Abstract
We aimed to investigate the prevalence of the JAK2 V617F mutation in Tunisian patients with myeloproliferative neoplasms (MPN) and to look for possible associations with diseases’ presentation. In this context, JAK2 V617F polymorphism was detected by PCR-RFLP and direct sequencing in 213 MPN patients (109 with polycythemia vera (PV), 93 with essential thrombocythemia (ET) and 11 with primary myelofibrosis (PMF)), 77 unclassified patients with thrombosis (UPT) and 95 healthy control subjects. The JAK2 V617F mutant allele was present by either PCR-RFLP or direct sequencing in 158 (74.17%) MPN patients while all UPT and controls were negative. Besides, the JAK2 V617F mutation was significantly more frequent in patients with PV 98 (89.9%) than in ET 54 (58.1%) and PMF 6 (54.5%) groups,
Introduction
Dysregulated proliferation and expansion arising from a transformed pluripotent hematopoietic stem cell could lead to clonal hematopoietic malignancies, called myeloproliferative neoplasms (MPNs). 1 In Philadelphia-negative MPN group there are three entities characterized by different clinical features: Polycythemia Vera (PV), Essential Thrombocythemia (ET), and Primary MyeloFibrosis (PMF). 1
The molecular characterization of MPNs was revolutionized in 2005 by the detection of a somatic mutation, the JAK2 V617F. 1 In fact, this peculiar mutation has been detected mostly in patients with PV (65%–97%) and more rarely in patients with ET (23%–57%) and PMF (35%–57%).2–6 Few years later, in 2008, a revised edition for the classification and diagnosis criteria of MPNs including the JAK2 V617F mutation as major criteria have been published by the World Health Organization (WHO). 7
Janus kinases (JAK) are a group of tyrosine kinases enzymes which play crucial roles in cytokines signal transduction controlling cell proliferation, survival, and apoptosis. 8 In this group, JAK1, JAK2, JAK3, and tyrosine kinase 2 (TYK2) are the major kinases. 8 JAK2, a cytoplasmic kinase, plays a critical role in multiple cytokines and hematopoietic growth-factors receptors signal transduction. 1 JAK2 structure includes four domains: N-terminal FERM domain which mediates receptor interaction, a Src homology-2 domain (SH2) which interacts with several signaling proteins, a C-terminal tyrosine kinase domain JH1 (Jak homology-1) which is negatively regulated by a pseudokinase domain (JH2). 1 The JAK2 V617F mutation consists in a G to T substitution at nucleotide 1849 of exon14. 3 This gain of function mutation results in a substitution in codon 617 from valine to phenylalanine of the protein. 3 The mutation occurs in the auto-inhibitory domain (JH2) and induces a constant hyperactivation of the JAK2 protein which enhances cytokine-independent growth of hematopoietic progenitors and eventually increases cell proliferation and resistance to apoptosis. 1
The discovery of the JAK2 V617F mutation has made a significant contribution to the understanding of the pathogenesis of MPNs and its high frequency makes it an important laboratory tool for both the diagnosis and the management of these diseases. Moreover, several clinical studies have quantified the V617F mutant allele, known as ‘allele burden’. The allele burden was disparately estimated within MPN, in fact, Allele burden was the lowest in ET patients, intermediate in those with PV and PMF, and the highest in post-PV myelofibrosis patients. 9
Therefore, we aimed in the present study to determine for the first time in Tunisia the frequency of the JAK2 V617F mutation in Tunisian patients with myeloproliferative neoplasms by performing PCR-RFLP and direct sequencing methods. We intended to compare these two methods because of the reported discrepancies in the threshold of the mutation’s detection. In fact, the direct sequencing is able to detect the mutation at an allelic ratio between 10% and 20% while the threshold for PCR-RFLP was estimated at 2%–10%. 10
Material and methods
Subjects
In this work we intended to perform a transversal comparative study between PCR-RFLF and direct sequencing for JAK2 V617F mutation. This study included 213 MPN patients, 77 unclassified patients with thrombosis (UPT) and 95 healthy blood donors who served as controls. Theoretical percentage (64.4%) of the JAK2 mutation in Tunisian MPN patients was obtained from the study of Mahjoub et al. 11 We used the following online calculator to determine the sample size: https://biostatgv.sentiweb.fr/>:?module=etudes/sujets#. For a Beta risk of 0.2, the estimated required sample size is 182 subjects.
