Abstract
This paper focused on evaluating the effect of TLR4-MyD88-NF-κB signaling pathway in the progression of secondary hepatic injury and fibrosis in hepatolithiasis. The levels of inflammatory factors (IL-1β, IL-6, TNF-α) and serum biochemical values (ALT, AST, Tbil, Dbil, ALP, GGT) were detected by ELISA. IHC was used to detected the expression level of TLR4 in liver tissues of hepatolithiasis patients and mice. The pathological changes of liver tissue were observed by HE staining. The levels of MyD88, NF-κB, IκB, Laminin (LN), and chitosan enzyme 3-like protein 1 (CHI3L1) were detected by western blotting. In hepatolithiasis patients, the levels of TNF-α, IL-1β, and IL-6 were distinctly raised and proteins associated with TLR4-MyD88-NF-κB signaling pathway (such as TLR4, MyD88, NF-κB, and IκB) in liver tissues were significantly up-regulated. In Bile duct ligation (BDL) model of mice, the results showed that in addition to the significant increase of inflammatory factors, liver function indexes, and fibrosis indexes in BDL mice were also significantly up-regulated. Additionally, TLR4-MyD88-NF-κB signaling pathway was activated in BDL mice. After TLR4 knockdown in BDL mice, inflammatory factors, liver function indexes, and fibrosis indexes were significantly down-regulated. TLR4-MyD88-NF-κB signaling pathway proteins were restrained. TLR4-MyD88-NF-κB signaling pathway took part in the progression of secondary hepatic injury and fibrosis in hepatolithiasis. Inhibition of TLR4-MyD88-NF-κB signaling pathway can reduce the progression of secondary hepatic injury and fibrosis in hepatolithiasis.
Introduction
Hepatolithiasis was common and refractory bile duct diseases, which was commonly in East Asian countries. 1 At present, the treatment for hepatolithiasis was mainly surgery, and the cure rate was high. 2 However, the secondary hepatic injury caused by hepatolithiasis and the potential risk of liver fibrosis may often be ignored. At present, little was known about the pathogenesis of secondary hepatic injury and fibrosis in hepatolithiasis.
The main reason why TLR4-MyD88-NF-κB signaling pathway can play a vital role in immune and inflammatory mechanisms is that it mainly contains some signaling pathway proteins, including toll-like receptor 4 (TLR4), myeloid differentiation factor 88 (MyD88), and nuclear factor-κB (NF-κB).3–5 It has been reported that Toll-like receptors (TLRs) are known as pathogen-associated molecular patterns (PAMPs). 6 As a vital member of the TLR family, TLR4 is a momentous inflammatory regulator. 7 TLR4-mediated downstream signaling pathways included primary MyD88 dependent and independent pathways. Primary MyD88 dependent pathway mainly regulated the expression of a variety of inflammation related genes, which transmitted intracellular signals through the TIR domain of MyD88. This could activate the transcription factor NF-κB, thus promoting the release of inflammatory factors IL-1, IL-6, and TNF-α.8–10 The discovery that TLR4-MyD88-NF-κB signaling pathway played a momentous role in many diseases had been described in many studies. For example, an important finding has shown that TLR4-MyD88-NF-κB signaling pathway had a certain effect on myocarditis after coronary microembolization (CME). 11 In addition, previous studies have reported that TLR4-MyD88-NF-κB signaling pathway was associated with contrast-induced renal tubular epithelial cell injury by inducing inflammation and apoptosis. 12 Hence, TLR4-MyD88-NF-κB signaling pathway played a significant role in the study of inflammatory diseases.
Studies have shown that secondary hepatic injury caused by hepatolithiasis was closely related to inflammatory response, and liver fibrosis was caused by the release of a large number of inflammatory factors.13,14 However, it was unclear the effects of TLR4-MyD88-NF-κB signaling pathway in the progression of secondary hepatic injury and fibrosis in hepatolithiasis. Therefore, this study intended to collect liver tissue and peripheral blood from normal subjects, mild hepatolithiasis patients and fibrotic hepatolithiasis patients, and evaluated the inflammatory response and activation of TLR4-MyD88-NF-κB signaling pathway in patients. We established an animal model of bile duct ligation (BDL) in mice to evaluate the biological role of TLR4-MyD88-NF-κB signaling pathway in the progression of secondary liver injury and fibrosis in hepatolithiasis, and TLR4 knockout mice were used to verify the effect of the TLR4-MyD88-NF-κB signaling pathway.
