Abstract
Background:
Treatment for interstitial pneumonia (IP) associated with collagen diseases has not been established. There is a need to elucidate the pathogenesis of IP and develop a novel therapy. We aimed to clarify the role of chemokine (C-X3-C motif) ligand 1 (CX3CL1, also known as fractalkine) in IP.
Methods:
Bleomycin (BLM) was intratracheally administered to C57BL/6 mice to induce IP. For treatment with control Ab or anti-CX3CL1 mAb, the mice were administered either Ab three times per week for 2 weeks from the day of BLM administration until euthanasia. Expressions of CX3CL1 and its unique receptor CX3CR1 in the lung tissue were examined by immunohistochemical analysis. Cellular infiltration and lung fibrosis were evaluated based on hematoxylin-eosin-staining and Sirius red staining of the lung tissue sections, respectively. Bronchoalveolar lavage fluid (BALF) cells were analyzed by flow cytometry.
Results:
CX3CL1 and CX3CR1 were strongly expressed in the lung tissue from mice with BLM-induced IP (BLM-IP). Treatment with anti-CX3CL1 mAb did not significantly alter inflammatory cell infiltration or fibrosis in the lung tissue. However, the number of M1-like macrophages in BALF was decreased and surface CD3 expression on T cells was increased by anti-CX3CL1 mAb treatment.
Conclusions:
Inhibition of CX3CL1 decreased inflammatory cells and may attenuate T cell activation in BALF. CX3CL1 inhibitor may have the potential to suppress the infiltration and activation of immune cells in IP.
Introduction
Interstitial pneumonia (IP) is a consequence of the loss of alveolar capillary barrier function. Continued replacement of alveolar tissue with fibrotic lesions distorts the lung architecture, resulting in a reduction in lung volume, impaired gas exchange, and ultimately in death. 1 Pathological findings reveal the accumulation of inflammatory cells and the proliferation of fibroblasts in IP. IP is classified by its etiology, including idiopathy, infections, environmental factors, drug-induced, malignancy, and rheumatic diseases. IP associated with rheumatic diseases, including rheumatoid arthritis (RA), is progressive and has a poor prognosis, due to the lack of an established treatment strategy.2,3
Chemokines have been suggested as a therapeutic target molecule for RA. 4 Previously, our group demonstrated their involvement in the pathogenesis of RA and the therapeutic effects of the blockade of several chemokines in a murine model of arthritis.5–8 We showed that chemokine (C-X3-C motif) ligand 1 (CX3CL1, also known as fractalkine) and CX3C chemokine receptor 1 (CX3CR1), a receptor for CX3CL1, were strongly expressed in the synovial tissue of RA, and that an anti-CX3CL1 monoclonal antibody (mAb) ameliorated arthritis in a murine model.5,9 Currently, humanized anti-human CX3CL1 mAb is undergoing clinical trials for RA. The findings of a phase I/IIa study revealed that this treatment was tolerated well, suggesting is clinical efficacy. 10
We also reported that CX3CL1 was expressed in IP associated with polymyositis/dermatomyositis, and serum levels of CX3CL1 were associated with the disease activities of myositis and IP. 11 The upregulation of CX3CL1 expression in IP has also been demonstrated in patients with systemic sclerosis. 12 These findings indicated that the inhibition of CX3CL1 has the potential to be effective for IP as well as arthritis in RA.
Bleomycin-induced interstitial pneumonitis (BLM-IP) is an established model of human idiopathic pulmonary fibrosis (IPF). RA-associated interstitial lung disease has some similarities to idiopathic IP including common environmental risk factors, the usual interstitial pneumonia pattern on imaging, progressive fibrosis, and a comparable prognosis. 13 In addition, MUC5B promoter variant, which is a genetic risk factor of IPF, is also associated with RA-ILD. 14 Therefore, we used a BLM-IP model to clarify the involvement of the CX3CL1-CX3CR1 in the pathogenesis of rheumatic disease-associated IP and the anti-fibrotic effects of its blockade on the diseases. We found that the inhibition of the CX3CL1-CX3CR1 did not suppress lung fibrosis in mice with BLM-IP. However, it decreased the number of immune cells and increased surface expression of CD3 on T cells in bronchoalveolar lavage fluid (BALF). Our results suggest that simply blocking CX3CL1 may not overcome lung fibrosis in BLM-IP mouse models, although it could suppress the infiltration and activation of immune cells in BALF.
