Abstract
Ulcerative proctitis (UP) is a common form of ulcerative colitis (UC), the number of patients with UP is increasing. Suppository treatment containing mesalamine is usually the first-line choice for mild to moderate UP, here, we review the publications on mesalamine suppository in UP, to better understand its dosage, duration of therapy and adverse events in both induction and maintenance phases. We also review articles on budesonide, beclomethasone, tacrolimus, herbal medicine and acetarsol suppositories in UP, to present therapeutic strategies for patients with UP refractory to mesalamine suppository.
Introduction
Ulcerative proctitis (UP) is a form of ulcerative colitis (UC), a chronic inflammatory condition that causes long-lasting inflammation and ulcers of a variable extent of the colon, the characterized symptoms of UC include rectal bleeding, tenesmus, periodic diarrhea or constipation, and rectal pain. In UP, the inflammation is confined to the rectum. 1 About 37% and 32% of people diagnosed with UC in Asia and Australia is actually UP. 2 Rectal therapy of mesalamine in the form of suppositories or enemas has demonstrated high concentrations of the active drug at the site of inflammation, and low systemic absorption, 3 and is therefore usually the first-line choice for UP recommended in several international guidelines.1,4–6 Suppositories are more appropriate than enemas as they deliver the drug optically to the rectum or adjacent sigmoid colon, and are more acceptable for patients with UP, and they were retained for three hours.7,8 The aim of this article is to review the current options and novel developments of suppository treatment in UP.
Mesalamine
Mesalamine (5-aminosalicylic acid) is the active component of sulfasalazine, it has multiple anti-inflammatory effects, such as inhibition of cyclooxygenase, lipoxygenase, B-cells, and several key inflammatory cytokines. 9 A Cochrane review 10 of 38 clinical studies has shown rectal mesalamine superiority in comparison to placebo for inducing clinical, endoscopic and histological improvement and remission in patients with distal UC, though this review assessed treatment of distal UC, which included both UP and left-sided UC. Another meta-analysis 11 also reported 62% patients with UC achieved mucosal healing by mesalamine suppository treatment.
Dosage and duration of therapy
Induction phase
In 1987, Williams et al. 8 firstly reported the efficacy of mesalamine suppository 0.5 g three times daily (TID) in patients with distal proctitis (a subset of idiopathic UC) was 78.6%. Campieri et al. 12 later confirmed the efficacy of mesalamine 0.5 g TID to treat distal UC (<20 cm); after 1 month treatment, 28/32 (87%) patients in mesalamine group were either in clinical remission (n = 18) or improved (n = 10) compared with 10/30 (33%) patients in the placebo group. Authors suggested further study to find out the minimum dosage necessary to give the maximum benefit. Then in 1990, Williams 13 reported mesalamine suppositories 0.5 g twice per day (BID) and 0.5 g TID have similar efficacy in treating UP. After that, the dosage of 1 g QD was frequently used in trials.
In 1992, Ngô et al. 14 reported the efficacy of mesalamine suppository 1 g once daily (QD) in a double-blind, placebo-controlled, comparative study of 50 patients with UP. Both endoscopic and clinical remission were seen in 65% of mesalamine-treated patients and in 25% of placebo-treated patients respectively (p ⩽ 0.01), after 2-week treatment. In 2013, Watanabe et al. 15 reported an endoscopic remission (mucosal score ⩽1) rates after 4-week treatment with1 g mesalamine suppository QD of 83.8%, through a multicenter, randomized, double-blind, placebo-controlled study. Yamamoto et al.’s 16 study in 2015 demonstrated a clinical remission of 74% and an endoscopic healing (score 0) rate of 55% after 8-week treatment of mesalamine 1 g suppository QD.