Together, the main purpose of this paper was to explore the effects of TLR4-MyD88-NF-κB signaling pathway in the progression of secondary liver injury and fibrosis in hepatolithiasis. This may provide some enlightenment for the clinical treatment and prognosis of hepatolithiasis.
Materials and methods
Animal
Animal Ethics Committee of Hunan Provincial People’s Hospital approved the implementation of all programs (No. 2019. Wei. 74). All protocols were operated according to the requirements for the application of laboratory animals issued by the National Institutes of Health. Forty mice (20–35 g) were kept in captivity at 25°C for 12 h/12 h light/dark periods, and they can get standard rat food and tap water from the Medical Laboratory Animal Center of Hunan Provincial People’s Hospital for free.
Clinical tissue samples and treatment
The patients who were diagnosed as hepatic hemangioma and hepatolithiasis by imageological diagnosis, serological, or histopathological examination from the Department of Hepatopathy, The Hunan Provincial People’s Hospital from November 2019 to June 2020 were collected. Among them, patients with hepatolithiasis met the diagnostic criteria of hepatolithiasis in the 2006 Guidelines for the Diagnosis and Treatment of Hepatolithiasis of the Biliary Surgery Group of the Chinese Medical Association. The METAVIR scoring system was used to determine the stage of fibrosis, 15 Mild hepatolithiasis was in F0 stage, and Fibrotic hepatolithiasis was in F3 stage. Inclusion criteria were as follows: (1) Conformed to the diagnostic criteria of hepatolithiasis in the 2006 Guidelines for the Diagnosis and Treatment of Hepatolithiasis of the Biliary Surgery Group of the Chinese Medical Association. (2) The clinical pathological data are kept intact. (3) patients have informed consent and cooperated with the research. The exclusion criteria were: (1) Combined with viral hepatitis, autoimmune liver disease, alcoholic liver disease, non-alcoholic fatty liver disease, and other liver diseases. (2) Merged liver and other types of malignant tumors. (3) There were systemic or local uncontrolled active infections. (4) Merged autoimmune system diseases. (5) Patients with missing pathological data. We have obtained the written informed consent of the subjects before the start of the study, and obtained the approval of the Medical Ethics Committee of Hunan Provincial People’s Hospital (No. 2019. 74). Liver tissue and peripheral blood were collected from hepatic hemangioma patients (Normal control), mild hepatolithiasis patients, and fibrotic hepatolithiasis patients. The collected liver tissue and peripheral blood were used to detect the inflammatory response and the activation of TLR4-MyD88-NF-κB signaling pathway. We froze tissue samples at −80°C in liquid nitrogen and stored them for western blotting and ELISA. At the same time, we fixed the liver tissue samples with 4% paraformaldehyde, and then embedded it with paraffin. After that, sections (4 μm thickness) were made. The prepared sections were used for IHC detection.
Bile duct ligation (BDL) model of mice
Male C57 mice aged 4–8 weeks (weight about 20–35 g) were selected for preoperative fasting for 12 h. We divided thirty male C57 mice randomly into Normal, Sham and Model groups (
HE staining
HE staining was performed to evaluate the pathological changes of liver tissue. The tissue sections were roasted at 60°C for 1–2 h. The slices were immersed in xylene for 10 min until dewaxing twice to water. Next, we put them in 100%, 95%, 85%, and 75% ethanol for 5 min. After that, we soaked the slices in distilled water for 5 min, set them with hematoxylin for 5–10 min, rinsed them with distilled water and blued them with PBS. Then the slices were soaked in distilled water for 5 min. We used hematoxylin to shape the tissue sections for about 5–10 min, then washed them with distilled water, and finally blued them with PBS. After dyeing with eosin for 3–5 min, the slices were rinsed with distilled water. The slices were dehydrated with 95%–100% alcohol for 5 min each time. The slices were removed and immersed in xylene for 10 min, and repeated twice. Then we sealed the sections with neutral gum and observed them under microscope.
