to present our institution data and to perform systematic review of studies suggesting that the differentially expressed gene such as PPAR-γmRNA in macrophages of peripheral blood of patients with IAs involved in the mechanism of occurrence, development and rupture of IA.
Materials and methods
Subjects
The clinical data of 107 cases of saccular IAs in Xianyang first people’s Hospital and Xianyang Hospital of Yan’an University from March 2015 to March 2019 were retrospectively analyzed. Ethical approval for human subjects was obtained from the research ethics committee of Xianyang First People’s Hospital and Xianyang Hospital of Yan’an University, and the requirement for informed consent was waived by the ethics commissions. There were 50 cases of ruptured IAs and 57 cases of unruptured IAs, with an average age of 51.23 ± 9.71 years. The diagnosis of saccular IAs was based on cerebral angiography (bilateral vertebral artery, bilateral internal carotid artery, bilateral external carotid artery). The subarachnoid hemorrhages caused by ruptured IAs were confirmed via CT scan. The clinical and imaging data were completed. Subarachnoid hemorrhage and aneurysm were confirmed by DSA. Inclusion criteria: Both unruptured and ruptured aneurysms were diagnosed by CT; the clinical data and imaging data of the patients were complete. The excluded cases contained the patients with the severe heart, liver, and kidney dysfunction as well as the cerebral infarction secondary to vasospasm. Of these patients, 50 cases were treated by interventional embolization and 57 cases were treated by surgical clipping.
Definition of parameters for IAs
The parent artery diameter was obtained by averaging two representative vessel cross sections of the aneurysm. 14 The maximum diameter of the neck plane was calculated as the neck diameter. 15 The maximum aneurysm height (aneurysm size) was measured as the maximum a clinic distance between the dome and the neck plane. The SR was defined as the ratio of the maximum aneurysm height to average of the parent diameter (Figure 1). 14
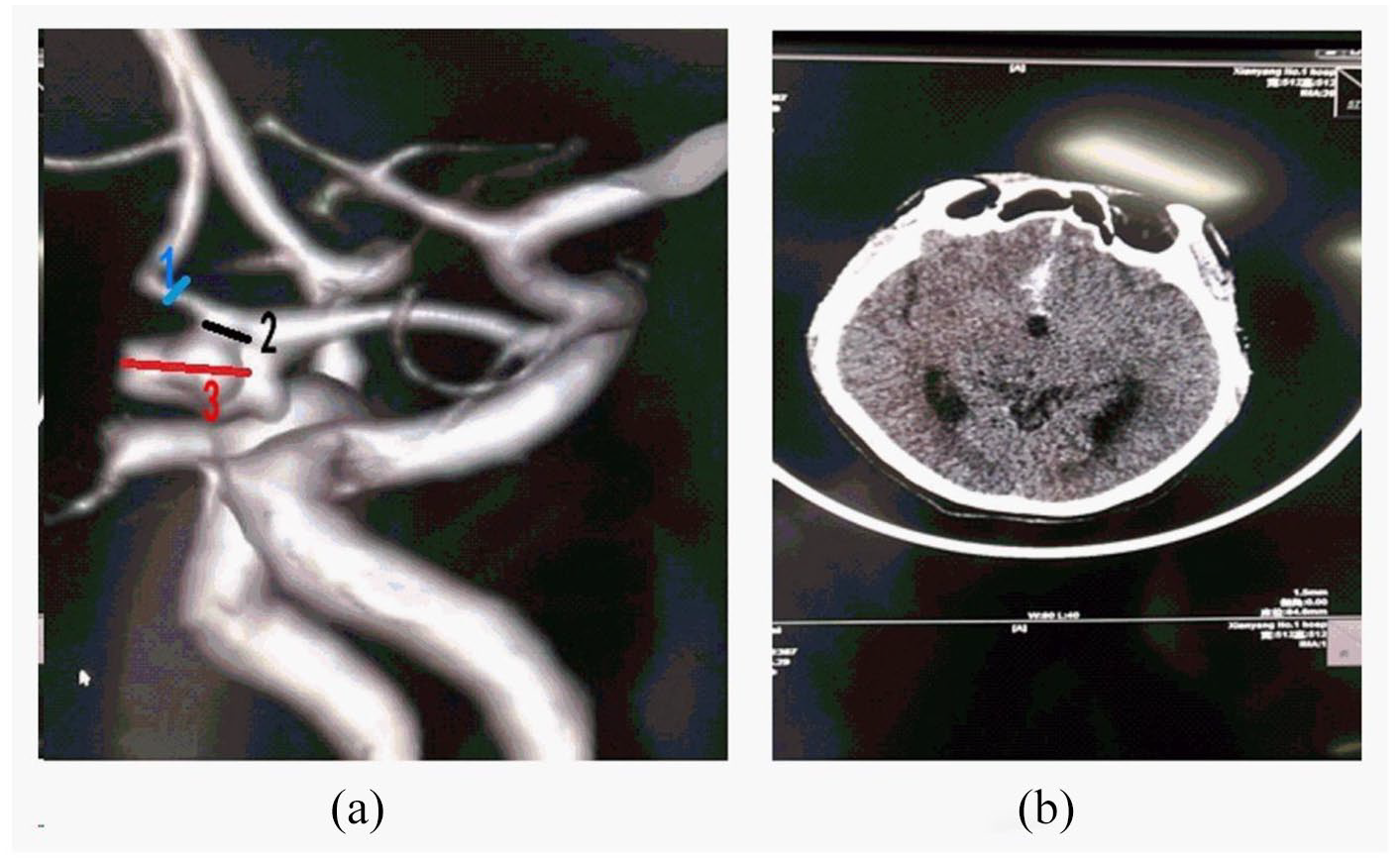
Definition of IA from the results of cerebral angiography: (a) showed IA located in ACOM with a height of 0.75 cm, in which blue 1 expressed the parent artery diameter, black 2 expressed the aneurysm neck diameter, and red 3 expressed the aneurysm size and (b) showed the ruptured IA from the results of CT scan.