Methods
Mice
Specific pathogen free (SPF) 8-week-old male C57BL/6J mice (
Histopathological and immunohistochemical analyses
The left lung was fixed in 10% formalin buffered with PBS (pH 7.2) and then embedded in paraffin. Tissues were sectioned (thickness of 3 μm) and stained with hematoxylin and eosin or Sirius red. For quantitative morphometric analysis, images of the whole area of lung sections were acquired using a BX63 microscope with the image analysis software cellSens (Olympus, Tokyo, Japan) and subsequent analyses were conducted. For evaluation of cellular infiltration, individual images were divided into four areas, and inflammatory severity was evaluated in each area, scored as follows: 0, no inflammatory cells; (1) presence of inflammatory cells in less than 20% of the area; (2) inflammatory cells in 20% to 50% of the area; (3) inflammatory cells in more than 50% of the area. The lung inflammatory score was defined as the sum of the scores in the four areas. 18 For evaluation of lung fibrosis, the Sirius red-positive area was measured by ImageJ (NIH, USA).
An immunohistochemical analysis was also performed. Sections (thickness of 3 μm) were immersed in ethanol containing 3% (v/v) H2O2 for 20 min to block endogenous peroxidase activity, and then incubated with Protein Block Serum-free (DAKO, Tokyo, Japan) for 20 min to block non-specific binding. Sections were subsequently incubated with 100 μg/mL of a rabbit anti-CX3CL1 polyclonal antibody (pAb) (BOSTER, Pleasanton, CA, USA), a rabbit anti-CX3CR1 pAb (Abcam, Cambridge, MA, USA), or isotype control (DAKO) as the primary antibody at room temperature for 3 h. Expression was detected using an EnVision+ kitTM (DAKO) and counterstained with hematoxylin.
Flow cytometric analysis in BALF
Following Fc blocking with anti-mouse CD16/CD32 (clone; 93, BD, Frankline Lakes, NJ, USA), single cell suspensions of BALF were stained with PE-conjugated rat anti-mouse CD68 mAb (clone; FA-11, BioLegend, San Diego, CA, USA), BV421-conjugated rat anti-mouse CD86 mAb (clone; GL-1, BioLegend), APC-conjugated rat anti-mouse CD206 mAb (clone; C068C2, BioLegend), BV510-conjugated rat anti-mouse CD3 mAb (clone; 17A2, BioLegend), FITC-conjugated rat anti-mouse CD19 mAb (clone; 1D3/CD19, BioLegend), APC/Cy7-conjugated rat anti-mouse CD45 mAb (clone; 30-F11, BioLegend), and PE/Cy7-conjugated rat anti-mouse CX3CR1 mAb (clone; SA011F11, BioLegend). Flow cytometric analysis was performed by BD LSRFortessa® (BD) and data analysis was conducted using FlowJo software (BD).
Statistical analysis
Statistical analyses were performed using Prism ver. 7.0 software (Graphpad Software, San Diego, CA, USA). The Kruskal-Wallis test was used for the three-group comparison and the Mann-Whitney test was used for the two-group comparison to assess differences between continuous variables with non-parametric distribution. A Student’s t-test was used for the two-group comparison to assess differences with parametric distribution. P-values less than 0.05 were considered significant. All data were expressed as the mean±standard error (SEM).
Results
BLM-IP
After the treatment of C57BL/6J mice with an intratracheal BLM injection on day 0, we resected the lung on days 14 and 21. Histological sections were examined under hematoxylin and eosin staining. We confirmed that cellular infiltration and lung fibrosis were observed in BLM-instilled mice on both days 14 and 21 (Figure 1(b) and (c)), whereas mice without BLM instillation showed no infiltration and normal alveolar structure (Figure 1(a)). However, the severity of inflammation was slightly reduced on day 21 due to the self-limited diseases in this model (Figure 1(c)). Therefore, we decided to evaluate the progression of IP by BLM on day 14.

Histopathological findings of BLM-IP. C57BL/6J male mice of 8 weeks of age were intratracheally administered with BLM. The left lung was dissected and stained with hematoxylin and eosin on indicated days (
Increase in the expressions of CX3CL1 and CX3CR1 in the BLM-IP lung tissue
Based on increased expressions of CX3CL1 and CX3CR1 in the lung tissue from patients with IP associated with SSc or polymyositis/dermatomyositis,11,19 we immunohistochemically investigated the expression of CX3CL1 and CX3CR1 in lung tissues harvested from normal mice and BLM-IP mice. CX3CL1 was expressed on column-shaped type 2 alveolar cells in normal mice (Figure 2(a)). In BLM-IP mice, mononuclear cells infiltrated in the interstitial space expressed CX3CL1 in addition to the type 2 pneumocytes (Figure 2(c)).