Enzyme linked immunosorbent assay (ELISA)
The levels of inflammatory factors (IL-1β, IL-6, TNF-α) and liver function indexes alanine aminotransferase (ALT), aspartate aminotransferase (AST), total bilirubin (Tbil), direct bilirubin (Dbil), alkaline phosphatase (ALP), and γ-glutamyl transpeptidase (GGT) were detected by ELISA. IL-1β (#CSB-E08053h, C23037037, CUSABIO), IL-6 (#CSB-E04638h, E31033828, CUSABIO), TNF-α (#CSB-E04740h, P05031137, CUSABIO), ALT (#C009-2-1, Nanjing Jiancheng Bioengineering Institute), AST (#C0010-2-1, Nanjing Jiancheng Bioengineering Institute), Tbil (#C019-1-1, Nanjing Jiancheng Bioengineering Institute), Dbil (#C019-2-1, Nanjing Jiancheng Bioengineering Institute), ALP (#A0509-2-1, Nanjing Jiancheng Bioengineering Institute), and GGT (#C017-1-1, Nanjing Jiancheng Bioengineering Institute) quantitative ELISA kit were used to detect the expression of inflammatory factors IL-1β, IL-6, TNF-α, and liver function indexes ALT, AST, Tbil, Dbil, ALP, and GGT according to the instruction. The OD value was read by Bio-Tek microplate reader (MB-530, Heales, China).
Immunohistochemistry (IHC)
Tissue slices were baked at 60°C for 12 h successively. We immersed the tissue sections in xylene for 20 min, and repeated three times. After that, the tissue sections were soaked with different concentrations of ethanol (100%, 95%, 85%, 75%) for 5 min. Then they were soaked in distilled water for 5 min. We put the slices into 0.01 M citrate buffer solution (PH6.0) and then heated them in electric furnace or microwave oven until boiling. After cooling, they were washed with 0.01 M PBS (pH 7.2–7.6) for 3 min every time, added 1% periodate and inactivated endogenous enzymes at room temperature for 10 min. PBS rinsed for 3 min every time. Then, a suitably diluted primary antibody TLR4 was added to it at 4°C overnight. The PBS buffer solution was used to wash them for 5 min, and repeated twice. The secondary antibody was incubated by dropping 50–100 μL anti-rabbit-IgG antibody-HRP polymer and incubated at 37°C for 30 min, then washed with PBS for 5 min each time. We dropped the prepared color developing agent DAB working solution of 50–100 μL, incubated for 1–5 min at room temperature, controlled the reaction time under the microscope and washed with distilled water. Hematoxylin was dyed for 5–10 min and washed with distilled water, and finally blued them with PBS. All levels of alcohol are dehydrated for 5 min. After taking it out, it was placed in xylene for 10 min twice and then sealed with neutral gum and observed under microscope. The above operations were repeated three times.
Western blotting
Total protein was extracted from liver tissues of normal subjects, mild hepatolithiasis patients and fibrotic hepatolithiasis patients, and BDL mice. Then the total protein was separated by 10% SDS-PAGE and transferred to PVDF membrane (millipore, Atlanta, Georgia, USA). About 5% skim milk was used to seal these membranes at room temperature. Then the primary antibodies against MyD88, NF-κB, IκB, Laminin (LN), and chitosan enzyme 3-like protein 1 (CHI3L1) were incubated overnight at 4°C. Primary antibodies specific to MyD88, NF-κB, IκB were supplied by Proteintech (USA). Primary antibodies targeting LN, CHI3L1 were obtained from Abcam Biotech (Cambridge, UK). After washing with PBS for five times, the secondary antibodies were incubated in PBS for 2 h at room temperature. Odyssey infrared imaging system (Li cor Biosciences, Lincoln, NE, USA) was used to detect the protein band, and β-actin (1: 5000, 66009-1-Ig, proteintech) was used as the internal reference.