We determined the size of the aneurysm via analyzing the mean measurements obtained by two readers. The aneurysmal measurements were operated by two neuroradiologists (with an average of 10 years of practical academic experience) and a clinical technologist (Dr. Kang). Repeated measurements were done by Dr. Kou and a technician.
Sampling
The participants’ blood was collected before treatment. After fasting for 8 h, 15 ml of the participants’ venous blood were collected. Five milliliter venous blood was separated at the speed of 3000 R/min for 15 min to determine the levels of IL-6 and FBG. While 10 ml of blood were supplied for PPAR-γmRNA analysis with EDTA anticoagulant. The separated products were removed to the freezing pipes, and preserved at −80°C until assays.
Isolation of monocytes
Ten milliliter of blood anticoagulated with EDTA was diluted with equal volume of normal saline and added into the lymphocyte separation solution. The peripheral blood mononuclear cells were isolated by density gradient centrifugation at a speed of 2000 R/min and 4°C for 20 min. The isolated peripheral blood mononuclear cells were added into RPMIll640 medium containing 10% fetal bovine serum, and cultured in incubator at 37°C and 5% CO2 for 4 h. The non adherent cells were discarded and the adherent monocytes were collected by 0.25% trypsin. The adherent monocytes were washed with normal saline once and packed in 1.5 ml Eppendorf tube. The purity of purified monocytes was more than 80% by flow cytometry and Wright’s staining.
Research methods
The expression of PPAR-γmRNA in monocytes was detected by qRT-PCR. Serum IL-6 level was determined by the immunoenzymatic method using standard laboratory techniques in accordance with the manufacturer’s specifications (Quantikine Human IL-6 No. D6050 kits, R&D Systems, USA). Serum FBG level was determined using the diagnostic kits (ROCHE) by MODULARP800 automatic biochemical analyzer (ROCHE, Switzerland). Meanwhile, the patients’ blood pressure information was collected. The aneurysm parameters of all the participants were obtained through the cerebral angiography in 64 spiral CT (Philips Brilliance, GE Co., USA).
RNA isolation
According to the manufacturer’s process, total RNA was extracted from monocytes by using Trizol reagent (Invitrogen), and its purity was determined according to the optical density ratio of 260 and 280 nm. If the ratio of optical density was between 1.7 and 2.0, the RNA solution was considered without impurities, and stored at −80°C immediately.