The 213 MPN patients were followed-up at the departments of hematology and internal medicine between 2016 and 2019. MPN patients were classified into three subgroups: 109 with PV, 93 with ET, and 11 with PMF. Inclusion conditions were patients meeting the 2008 WHO classification criteria for MPN. 7 Unfortunately, and because of limited resources we were not able to investigate the JAK2 exon 12, Calreticulin (CALR), and MPL mutations to apply the 2016 WHO classification criteria for MPN. 12 Exclusion criteria were: BCR-ABL1 positive karyotype and bone marrow non-conclusive findings.
We recruited an UPT group because thrombosis is a frequent and major complication in MPN. In the UPT group, thrombotic events included acute limb ischemia, stroke, splanchnic thrombosis, etc.
Healthy controls were used to verify the specificity of the JAK2 V617F mutation detection in MPN diagnosis as the mutation has been noted in the general population in some studies. In fact, Cordua et al. 13 reported a prevalence of 3.1% in a danish general population which included 19958 healthy participants.
In order to be included in this study, a written informed consent was obtained from all patients and controls. This study was approved by the local Ethics’ committee of Charles Nicolle Hospital.
Methods
A standard salting-out procedure 14 was used to extract genomic DNA for patients and controls from EDTA peripheral blood samples. The detection of the JAK2 V617F mutation was performed by both RFLP-PCR and direct sequencing procedures.
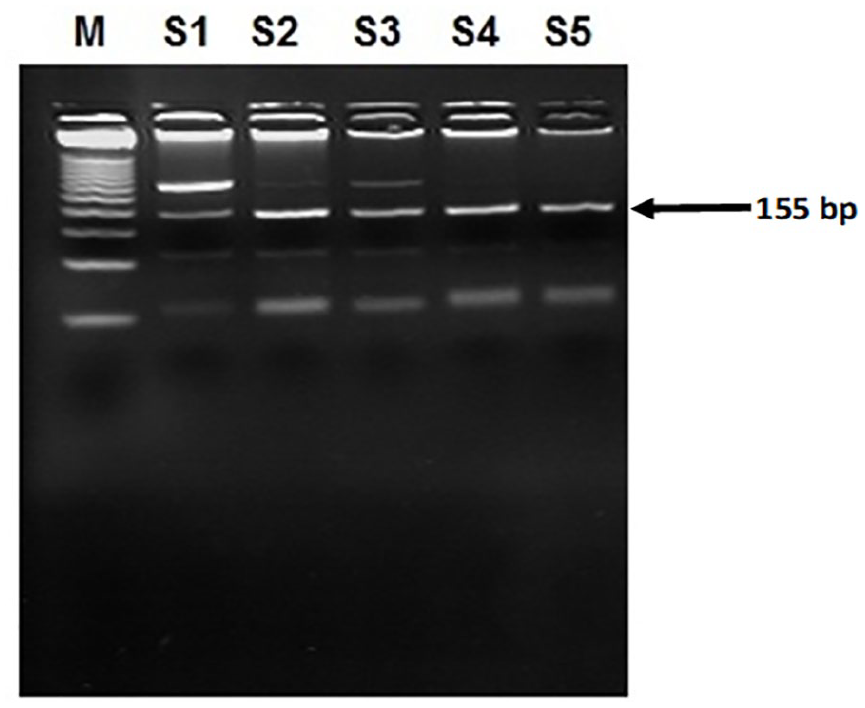
Gel analysis for the JAK2 V617F mutation detection in genomic DNA by PCR-RFLP.