Andus et al. 17 confirmed the noninferiority of 1 g mesalamine suppository QD administration in comparison to 0.5 g suppositories TID treatments, in 2010, by conducting a randomized, investigator-blinded, comparative trial. The clinical remission rates, defined as Disease Activity Index (DAI) <4 at the final visit (the sixth week), were 87.9% in the 1 g QD group and 90.7% in the 0.5 g TID group in the per protocol analysis (p = 0.00027 for non-inferiority); and they were 84.0% versus 84.7% (p < 0.00008) in the intention-to-treat analysis. However, as to the histological assessment, the 0.5 g mesalamine TID group achieved higher remission rates of 67.9% than 1 g mesalamine QD group (55.3%). Moreover, 86% of the patients preferred the 1 g suppository QD. Similar efficacy of mesalamine suppository 1 g once nightly (QHS) and 0.5 g BID was later reported by Lamet 18 in 2011.
Furthermore, the effect of mesalamine suppository in children and adolescents was also demonstrated. Heyman et al. 19 conducted a multicenter, open-label trial to evaluate the efficacy of 0.5 g mesalamine suppositories in treating pediatric patients with mild-moderate UP. The mean DAI value decreased from 5.5 at baseline to 1.6 and 1.5 after 3- and 6-weeks treatments, respectively (p < 0.0001). Summary of the trials is shown in Table 1.
Remission rates of mesalamine suppositories in UP.
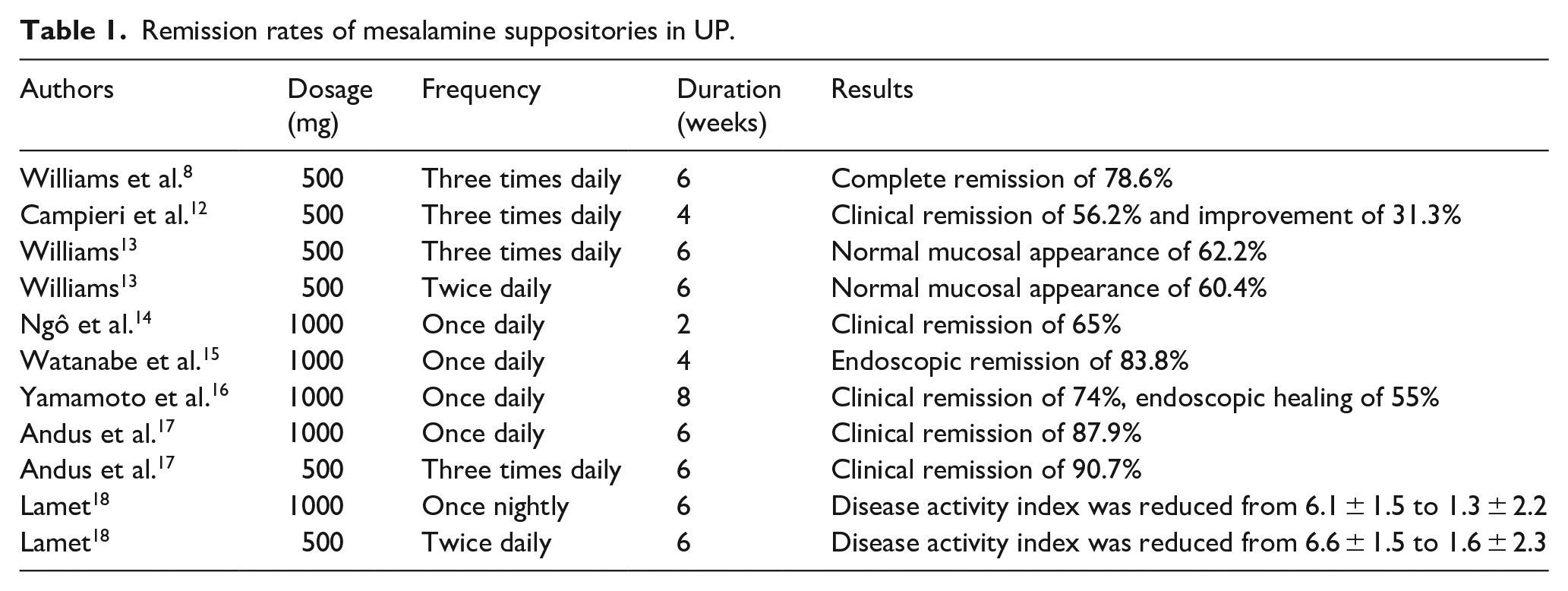
In conclusion, mesalamine suppository QD is as effective as divided doses, and dose-ranging studies support the use of QD administration of 1 g mesalamine suppositories for the induction of clinical and endoscopic remission in patients with mild-to-moderate UP. Thus, mesalamine 1 g suppository QD is recommended as the first-line therapy for mild or moderately active proctitis by European (ECCO), British, and American Guidelines.1,4–6 Nevertheless, higher histological remission rate was found in 0.5 g mesalamine suppositories TID compared to 1 g QD in Andus et al.’s 17 study, and an inverse correlation between mucosal concentrations of mesalamine and the level of disease activity in UC patients was found in Frieri et al.’s 20 study. Thus, further study is necessary to assess the significance of these observations. The duration of therapy for induction was not constant in all studies, 2, 4, 6, or 8 weeks? It is difficult to make a recommendation, further study on this subject is obligatory for a decision.
Maintenance phase