Statistical analysis
SPSS22.0 and GraphPad Prism 8.0.1 statistical analysis software were used for data statistics. Measurement data were expressed as mean ± standard deviation (Mean ± SD). Differences between two or more groups were analyzed using unpaired
Results
Inflammatory response and activation of TLR4-MyD88-NF-κB signaling pathway in hepatolithiasis patients
In order to determine the levels of IL-1β, IL-6, and TNF-α in peripheral blood and activation of TLR4-MyD88-NF-κB signaling pathway in hepatolithiasis patients, we performed histological and molecular biological studies in hepatolithiasis patients. ELISA results showed that the expression of IL-1β, IL-6, and TNF-α was positively correlated with the occurrence of hepatolithiasis. The levels of IL-1β, IL-6, and TNF-α in Fibrotic hepatolithiasis group were significantly higher than those in Mild hepatolithiasis group (

Inflammatory response and activation of TLR4-MyD88-NF-κB signaling pathway in hepatolithiasis patients: (a) ELISA was used to detect the levels of inflammatory cytokines (IL-1β, IL-6, and TNF-α) in peripheral blood of hepatolithiasis patients, (b) IHC detected TLR4 expression in liver tissues of hepatolithiasis patients. Bar = 25 μm (×400) or 100 μm (×100), (c) western blotting was performed to detect the expression of MyD88, NF-κB, and IκB in liver tissues of hepatolithiasis patients, and (d) *
Establishment and validation of animal model of BDL mice
It is well known that bile duct stones are the main cause of biliary obstructed.
17
In order to further verify BDL in mice, we first used ELISA to detect the levels of inflammatory factors IL-1β, IL-6, and TNF-α in the peripheral blood of mice. Compared with the Normal group and Sham group, the levels of IL-1β, IL-6, and TNF-α in Model group were significantly increased (

Establishment and validation of animal model of BDL mice: (a) ELISA was used to detected the level of inflammatory factors IL-1β, IL-6, and TNF-α in the peripheral blood of mice, (b) the liver function indexes ALT, AST, Tbil, Dbil, ALP, and GGT concentrations were detected by ELISA, (c) HE staining of liver tissue to assess pathological changes. Bar = 25 μm (×400) or 100 μm (×100), and (d) the expressions of CHI3L1 and LN were detected by western blotting.*
Activation of TLR4-MyD88-NF-κB signaling pathway in BDL mice
In order to determine the activation of TLR4-MyD88-NF-κB signaling pathway in BDL mice, the histological and molecular biological studies of liver tissues were studied in each group. IHC results showed that the levels of TLR4 between the Sham group and the Normal group had no statistical significance, but the expression of TLR4 was significantly enhanced in the Model group (Figure 3(a)). Compared with the Normal group and Sham group, the expression levels of MyD88 and NF-κB increased significantly (

Activation of TLR4-MyD88-NF-κB signaling pathway in BDL mice: (a) IHC was used to detected the level of TLR4 in liver tissue of mice. Bar = 25 μm (×400) or 100 μm (×100), (b) western blotting detected the level of MyD88, NF-κB and IκB, and (c) *
Establishment and validation of animal model of BDL in TLR4 knockout mice
First, we established an animal experimental model of BDL in TLR4 knockout mice to observe the inflammatory response and the progression of liver injury and fibrosis. ELISA results showed that the levels of IL-1β, IL-6, and TNF-α in BDL mice were significantly down-regulated after TRL4 knockout (Figure 4(a)), indicating that TRL4 knockout could alleviate the occurrence of inflammatory response. Meanwhile, the concentrations of liver function indexes ALT, AST, Tbil, Dbil, ALP, and GGT were also significantly down-regulated (Figure 4(b)), indicating that TLR4 knockout could reduce liver injury. After TLR4 knockout, the degree of liver fibrosis in BDL mice was decreased (Figure 4(c)) and the levels of liver fibrosis indexes CHI3L1 and LN were significantly decreased (Figure 4(d) and (e)), indicating that TLR4 knockout could alleviate the progression of liver fibrosis in BDL mice.