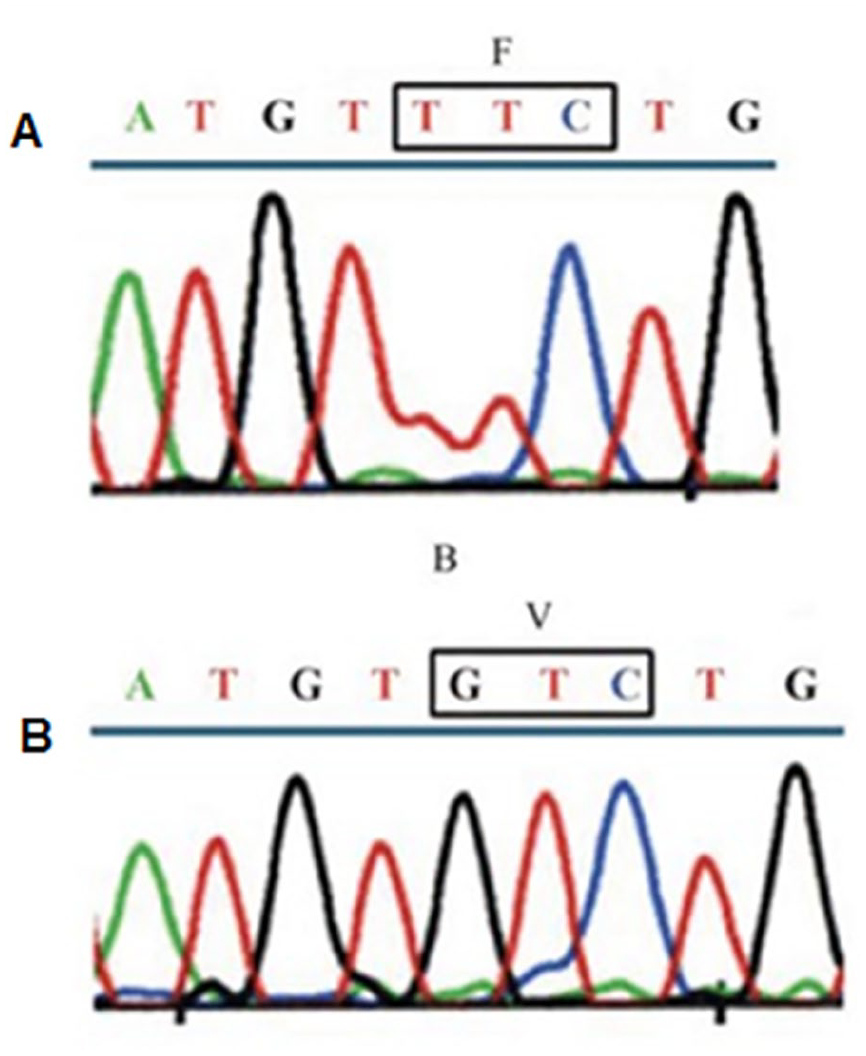
DNA sequence chromatograms of the JAK2V617F mutation.
Statistical analysis
Statistical evaluation was carried out using the Statistical Package for the Social Sciences (SPSS) version 11 (IBM®, Armonk, USA). The threshold of significance was fixed at 0.05.
Chi-square or Fisher exact tests were used to test the association between categorical variables. Odds ratio (OR) together with 95% confidence intervals (95%CI) were calculated to estimate the strength of the association.
ANOVA, Mann–Whitney
Results
In total, 213 patients with MPN, 77 unclassified patients with thrombosis and 95 controls were included in this study. Mean age was at 57.28 ± 13.9 years for MPN patients, 47.82 ± 15.01 years for the UPT group and 53.19 ± 14.51 years for the controls. The sex ratio (Men/Women) was 0.95 (104/109) for the MPN group, 1.48 (46/31) for the UPT group and 0.93 (41/44) for the controls group (Table 1). Twenty-five (11.7%) MPN patients had a portal vein thrombosis which caused a Budd-Chiari Syndrome.
Characteristics of the study population.
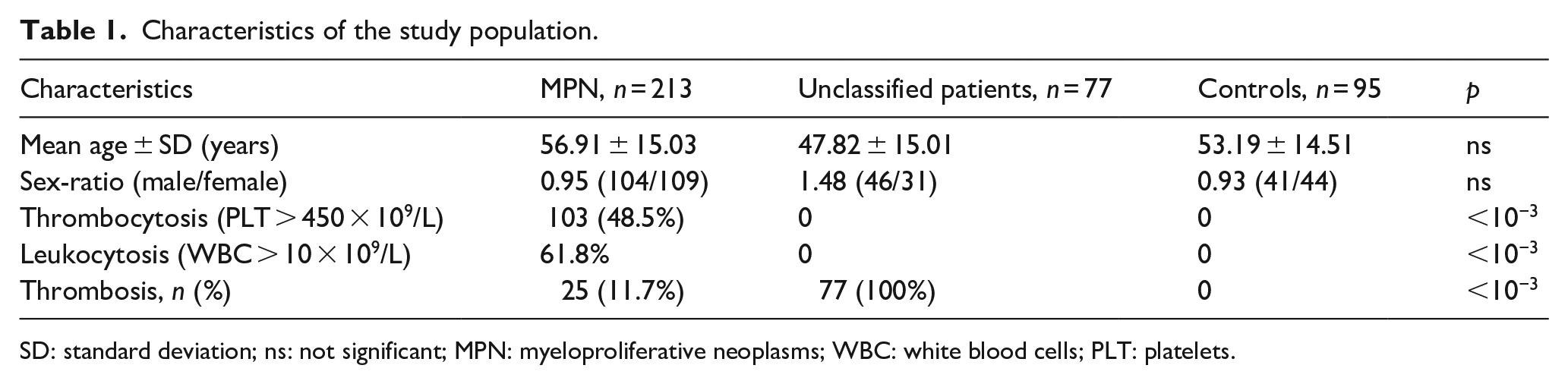
SD: standard deviation; ns: not significant; MPN: myeloproliferative neoplasms; WBC: white blood cells; PLT: platelets.