Quantitative reverse transcription-polymerase chain reaction (qRT-PCR)
The primers (Table 1) were designed by primer 5 software. Beta-actin was used as the internal reference. The reverse transcription reaction was carried out using the TaqMan Reverse Transcription Kit (Applied Biosystems, USA) according to the manufacturer’s instructions. Quantitative polymerase chain reaction was performed using TaqMan Universal PCR Master Mix (Applied Biosystems) in an ABI 7500 Real-Time PCR system (Applied Biosystems) with the following conditions: an initial polymerase activation step for 15 min at 94°C, followed by 40 cycles of denaturation for 10 s at 94°C, annealing for 20 s at 60°C, and elongation for 30 s at 72°C. The length of the amplification product for PPAR-γmRNA was 120 bp, and for β-actin, the length was 115 bp.
Primers used in qPCR.
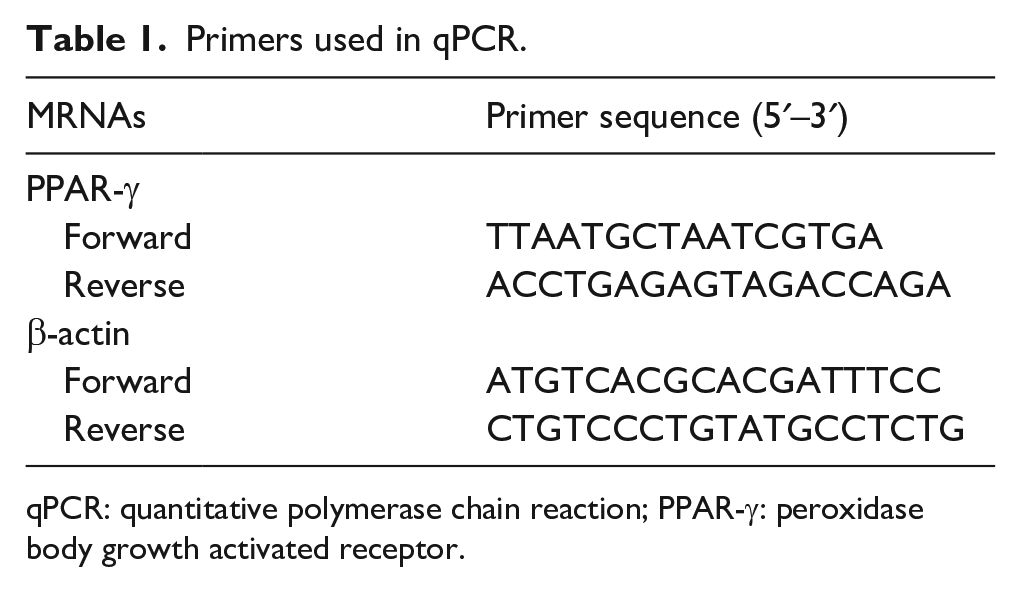
qPCR: quantitative polymerase chain reaction; PPAR-γ: peroxidase body growth activated receptor.
Analysis of gene sequences
The sequences of PCR products were determined by the sequencing machine. The sequencing results were homologously compared with the known sequences in GenBank by BLAST application software.
Detection of PPAR-γmRNA relative expression
The amplification fragments were used electrophoresis scanning to record and calculate CT values, its changes (∆CT) were used expressing mRNA expression quantity. ∆CT = (Xn) CT−(β-actin) CT, (β-actin) CT showed CT values of the internal reference, (Xn) CT showed CT values of the research objects. The relative expression quantity of mRNA was calculated by 2−∆ct.
Statistical analysis
All data were expressed as mean ± standard deviation. Chi-square test was applied to analyze the risk factors of aneurysms such as heavy drinking. The levels of mRNA, FBG, SBP, IL-6, and aneurysm characteristics were compared between two groups using Student’s t test and Mann–Whitney test. Pearson method was used in correlation analysis. Logistic regression was used to establish the relationship model between dependent variables and risk factors. The results were expressed by odds ratio (OR) and 95% confidence interval (CI). ROC curve analysis was established determining the diagnostic value of PPAR-γmRNA in rupture of IAs. The statistical significance was accepted at P < 0.05. SPSS 21 was served as the statistics software.