A Cochrane meta-analysis 21 of nine studies (484 patients) assessed the efficacy and safety of rectal mesalamine for maintaining remission of distal UC. The results showed that rectal mesalamine was significantly superior to placebo for maintenance of both symptomatic and endoscopic remission over a period of 12 months. Again, this review included patients with not only UP, but also left-sided UC.
Hanauer et al. 22 performed a 24-month, multicenter, double-blind trial in patients with UP in endoscopic remission, in order to evaluate the efficacy and safety of 0.5 g mesalamine suppository QHS as a maintenance therapy for these patients. Sixty-five patients were randomly assigned to either 500-mg mesalamine suppository QHS or a matching placebo treatment. The remission rate in mesalamine group was 68% (vs 14% in placebo group, p < 0.001) at 12 months, and 54% (vs 11% in placebo group, p = 0.001) at 24 months. Mean time to relapse was 453.4 days in mesalamine group and 158.0 days in placebo group. This result is consistent with those from Marteau et al., 23 the later reported mesalazine suppositories 1 g three times a week were effective to prevent relapses of UP, the remission rate was 62% at 9 months (vs 38% in placebo group, p = 0.031). While D’Arienzo et al. 24 in 1990 reported a cumulative remission rate of 92% in mesalamine suppositories (400 mg BID) group, and 21% in the placebo group, at 12 months. Yamamoto et al.’s 16 reported a remission rate of 70% with 1g QD at 10 months.
As to the frequency of therapy, in Yamamoto et al.’s 16 study, 20 out of 38 (53%) patients who ceased the suppository treatment relapsed while 24/80 (30%) of those who continued the treatment relapsed (p = 0.03); while the relapse rate was similar between patients with QD administration and those who decreased frequency (22% vs 43%, p = 0.08). This result is consistent with those found in mesalamine enema, which is not necessary on a daily basis in preventing relapse but can be applied less frequently as for example 7 days per month 25 or two to three times per week. 23 Summary of the trials is shown in Table 2.
Maintenance treatment of mesalamine suppositories in UP.

In summary, mesalamine suppository is effective for maintaining remission of UP with indefinite dosage, ranging from 1 g QD to 1 g three times per week. Though British and American Guidelines4,6 recommended 1 g QD to maintain remission, more data are needed to decide the dosage and frequency. As to the duration, we recommend as long as possible, as UP is a lifelong disease.