Immunohistochemical analysis of CX3CL1 expression in the lung tissue from mice with BLM-IP. The lung tissue from normal mice (a and b) or mice on 14 days post BLM treatment (c and d) was stained with anti-CX3CL1 pAb (a and c) or rabbit IgG (b and d). Scale bars indicate 50 µm. Original magnification is ×100. A representative section from 5 mice per each group is shown.
The CX3CR1 was also expressed on interstitial infiltrating cells, alveolar macrophages and epithelial cells in BLM-IP mice (Figure 3(c)). However, the expression of CX3CR1 was minimal in the lungs of normal mice (Figure 3(a)). These results are consistent with other studies20,21 and suggest that an interaction between CX3CL1 and CX3CR1 plays a role in the pathogenesis of BLM-IP.

Immunohistochemical analysis of CX3CR1 expression in the lung tissue from mice with BLM-IP. The lung tissue from normal mice (a and b) or mice on 14 days post BLM treatment (c and d) was stained with anti-CX3CR1 pAb (a and c) or rabbit IgG (b and d). Scale bars indicate 50 µm. Original magnification is ×100. A representative section from 5 mice per each group is shown.
Little effect of the inhibition of CX3CL1 on the progression of cellular infiltration and lung fibrosis in BLM-IP
Therefore, we conducted anti-CX3CL1 mAb treatment for BLM-IP in mice. C57BL/6J mice were administered anti-CX3CL1 mAb three times per week for 2 weeks from a day on which IP was induced by the administration of BLM. On day 14, cellular infiltration was evaluated as a histological score. We found no significant differences in cellular infiltration scores between control Ab- and anti-CX3CL1 mAb-treated mice (Figure 4(a) and (b)). We also assessed its effect on lung fibrosis by quantitative morphometric analysis of Sirius red-stained lung tissue sections. There was no significant difference in the collagen-stained area of the lung tissue between control Ab- and anti-CX3CL1 mAb-treated mice with BLM-IP (Figure 5(a) and (b)).

Anti-CX3CL1 mAb treatment has little impact on cellular infiltration in the lung in BLM-IP. Murine BLM-IP was treated with control Ab (
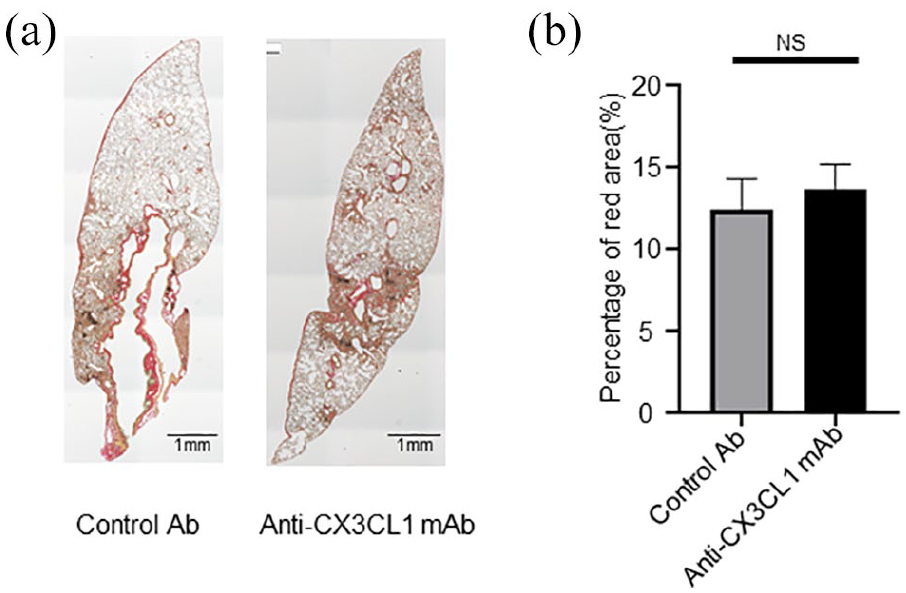
Anti-CX3CL1 mAb treatment has minor impact on tissue fibrosis in the lung in BLM-IP. The lung tissue sections from mice with BLM-IP in Figure 4 was stained with Sirius red (a). Red-colored area and total cross-sectional area was quantitated and calculated the percentage of red area in total area (b). NS: not significant. Original magnification is ×40 for (a).