As shown in Table 2, in the MPN group, the mean age was similar in PV, ET, and PMF patients. Inversely, the sex-ratio (Male/Female) was significantly higher in PV group (1.53) than in ET and PMF groups (0.63 and 0.22),
Hematological features of the MPN patients.

PV: polycythemia vera; ET: essential thrombocythemia; PMF: primary myelofibrosis; Hb: hemoglobin; HCT: hematocrit; WBC: white blood cell; PLT: platelets; ns: not significant.
Results of the JAK2 genotyping
The JAK2 V617F mutant allele was positive in 154 (40%) subjects by PCR-RFLP and in 150 (38.96%) subjects by direct sequencing. The concordance rate between the two procedures was at 96.88% (373/385), κ = 0.935,
Comparative analysis between PCR-RFLP and direct sequencing in study population.

κ = 0.935,
Considering positive for the JAK2 V617F SNP any detected mutation by either PCR-RFLP or direct sequencing, 158 (74.17%) MPN patients were positive for V617F mutation while all unclassified patients with thrombosis and controls were negative (Table 4). Moreover, the JAK2 V617F mutation was significantly more frequent in patients with PV 98 (89.9%) than in ET 54 (58.1%) and PMF 6 (54.5%) groups,
Jak2 V617F mutant allele frequencies by PCR-RFLF and direct sequencing.

Analytic results of the JAK2 V617F mutation in MPN patients
In MPN patients, the V617F mutant allele was significantly correlated to patients’ age (59.35 ± 13.64 vs 49.91 ± 16.69),
Analysis of the Jak2 V617F mutation with clinical and biological features of MPN.

In PV and PMF subgroups, the JAK2 V617F polymorphism was not associated to either hemoglobin level or hematocrit or leukocytes and platelets counts or the occurrence of thrombosis (Table 6). Inversely, in the ET subgroup the V617F mutant allele was associated to a higher hematocrit level (39.3 ± 3.34 vs 35.78 ± 4.1) and platelet count (872 vs 719);
Analysis of the Jak2 V617F mutation with clinical and biological features of PV, ET, and PMF.

Discussion
The discovery of a gain-of-function somatic mutation in the JAK2 gene, baptized JAK2 V617F, has opened a new era in the understanding of Philadelphia-negative (PN) MPNs.
9
Currently, the JAK2 V617F mutation screening has become a routine diagnostic workup for patients suspected to have PN-MPN. Thus, we aimed to study the prevalence of the JAK2 V617F mutation in MPN patients (
The V617F mutation in the exon 14 of the JAK2 gene was detected by two molecular procedures: PCR-RFLP and direct sequencing. The PCR-RFLP was positive in 154 samples, while the direct sequencing detected only 150 positive patients for the V617F mutation. The concordance rate between the two tests was 96.88% (373/385), κ = 0.935. This, slightly higher sensitivity of PCR-RFLP comparatively to direct sequencing in JAK2 V617F mutation detecting corroborates previous reports. In fact, it has been reported that PCR-RFLP is able to detect the mutation with an allele burden of 2%–10% while direct sequencing with a burden varying between 10% and 20%. 10
In the present study, the JAK2 V617F mutation was detected in 158 (74.17%) MPN patients, while all the UPT and controls were negative. This finding corroborates the results of a Portuguese study 16 in which 75% of MPN patients and none of the controls were positive for JAK2 V617F mutation. Interestingly, the JAK2 V617F have been detected in cohorts of healthy subjects. Using qPCR, Nielsen et al. 17 found the JAK2 V617F prevalence to be 0.1% in a Danish general population. Another study in a Danish general population reported the V617F mutation 613 (3.1%) out of 19958 healthy subjects. 13 The authors explained their finding by MPN underdiagnosis in the general population. 13 Besides, in our study, the V617F mutation was present in 98 (89.9%) PV patients, 54 (58.1%) ET patients, and 6 (54.5%) PMF patients, which are higher frequencies comparatively to those reported by the first five JAK2 V617F-discovering studies in 2005.2–6 In fact, pooled data from the five studies indicated that the mutation was found in 77% (335/434) of PV, 35% (96/280) of ET, and 43% (40/93) of PMF patients. 1 These discrepancies could be explained by different diagnostic criteria which have been updated in 2008 7 and 2016. 