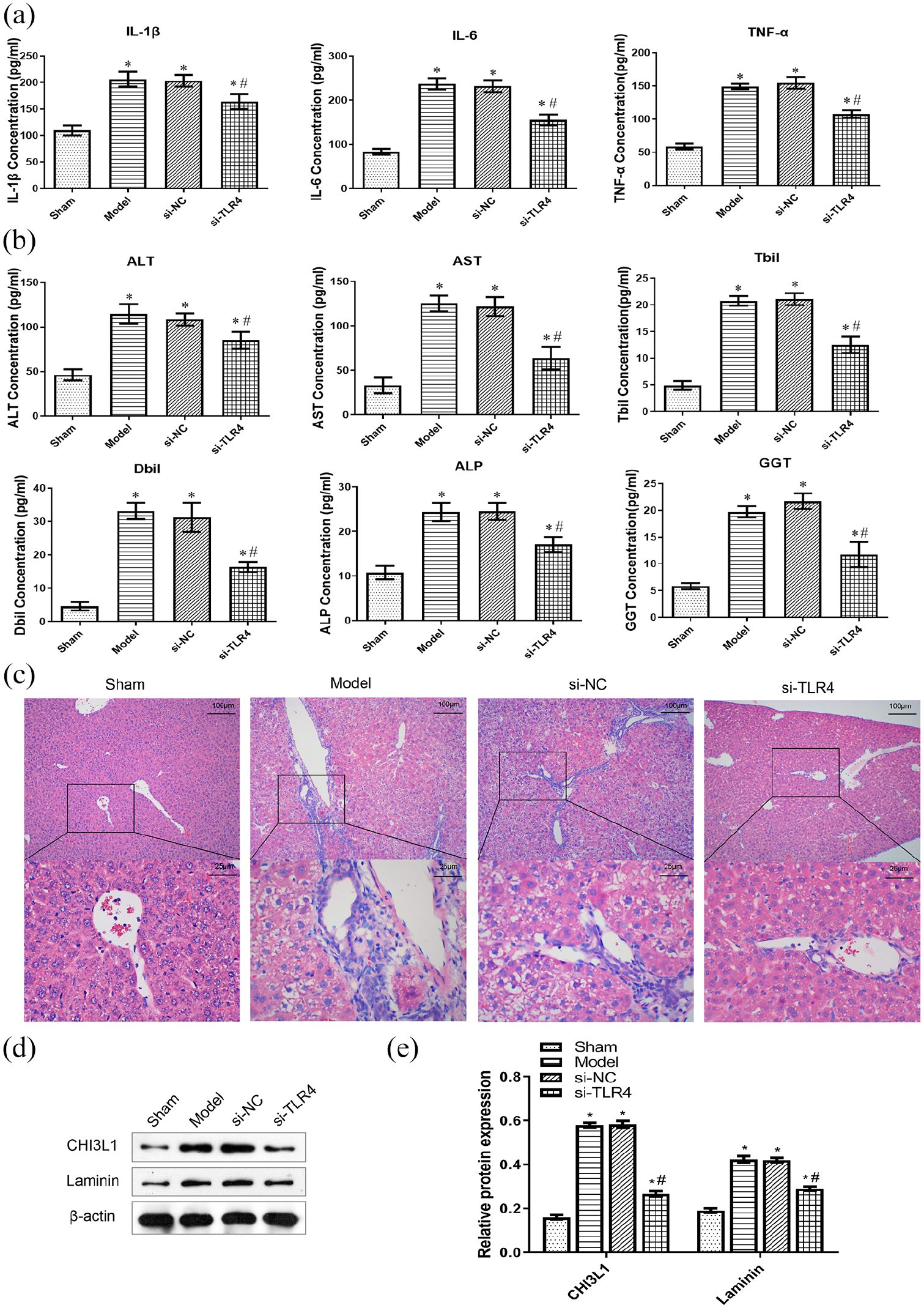
Establishment and validation of animal model of BDL in TLR4 knockout mice: (a) ELISA was used to detect the levels of inflammatory factors IL-1β, IL-6, and TNF-α in the peripheral blood of mice, (b) The liver function indexes ALT, AST, Tbil, Dbil, ALP, and GGT concentrations were detected by ELISA, (c) HE staining of liver tissue was used to evaluate the pathological changes. Bar = 25 μm (×400) or 100 μm (×100), (d) western blotting detected the levels of CHI3L1 and LN, and (e) the relative protein expressions of CHI3L1 and LN. *
Inhibition of TLR4-MyD88-NF-κB signaling pathway of BDL in TLR4 knockout mice
After knockout of TLR4 in BDL mice, the expression of TLR4 was significantly decreased (Figure 5(a)). Meanwhile, after TLR4 knockout in BDL mice, the expression of TLR4-MyD88-NF-κB signaling pathway related proteins MyD88 and NF-κB were significantly down-regulated, while IκB was up-regulated (Figure 5(b) and (c)). These results indicated that TLR4 knockout could effectively inhibit TLR4-MyD88-NF-κB signaling pathway.