Decrease of inflammatory cells in BALF by the inhibition of CX3CL1
BAL is a safe and effective means for obtaining inflammatory cells from the lower respiratory tract and the cell number and components in BALF reflect the level of inflammation in lung diseases. 22 The total number of both cells and leukocytes in the BALF obtained from mice with BLM-IP were significantly increased compared to normal mice that were not administered BLM (Figure 6(b) and (c)), indicating that BLM-IP induced the infiltration of immune cells in BALF. Interestingly, treatment with anti-CX3CL1 mAb substantially reduced the number of total cells and leukocytes (Figure 6(a) left panels, (b) and (c)). To clarify the changes in cell components by treatment with anti-CX3CL1 mAb, we further examined individual subsets of lymphocytes and macrophages and compared control Ab- and anti-CX3CL1 mAb-treated BLM-IP mice. Because we observed almost no cells on cytograms of CD3+, CD19+ or CD13–CD19– cells in mice without BLM (data not shown), we conducted a comparative analysis between control Ab- and anti-CX3CL1 mAb-treated mice with BLM administration. No significant difference was observed in the number of CD3+ T cells in BALF from anti-CX3CL1 mAb-treated mice compared to that from control Ab-treated mice (Figure 6(a) middle panels and (d)). However, both the number of CD19+ B cells and non-T non-B cells decreased by anti-CX3CL1 mAb treatment, although the difference in CD19+ B cells was not statistically significant (Figure 6(a) middle panels, (e) and (f)). Interestingly, the number of CD86+ M1-like macrophages, which have a proinflammatory phenotype, was significantly lower in BALF from anti-CX3CL1 mAb-treated BLM-IP mice than from control Ab-treated BLM-IP mice (Figure 6(a) right panels and (g)). On the other hand, there was no significant difference in the number of M2-like CD206+ macrophages between control Ab- and anti-CX3CL1-treated BLM-IP mice (Figure 6(a) right panels and (h)). The surface expression level of CD3 on BALF T cells in anti-CX3CL1-mAb-treated mice was significantly increased compared to that in control Ab-treated mice (Figure 6(i)), suggesting that the activation of T cells in BLM-IP was suppressed by blocking CX3CL1. Thus, the inhibition of CX3CL1 decreased the number of M1-like macrophages and may attenuate the activation of T cells in BALF.

Inhibition of CX3CL1 decreased M1 macrophages and increased surface expression of CD3 on T cells in BALF. Flow cytometric analysis of BALF cells from mice with BLM-IP treated with control Ab- or anti-CX3CL1 mAb. (a) Representative flow cytometry scatter plots for live cells with side scatter (SSC) and CD45 axes (left panels), CD45+-gated cells with CD3 and CD19 axes (middle panels), and CD3-CD19--gated cells with CD86 and CD206 axes (right panels). (b–h) Number of total cells (b), leukocytes (CD45+ cells) (c), T (CD3+CD19-) cells (d), B (CD19+CD3-) cells (e), non-T non-B (CD3-CD19-) cells (f), CD86+M1-like cells (g) and CD206+M2-like cells (h) of pooled samples. (i) Mean fluorescence intensity of CD3 of T cells. Values represent mean ± SEM (
Discussion
In this study, we demonstrated (1) the expression of CX3CL1 and CX3CR1 was upregulated in the lung tissue in mice with BLM-IP, and (2) the inhibition of CX3CL1 reduced the number of M1-like macrophages and increased surface CD3 expression on T cells in BALF. However, (3) we found no significant effect of CX3CL1 inhibition on reductions in cellular infiltration and fibrosis in lungs with BLM-IP.
Both expressions of CX3CL1 and CX3CR1 were increased in the lung tissue in BLM-IP, suggesting that the CX3CL1-CX3CR1 was involved in the pathogenesis of IP. Our findings were consistent with other studies showing that the expression of CX3CL1 and/or CX3CR1 were upregulated in the fibrosis of many organs including lungs, liver, and kidneys.20,23–26 Particularly, a recent study identified three subsets of macrophages during BLM-IP and demonstrated that a CX3CR1+ subset of the three promoted fibroblast proliferation by secreting platelet-derived growth factor-aa. 20 This provides strong evidence of the contribution of the CX3CL1-CX3CR1 signal to lung fibrosis and its potential as a therapeutic target.