12 Inversely, recent studies performed in patients diagnosed with the updated criteria have reported higher frequencies of the JAK2 V617F mutation in MPN patients.16,18–21 In fact, the frequency of the JAK2 V617F mutation in PV was 100% in Lebanese 19 and Iranian, 20 87.2% in Portuguese, 16 82% in Indian, 21 and 81% in Egyptian 18 populations. For the ET patients, the prevalence of the mutant allele was 73.4% in Portuguese, 16 70% in Indian, 21 68.29% in Lebanese, 19 50% in Egyptian, 18 and 45.66% in Iranian 20 studies. Besides, the JAK2 V617F mutation frequencies in PMF patients were 52% in Indian, 21 50% in Portuguese 16 and Egyptian, 18 and 39.7% in Iranian 20 reports. The differences between the reported mutation frequencies in PV, ET, and PMF could be due to diverse study designs and different genetic backgrounds.
In this study, we found that MPN JAK2 V617F-positive patients had significantly higher hemoglobin levels and hematocrit which corroborates the results of previous reports.18,20,21 Inversely, in our PV and PMF subgroups, the JAK2 V617F mutation did not influence either hemoglobin levels, or hematocrit, or leukocytes, and platelets count. These results agree with those of the Iranian 20 and Indian 21 studies. Inversely, the Egyptian report 18 noted significant associations with higher leucocytes and platelets counts in V617F positive PV and PMF patients, but the differences were not relevant (leukocytes: 13.15 vs 14.34 and platelets: 508 vs 591.2). Besides, we noted in the present study that ET JAK2 V617F-positive patients had significantly higher hematocrit and platelets count which has not been reported before in previous studies. In fact, while most of the reports16,20,21 had not found any associations, the Egyptian report noted higher hemoglobin levels but lower platelets count in ET patients carrying the JAK2 V617F mutation 18 The Egyptian unique finding 18 led the authors to suggest that JAK2 V617F-positive ET are more closely resembling to PV.
Besides, in the present study the ET JAK2 V617F-positive patients had significantly a higher risk (≈5.05) for developing thrombosis. This significant association between the JAK2 V617F mutation and thrombosis has also been reported in previous studies22,23 and confirmed by a meta-analysis of 17 studies 24 in which the estimated OR [95% CI] was 1.84 [1.4–2.43]. The higher risk of thrombosis in our ET JAK2 V617F-positive patients could be explained by the noted significantly higher hematocrit. Indeed, and in contrast to hematocrit, even extreme elevations in platelet counts do not contribute to whole blood viscosity. 25 Despite of the intuitive consideration that thrombocytosis increases the risk of thrombosis, most of previous reports failed to confirm it. 25 Moreover, the increased risk of thrombosis is not only the consequence of quantitative cellular variations but also qualitative changes. In fact, platelets from patients carrying the JAK2 V617F mutation exhibit biological differences such as increased P-selectin expression. 26
In summary, our findings corroborate those of previous reports with a majority of PV patients carrying the JAK2 V617F mutation, while it was present in approximately half of ET and PMF cases. Therefore, screening for the JAK2 V617F has to become a routine approach for MPN diagnosis in the Tunisian population. Nonetheless, the association with thrombosis events in ET patients needs to be replicated on independent cohorts.
In this study, there are some limitations. For instance, the PMF group was too small (
Conclusion
Based on these findings, the frequency of the JAK2 V617F mutation in Tunisian MPN patients corroborate those of previous studies. In ET patients, the V617F mutation seems to be predictive of thrombosis occurrence.
Footnotes
Acknowledgements
This study was granted and supported by the Research Laboratory in Immunology of Renal Transplantation and Immunopathology (LR03SP01), Charles Nicolle Hospital, Tunis El Manar University, Tunisia.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from the local Ethics’ committee of Charles Nicolle Hospital (Approval number: HCN_2016_21; January 07, 2016).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from all subjects before the study.