Inhibition of TLR4-MyD88-NF-κB signaling pathway of BDL in TLR4 knockout mice: (a) IHC was used for detecting the levels of TLR4. Bar = 25 μm (×400) or 100 μm (×100), (b) western blotting detected the levels of MyD88, NF-κB and IκB, and (c) the relative protein expressions of MyD88, NF-κB and IκB. *
Discussion
In this study, we founded that the activation of TLR4-MyD88-NF-κB signaling pathway could promote the progression of secondary hepatic injury and fibrosis in hepatolithiasis. However, TLR4 knockout slowed down the progression of secondary hepatic injury and fibrosis in hepatolithiasis.
Hepatolithiasis was closely related to cholangitis and liver fibrosis.18,19 Studies have shown that the secondary hepatic injury in hepatolithiasis to liver function was closely related to the inflammatory response and liver fibrosis was caused by the release of a large number of inflammatory factors.20,21 It was reported that cell death and inflammation were two central elements in the development of liver fibrosis. 22 Interestingly, TLR4-MyD88-NF-κB signaling pathway was a classical inflammatory pathway and also played a vital role in the progression of liver fibrosis. We found that hepatolithiasis was associated with the occurrence of inflammatory response, and then up-regulated the levels of IL-1β, IL-6, and TNF-α. Meanwhile, the expression of TLR4, MyD88, and NF-κB were significantly increased, which indicated that the TLR4-MyD88-NF-κB signaling pathway was activated.
Activation of TLR4 could transduced transmembrane signaling through MyD88-dependent pathway, activated transcription factors such as NF-κB, and promoted subsequent release of multiple cytokines and inflammatory factors.20,23 Studies have shown that intrahepatic bile duct stones can cause recurrent inflammation. 24 IL-1β, IL-6, and TNF-α are important inflammatory factors in the body, which are closely related to the occurrence and development of diseases.9,25,26 As found in this study, the levels of IL-1β, IL-6, and TNF-α in hepatolithiasis patients were significantly up-regulated with the inflammatory state. Meanwhile, we observed significant upregulation of TLR4, MyD88, and NF-κB, suggesting that TLR4-MyD88-NF-κB signaling pathway was activated in hepatolithiasis patients. These showed that TLR4-MyD88-NF-κB signaling pathway was able to aggravate the inflammatory response and liver injury in hepatolithiasis patients.