Results
Analysis of risk factors of IAs
The clinical data of 107 patients with saccular IAs were analyzed retrospectively. There were 50 cases with ruptured IAS and 57 cases without ruptured IAs. The levels of SBP and IL-6 in rupture group were higher than those in unrupture group (respectively, all P < 0.01). Of the studied factors, heavy drinking achieved statistical significance between ruptured and unruptured groups (P < 0.05). There was no difference in serum FBG level between the two groups (P > 0.05) (Table 2 and Figure 2).
Description of risk factors of IAs between the two groups.

FBG: fasting blood glucose; IL-6: interleukin-6; SBP: systolic blood pressure.

The risk factors of IAs: (a–c) showed the levels of FBG, SBP, and IL-6 in the rupture group and the unrupture group, respectively (Compared with unrupture group, **P < 0.01).
Analysis of IA characteristics
Our results showed that the increased presences of SR (P < 0.01) and maximum aneurysm height (P < 0.05) were described in the rupture group compared with those in the unrupture group. Patients with IAs ⩾7 mm in diameter exhibited a significantly augmented rupture risk (P < 0.05), and a similar trend in the patients with IAs in ACOM or PCOM was also found (P < 0.05) (Table 3).
Comparison of aneurysm characteristics between groups.

MAH: maximum aneurysm height; AL: aneurysm location; PCOM: posterior communicating artery; ACOM: anterior communicating artery; SR: size ratio.
Analysis of PPAR-γmRNA expression
RT-PCR analysis confirmed that the expression of PPAR-γmRNA in the rupture group was significantly lower than that in the unrupture group (P < 0.01, Figure 3).

The expression of PPAR-γmRNA in monocytes from the patients with IAs: (a) M showed DL 2000 DNA Marker (Baori Medical Biotechnology Co., Ltd., Beijing, China); 1 and 2 showed the expression of PPAR-γmRNA in monocytes of ruptured and unruptured IA patients, respectively; 3 and 4 showed β-actin and negative control group, respectively and (b) the expression of PPAR-γmRNA in the rupture group and the unrupture group were (0.47 ± 0.12, 0.86 ± 0.20, respectively) (Compared with unrupture group, **P < 0.01).
Correlation analysis
Our results showed that the PPAR-γmRNA expression was negatively correlated with IL-6 level, SR, and SBP in rupture group, respectively (all P < 0.01) (Figure 4).

Correlation analysis: (a–c) showed the correlation analysis between the expression of PPAR-γmRNA and IL-6, the expression of PPAR-γmRNA and SR, the expression of PPAR-γmRNA and SBP (respectively, all P < 0.01).
Logistic regression analysis
Logistic regression analysis showed that maximum aneurysm height ⩾7 mm (OR = 0.282, 95% CI: 1.107–12.618, P < 0.05), IAs in PCOM or ACOM (OR = 1.50, 95% CI: 1.097–8.913, P < 0.05), increased IL-6 level (OR = 2.84, 95% CI: 3.358–18.912, P < 0.05), increased SR (OR = 0.923, 95% CI: 1.361–7.172, P < 0.05), increased SBP (OR = 1.50, 95% CI: 1.584–7.865, P < 0.05), heavy drinking (OR = 0.785, 95% CI: 1.263–6.872, P < 0.05) and decreased PPAR-γmRNA (OR = 3.54, 95% CI: 6.407–43.339, P < 0.05) were independent risk factors for IAs rupture.
Analysis of receiver operating characteristic (ROC) curve
The analysis of ROC curve confirmed that the variant expression of PPAR-γmRNA could be available for prediction of IAs rupture (Figure 5).

ROC curve of PPAR-γmRNA in diagnosis of ruptured IAs. The AUC of PPAR-γmRNA was 0.867 (P < 0.01; 95% CI, 0.801–0.957). The sensitivity and specificity of PPAR-γmRNA at the best threshold for diagnosis of ruptured IAs were 87.7% and 93.9%, respectively.
Discussion
In the present study, the risk factors related to IAs rupture were clarified on the basis of the cerebral angiogram. These presences of maximum aneurysm height ⩾7 mm, aneurysm location in the PCOM or ACOM, increased SR and increased SBP were observed to be associated with IAs rupture. Furthermore, the higher levels of IL-6 and PPAR-γmRNA were revealed for rupture IAs. Logistic regression analysis showed that maximum aneurysm height ⩾7 mm, IAs in PCOM or ACOM, increased IL-6 level, increased SR, increased SBP, heavy drinking and decreased PPAR-γmRNA were independent risk factors for IA rupture. Expression of PPAR-γmRNA presented a negtive correlation with SBP, SR, and IL-6 level in the rupture group, respectively. Heavy drinking achieved statistical significance between ruptured and unruptured groups. There was no difference in serum FBG level between the two groups. ROC curve showed that AUC of PPAR-γmRNA was 0.867 and confirmed the prediction of IA rupture via the variant expression of PPAR-γmRNA.