Adverse events
The safety profile of mesalamine suppositories was favorable in patients with UP. Adverse events (AEs) reported were comparable between mesalamine and placebo groups in the available randomized blind controlled trials, the incidence was 15.4%–23%. Frequently reported AEs were flatulence, diarrhea, abdominal pain, headache, etc.14,16,19 Gastrointestinal disorders (mainly abdominal pain, diarrhea, and vomiting) were reported in about 60% of the children and adolescent patients. 18 Besides, Kim et al. 26 reported a case of acute eosinophilic pneumonia related to a mesalamine suppository, the patient recovered after stopping the treatment and starting systemic steroid. Ding et al. 27 reported a case of UC flair induced by mesalamine suppositories hypersensitivity.
Mesalamine suppository in relapsed UP
Intermittent data from Kato et al. 28 demonstrated that mesalamine suppository was superior to dose intensification of oral medication for treating aggravation of UP. They conducted a randomized prospective study of 35 patients with mild UP taking 2.4 g oral mesalamine daily as the maintenance regimen, these patients were having bloody stools when enrolled. Patients were randomized to receive mesalamine suppositories (1 g/day) or oral mesalamine (3.6 g/day). More patients achieved the primary endpoint (disappearance of bloody stool after 2 weeks treatment) in the suppository group (75%, 12/16) than in the oral group (26%, 5/19, p < 0.01). Authors also reported a significantly higher decreased level of proctoscopic scores from the baseline in the mesalamine suppository group than that in the oral group, but data were not shown. More data are needed to identify the efficacy of mesalamine suppository in relapsed UP.
Rectal corticosteroids
Current guidelines1,4–6 advice locally administered corticosteroids in those who failed mesalamine suppository, however, rectal corticosteroids induce remission in only 46% of patients 29 and put patients at risks of systemic side effects, such as suppression of the hypothalamic-pituitary-adrenal axis. 30 Budesonide and Betamethasone are commonly used as topical formulations, depending on the region.
Budesonide
Budesonide is an anti-inflammatory drug. Kruis 31 conducted a double-blind trial in 337 patients with active UP, in order to compare the efficacies and safeties of four different suppositories: 2 mg budesonide, 4 mg budesonide, 1 g mesalamine, and 2 mg budesonide plus 1 g mesalamine. The mean time to resolution of symptoms in the 4 mg budesonide (29.8 days) and combination (29.3 days) groups was similar with that of 1 g mesalamine treatment (29.2 days). Proportions of patients with clinical, endoscopic, and mucosal healing were similar among the four groups. Authors concluded that budesonide suppositories offered an alternative therapy to mesalamine for topical treatment of UP. Further studies are needed to find out whether single 4 mg budesonide suppository is an option for patients with UP refractory to mesalamine suppositories.
Beclomethasone
Beclomethasone is a second-generation corticosteroid. Many studies have demonstrated the efficacy and safety of topical beclomethasone (enema and foam) in active mild-moderate distal UC.32–34 Recently, Lie et al. 35 conducted a randomized controlled, double-blind study to compare beclomethasone suppositories (3 mg/day) with tacrolimus suppositories (2 mg/day) for 4 weeks in 85 patients with UP refractory to mesalamine. After 4-week treatment, similar amount of patients achieved clinical remission (38.5% vs 45.7%) and clinical response (59.5% vs 62.9%) in beclomethasone group and tacrolimus group. Besides, non-significant difference was found in AEs between groups.