TLR4 activated NF-κB by binding with corresponding ligands, which further promoted the up regulation of various inflammatory factors. 27 Study found that Forsythiae Fructuse water extract could inhibit the expression of inflammatory factors and fibrotic cytokines, reduce liver injury, and inhibit the development of liver fibrosis through TLR4/MyD88/NF-κB and TGF-β/smads signaling pathways. 28 Thus, in order to further elucidate the effect of TLR4-MyD88-NF-κB signaling pathway in the progression of secondary hepatic injury and fibrosis in hepatolithiasis, an animal model of BDL was established. What interesting is that the results in the animal model were consistent with those in clinical trials. Serum biochemical values ALT, AST, Tbil, Dbil, ALP, and GGT are important indexes to evaluate whether liver function is normal or not.29–32 Our study found that the liver function indexes ALT, AST, Tbil, Dbil, ALP, and GGT were significantly up-regulated in BDL mice, indicating that hepatolithiasis promoted liver injury. It is well known that CHI3L1 and LN are important indicators for the evaluation of liver fibrosis.33,34 CHI3L1 was an abundantly expressed liver gene, whose expression was highly enriched in the liver. 35 Our study found that liver fibrosis indexes CHI3L1 and LN were significantly up-regulated in hepatolithiasis patients, indicating that hepatolithiasis can cause liver injury and then promote the progression of liver fibrosis. Based on these results, we hypothesized that TLR4-MyD88-NF-κB signaling pathway may be involved in the progression of secondary hepatic injury and fibrosis in hepatolithiasis. Therefore, we established an animal model of BDL mice to evaluate the biological role of TLR4-MyD88-NF-κB signaling pathway in the progression of secondary hepatic injury and fibrosis. We found that increased levels of inflammatory factors IL-1β, IL-6, and TNF-α in BDL mice promoted liver injury and fibrosis through the TLR4-MyD88-NF-κB signaling pathway. TLR4-MyD88-NF-κB signaling pathway can promote secondary hepatic injury and fibrosis progression. To reduce the progression of liver injury and fibrosis, blocking the TLR4-MyD88-NF-κB signaling pathway may be considered.
It was reported that TLR4 silence could decrease inflammatory cytokines in corneal epithelial cells treated with inactivate staphylococcus epidermidis. 36 Blocking TLR4/MyD88/NF-κB signaling pathway may help reduce myocardial injury and improve cardiac function after coronary microembolization. 11 In order to confirm the hypothesis that blocking TLR4-MyD88-NF-κB signaling pathway could alleviate liver injury and fibrosis progression, we established an animal model of TLR4 knockout. 37 We studied the activation of TLR4-MyD88-NF-κB signaling pathway after hepatolithiasis, which is involved in liver inflammatory response, leading to liver injury and fibrosis. TLR4 knockout could reduce the level of IL-1β, IL-6, and TNF-α, which inhibited TLR4-MyD88-NF-κB signaling pathway. This significantly reduced liver injury and fibrosis caused by hepatolithiasis and improved liver function. However, this study was limited to a small number of observation time points and small sample size, and would be further improved in the future.
Conclusion
Activation of TLR4-MyD88-NF-κB signaling pathway may promote the inflammatory response and aggravate the progression of secondary hepatic injury and fibrosis in hepatolithiasis. Therefore, inhibition the TLR4-MyD88-NF-κB signaling pathway was one of the effective ways to improve liver function and slow down the secondary hepatic injury and fibrosis caused by hepatolithiasis. It provided new insights for the prevention and treatment of secondary hepatic injury and fibrosis in hepatolithiasis.
Footnotes
Acknowledgements
The authors gratefully acknowledge the financial supports by the General items of Hunan Provincial Health Commission (No. 20200554), the General items of Education Department of Hunan Province (NO.19C1120) and the General items of Science and Technology Bureau of Changsha City (No. kq1907066).
Author contributions
Yuan Fang and Lulu Zhou performed the experiment, analyzed the data, and wrote original draft; Jinmao Liao and Jingyun Guo performed the experiment; Zheng Zhang guided the experiment, reviewed, and edited the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the General items of Hunan Provincial Health Commission (No. 20200554), the General items of Education Department of Hunan Province (No. 19C1120) and the General items of Science and Technology Bureau of Changsha City (No. kq1907066).
Ethics approval
Ethical approval for this study was obtained from the Animal Ethics Committee of Hunan Provincial People’s Hospital (No. 2019. Wei. 74) and the Medical Ethics Committee of Hunan Provincial People’s Hospital (No. 2019. 74).
Informed consent
Written informed consent was obtained from all subjects before the study.
Trial registration
We have obtained the written informed consent of the subjects before the start of the study, and obtained the approval of the Medical Ethics Committee of Hunan Provincial People’s Hospital (No. 2019. 74).
Availability of data and materials
The authors confirm that all data underlying the findings are available. All relevant data are within the paper and its Supporting Information files.