It is reported that ruptured IAs are fatal in about 50% of cases and often cause subarachnoid hemorrhage. 1 However, the mechanism of IA rupture remains unknown at present. 16
There was an observation revealing that heavy drinking impaired outcome mainly through a poor initial condition and presence of intracerebral hematoma that contributed to the occurrence of aneurysmal subarachnoid hemorrhage, believing that alcohol consumption as a risk factor might cause the changes of blood pressure in IA patients to promote IA rupture. 6 The above evidence was also determined by ours. We believed that heavy drinking achieved statistical significance between ruptured and unruptured groups.
In analysis of the 1058 ruptured IA patients with type 2 diabetes, Lindgren et al. described that type 2 diabetes could not increase the risk of rupture of IA. 17 In this study, the FBG level did not significantly differ between ruptured and unruptured groups.
The indentation test of fresh aneurysm wall samples showed that the elastic capacity of the rupture area increased and the hardness of the remaining part of the aneurysm increased, suggesting that the biomechanical characteristics of aneurysm wall may be an integral part of the risk assessment of aneurysm rupture. 18
IA location was proposed to be associated with IA rupture. 8 A CT angiography analysis of two cases of intraventricular hemorrhage caused by ruptured anterior communicating artery aneurysms showed that the initial hemorrhage formed a high-density intraventricular angiography image, and then the injection of contrast agent could be observed. 19 In this study, IA location in the ACOM or PCOM achieved statistical significance between the two groups.
Aneurysm size has been studied extensively as a parameter for IA rupture risk prediction. A study found that subarachnoid hemorrhage caused by very small aneurysms (⩽3 mm) is not uncommon. Because of the spatial structure of the arteries and the thin and fragile walls of the arteries in the IA patients, very small aneurysms have higher treatment risk than large aneurysms. 20 some evidences revealed that small unruptured IAs (<7 mm) were significant risk factors for IA rupture.21,22 However, the contrary observation has also been reported. A study of 1256 cases of sporadic ruptured IAs in a single Chinese institution reported that ninety percent of patients had ruptured IAs <10 mm in size. The findings indicated that an unruptured IA of ⩾7 mm was an independent risk factor for the lifetime occurrence of SAH and the hazard ratio was more than 3.35. The rupture risk of an unruptured IA <7 mm was minimal (<1% per year). 9 In this study, compared with unruptured IAs, the maximum height of ruptured IAs ⩾7 mm was statistically significant, indicating that the larger IAs might contributed to a increased ruptured risk. In the study of Dhar et al., 14 a logistic regression analysis showed that SR was positively correlated with IA rupture. SR was more than 2.05 in 75% of ruptured aneurysms and <2.05 in 83% of unruptured aneurysms. In addition, SR was found to be (1.5 ± 0.7) easily contributing to IA rupture. 23 This is consistent with our results that the SR significantly increased the rupture risk of unruptured IAs. In this study, SR were found to be negatively correlated with the expression of PPAR-γmRNA. These evidences further support that PPAR-γ may be involved in the pathogenesis of IA rupture.
Inflammation is thought as a key component of the pathophysiology of IAs. Inhibition of inflammation is proposed as a modification of the natural process preventing the development of aneurysmal subarachnoid hemorrhage.24,25 The analysis of Cebral et al. 26 in tissue samples from 18 cases of saccular IAs indicated that the increased inflammation contributed to some changes of IA wall such as high wall shear stress, higher vorticity, higher viscous dissipation, and high shear rate, which was associated with degeneration of IA wall and rupture of IAs. Our study showed that the level of IL-6 was significantly increased in the patients with ruptured IAs, and and IL-6 was a risk factor for ruptured IAs. This confirmed the conclusion of the above literatures. Recent evidences suggested that PPAR-γ reduced inflammation and affected the progression and rupture of IA.27,28 These facts has also been demonstrated by Hasan et al. 29 and Shimada et al. 8 An animal experiment concluded that the levels of IL-6 and IL-1β were significantly lower in the aneurysm tissues collected from pioglitazone-treated mice (pioglitazone, PPARγ agonist) than those in the control group, further establishing the protective effect of PPAR-γ activation on the development of IA. 8 We provided the insight of the significantly decreased expression of PPAR-γmRNA in the ruptured group compared to that in the unruptured group for IA patients, and an inverse correlation was described between serum IL-6 level and PPAR-γmRNA expression in the rupture group, suggesting that PPAR-γ may participate in the pathological process of IA rupture induced by inflammatory reaction.