Tacrolimus
Tacrolimus is a calcineurin inhibitor, also known as FK506, is a potent immunomodulatory drug. 36 It is effective in the systemic treatment of inflammatory bowel disease (IBD), but the potential toxicity and side effects after oral intake limit its use. Topical tacrolimus has already been investigated in UP in several clinical trials,37–39 Rectal tacrolimus of ointment was used in Lawrance and Copeland 37 and Lawrance et al.’s 39 studies, showing an excellent efficacy and safety. Three patients (75%) went into remission in Lawrance and Copeland’s study. 37 Eight out of 11 (73%) patients in Lawrance et al.’s study 39 achieved mucosal healing after 8 weeks of treatment of rectal tacrolimus. Van Dieren et al. 38 confirmed the safety and efficacy of tacrolimus suppository in refractory UP. In this study, 8 out of 12 patients with refractory UP achieved significant improvement of histological appearance after 4-week treatment of daily tacrolimus 2 mg suppository, and a significant histological improvement was observed in the suppository group (67%, p = 0.004). None of the patients developed AEs, and the whole blood trough levels of tacrolimus were less than 5 µg/L. Recently, Jaeger et al. 40 also reported a remission rate of 52.3% after 2 mg tacrolimus suppositories treatment.
Taken together, tacrolimus suppositories can be considered as a promising alternative treatment for patients with UP who have failed traditional therapies. However, in view of the small sample size, these clinical trials call for the initiation of randomized, placebo-controlled trials with larger numbers. Besides, Tacrolimus suppositories actually may result in substantial plasma levels 40 and therefore are not without side effects, so close monitoring and dose adjustment are recommended to avoid toxicity.
Herbal medicine
Traditional Chinese medicine (TCM) originated in ancient China and has evolved over thousands of years, it belongs to Complementary and Alternative Medicine. The efficacy of two herbal preparations Xilei-san and Qingchang suppository, in treating UP, have been proved with randomized, controlled trials.
Xilei-san is a traditional herbal preparation with a history of healing power for inflamed mucosa. Fukunaga et al. 41 conducted a randomized, double blind trail to compare the effectiveness of topical Xilei-san (0.1 g QD, n = 15) with placebo (n = 15) in patients with intractable UP. On day 14, the number of patients who achieved clinical remission in Xilei-san group was significantly higher compared with placebo (45.5% vs 0%, p < 0.04). On day 180, one patient in the Xilei-san group relapsed while 10 patients in the placebo group relapsed (p < 0.001).
Qingchang suppository is an in house suppository from Longhua Hospital. Gong et al. 42 conducted a randomized, controlled trial in multicenter, to compare the efficacy and safety of 2-month treatment of Qingchang suppository (3 g QHS, n = 47) with sulfasalazine suppository (0.5 g QHS, n = 47). The number of patients who achieved complete remission, defined as no clinical symptom and normal endoscopic finding, was similar between groups, 61.70% in Qingchang suppository group versus 51.06% in sulfasalazine suppository group (p > 0.05). During the 1 year follow up, 4/47 (9.30%) patients in Qingchang suppository group relapsed, while 11/47 (26.83%) patients in sulfasalazine suppository group found to be relapsed (p < 0.05).
Besides, a single-agent Indigo Naturalis (IN) suppository was evaluated by Yoshimatsu et al. 43 in an open-label, single-center, prospective pilot study. A total of 10 patients, who were refractory to topical mesalamine or corticosteroids, were treated with 50 mg IN QD for 4 weeks. At week 4, the rates of clinical remission and mucosal healing were 30% and 40%, respectively. IN is a key ingredient in Xilei-san and Qingchang suppository.
In conclusion, for patients with active UP but showing poor response to the existing topical treatments, TCM suppository might be a considerable choice. Besides, it remains to be tested if Xilei San has better efficacy in treating naive patients with UP. And more prospective, randomized, controlled clinical trials are needed to justify the efficacy and safety of TCM suppository in treating patients with active UP.
Acetarsol
Acetarsol is a pentavalent arsenical compound derived from organic arsenic. Acetarsol suppository was first described as an effective treatment for proctitis in 1965. 44 Connell et al. conducted a randomized, double-blind trial of 44 patients with idiopathic proctitis. The patients received two nightly treatments for 3 weeks, with either 5 mg prednisolone suppositories (n = 21) or 250 mg acetarsol suppositories (n = 23). Twenty patients in each group attended for follow up and underwent sigmoidoscopy, nineteen patients (82.6%) in the acetarsol group and 17 (80.9%) patients in the prednisolone group achieved endoscopic improvement. One patient developed jaundice after 2 weeks of acetarsol treatment, which settled upon drug cessation.
In 2018, Kiely et al. 45 performed a retrospective analysis of 39 patients with refractory proctitis. Among these 39 patients, 29 were diagnosed with UC, 9 with Crohn’s disease, and 1 with indeterminate colitis. The median duration of disease for these patients was 6.9 years (SD ± 5.0), and they had tried a median of 5.5 ± 1.2 different interventions before commencing acetarsol, including oral or topical 5-ASA, systemic or topical corticosteroid, thiopurine, methotrexate and anti-TNF agents. The results showed in these 39 patients treated with acetarsol suppositories, 26 patients (68%) achieved clinical response, defined as improvement in symptoms to “normal” and/or an endoscopic sub-score of 1/0. Eleven out of 39 patients had endoscopic scores before and after treatment, and among them, 9 (82%) patients went into endoscopic improvement and 5 (45%) achieved complete remission (Mayo endoscopic sub-score = 0, p = 0.006). One patient experienced a macular skin rash (consistent with a drug rash) 1 week after starting acetarsol, and the rash resolved within 4 weeks of drug withdrawal. Argyriou et al. 46 retrospectively reviewed clinical records of 28 UP patients, who had failed mesalamine or corticosteroid topical therapy, received acetarsol suppositories at Nottingham University Hospitals since 2012, and found that 67.9% patients went into clinical response and 46.4% achieved clinical remission.
These studies demonstrated acetarsol suppository is effective in refractory UP, and with few AEs, but more prospective, randomized, controlled clinical studies are obligated to justify the effectiveness and safety of acetarsol suppository.
Conclusion
Suppository treatment is the mainstay in inducing and maintaining remission of UP. Mesalamine suppository 1 g QD is recommended as the first-line choice to induce remission, but it’s hard to say whether a longer topical mesalamine induction regimen offers further benefit for patients over a shorter induction regimen, basing on the data available. With respect to maintaining remission of UP, the mesalamine dosage is uncertain, ranging from 1 g QD to 1 g three times per week; and we suggest patients to maintain mesalamine suppository as long as possible, as UP is a lifelong disease. For patients refractory to mesalamine, budesonide, beclomethasone, tacrolimus, Xilei-san, and Qingchang suppository, as well as acetarsol suppository might offer benefit. No trials of maintenance rectal corticosteroids/tacrolimus/acetarsol were identified, and thus their long-term effectiveness and safety are unknown, patients refractory to mesalamine might use Xilei-san and Qingchang suppository as a maintenance therapy. A suggested algorithm based on current evidence for treatment of mild to moderate UP is shown in Figure 1.

Mesalamine suppository 1 g QD is recommended as the first-line choice to induce and maintain remission,1,4–6 with an efficacy above 80% 1 in induction phase. Budesonide/Beclomethasone/Tacrolimus/Acetarsol suppository might offer benefit for patients with UP refractory to mesalamine suppositories in induction phase.31,34,40,46 Herbal preparations of Xilei-san 41 and Qingchang suppository 42 might be a considerable choice for patients with UP refractory to mesalamine to induce and maintain remission.
Most patients with mild to moderate UP may benefit from the use of effective suppositories, however, it requires acceptance of both patients and doctors. Studies47,48 have expressed concern over the underuse of rectal therapies, though one retrospective study later 49 also found that majority of patients with UP were treated with topical drugs. To better popularizing suppositories in patients with UP, we suggest to emphasize education and training of physicians and patients, and adherence to therapy may also be optimized by better information and teaching of the patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Natural Science Foundation of China (No. 81703986), Scholar Project of Longhua hospital (No. LYTD-45), and Research Project of Longhua Hospital in 2016 (No. 2016YR02)