In analysis of mice IA induced by a combination of deoxycorticosterone acetate salt induced hypertension and a single injection of elastase into the cerebrospinal fluid, normalization of blood pressure after aneurysm formation prevented aneurismal rupture in mice, further revealing that relationship between changes of blood pressure and rupture of aneurysms, and the inhibition of the local renin-angiotensin system independent from the reduction of blood pressure can prevent aneurysmal rupture. 30 Our data indicated the increased SBP in the patients with the ruptured IAs compared to the unruptured IAs and the variation of blood pressure as a risk factor inducing rupture of IA. The findings of Jones et al. 31 clarified that the protective ability of PPAR-γ to the development and rupture of aortic aneurysm in angiotensin II—treated apolipoprotein E knockout mice. Despite different underlying pathologies between these two types of aneurysms, a similar mechanism that activation of PPAR-γ opposing to aneurysm rupture has been observed. In this study, SBP were found to be negatively correlated with the expression of PPAR-γmRNA, further proving that PPAR-γ was related to IA rupture.
In this study, PPAR-γmRNA had the higher AUC of 0.867 and nice sensitivity and specificity from ROC curve for ruptured IAs. Hence, we speculated that PPAR-γ had the strongest independent correlation with IA rupture.
Conclusion
The results of this article showed that IA location, heavy drinking, the increase of SR, SBP, and IL-6 level, as well as decreased PPAR-γmRNA expression may be related to rupture of unruptured IAs. And more interestingly, PPAR-γmRNA expression was negatively correlated with IL-6 level, SR and SBP for ruptured IAs, respectively, suggesting that PPAR-γ may be involved in the occurrence, development and rupture of IA. Hence, detecting PPAR-γmRNA expression can be saved as prediction for the rupture of unruptured IAs.
It is hoped that this paper will be of some help to provide a certain amount of information for exploring the occurrence and development of IA and its rupture mechanism, and for determining the susceptibility genes and screening susceptible population.
Limitations
PPAR-γ is a lipid sensing nuclear transcription factor with many intracellular ligands, which plays key roles in lipid storage and mobilization, glucose metabolism, morphogenesis, and inflammation. 32 The activation of PPAR-γ is also affected by other factors, such as activation of the canonical Wingless/Int (WNT)/β-catenin pathway induces inactivation of PPAR-γ. 33 Although the expression of PPAR-γ in macrophages can identify aneurysms at risk of rupture, it is impossible to rule out that these levels change only after IAs rupture. Therefore, the prediction of PPAR-γ on rupture of IAs in this study is only a preliminary analysis, and further diagnosis should be combined with other risk factors.
Compared with Hasan DM and Shimada K using animal samples, we analyzed the relationship between the level of PPAR-γ in human macrophages and the rupture of IAs, and further analyzed the relationship of the size and location of aneurysms, blood glucose and blood pressure of patients as well as heavy drinking with the rupture of IAs. In addition, in the methods, this study did not use software to calculate the power of statistics and justify the size of sample for different experiments.
Footnotes
Acknowledgements
Yi Liu, our colleague, master graduate students, offered writing assistance.
Author contributions
XZ, YX-K and YL-Z implemented the experiment and written. XZ, WK, TJ and YL-Z performed design topic, and modifyed in reviewing. XZ and TJ were responsible for the article. All the authors final approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from research ethics committee of Xianyang first people’s Hospital and Xianyang Hospital of Yan’an University (Approval number: 2019A-019).
Informed consent
The requirement for informed consent was waived by the ethics commissions (Waiver number: 2019B-003).
Consent for publication
All authors approved to submit this original article.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