As far as we know, we are the first to attempt to treat lung fibrosis with anti-CX3CL1 mAb in a mouse BLM-IP model. Unfortunately, we found little effect of anti-mouse CX3CL1 mAb treatment on the progression of lung fibrosis in BLM-IP. In contrast, two recent studies demonstrated a critical role of the CX3CL1-CX3CR1 in lung fibrosis in a BLM-IP mouse models. One study showed that the genetic deficiency in
Ishida et al. showed no difference in the number of lung-infiltrating cells including macrophages, T cells, and granulocytes between wild type mice and
We have observed a reduced number of CD86+ M1-like alveolar macrophages and elevated surface CD3 expression on T cells in BALF. Because surface CD3 on T cells is internalized and its expression is lost upon T cell receptor/CD3 stimulation, 28 the increased level of surface CD3 expression on BALF T cells may reflect the attenuation of activation of the T cells by anti-CX3CL1 mAb treatment. In addition, anti-CX3CL1 treatment may directly alleviate the activation of macrophages, as shown in a previous report on the activation of monocytes through the CX3CL1-CX3CR1 signal. 29 Since M1 macrophages are reported to increase in BALF during the acute phase of BLM-IP, 30 anti-CX3CL1 treatment appears to prevent the infiltration of macrophages in BALF in a BLM-IP mouse model.
This study has several limitations in addressing the effects of anti-CX3CL1 treatment in ILD. First, we utilized the histological score for evaluation of cell infiltration; however, the observation is dependent on observers and is also performed based a limited number of microscopy fields. For evaluation of fibrosis, we measured Sirius red-positive area also with a limited number of microscopy fields. This may result in under- or over-estimation of the severity of cell infiltration and fibrosis due to heterogeneous distribution of cell infiltration and fibrosis in the lung in BLM-IP model. Second, we found that M1 macrophages decreased in BLM-IP mice treated with anti-CX3CL1 mAb. However, the number and component of BALF cells reflect but do not directly represent the inflammation and/or fibrosis of the lung tissue inflammation or fibrosis, indicating that suppressive effects on the lung infiltrating cells by anti-CX3CL1 mAb have not been addressed yet in this study. Finally, the murine BLM-IP model resembles human IP in histological findings and cytokine production, however, it is a self-limited disease that is different from chronic progressive human IP.
Conclusion
We demonstrated that the CX3CL1-CX3CR1 is involved in the BLM-IP model and that its inhibition prevents infiltration and the activation of BALF immune cells. Although anti-CX3CL1 mAb treatment showed little effects on the progression of lung fibrosis, this target cannot be excluded as accumulating data suggest its critical role in the pathogenesis of lung fibrosis. Therefore, further studies are needed to elucidate more precise mechanisms of how the CX3CL1-CX3CR1 contributes to the progression of lung fibrosis.
Footnotes
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Authors’ contributions
SY and TN conceived the study. SY, ShM, JN, SaM, ZY, NK, HS, YK, KH-N, NI, and TM performed the majority of the laboratory work, statistics, and data analysis. SY wrote the manuscript. JN, TI, HN, SK, and TN did a critical review of the study protocol and of the manuscript. All authors have read and approved the manuscript.
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: I have read the journal’s policy and the authors of this manuscript have the following competing interests: YK, KH, NI, and TI are employees of KAN Research Institute, which is affiliated with Eisai Co., Ltd. In addition, YK and TI have a patent COMPOSITIONS AND METHODS FOR TREATING INFLAMMATORY DISORDERS issued.
SK received research grants from Eisai Co., Ltd. TN received research grants, consulting fees and speaking fees from Eisai Co., Ltd. All other authors have declared no conflicts of interest.
Ethics approval
Ethical approval for this study was obtained from Toho University Animal Care and User Committee (No. 17-51-367 and 18-51-398).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work supported in part by a Project Research Grant (No. 17-31) from Toho University School of Medicine to SY, a Research Promotion Grant from Toho University Graduate School of Medicine (No. 17-01) to TN, the Program for the Strategic Research Foundation for Private Universities (S1411015) from the Ministry of Education, Culture, Sports, Science, and Technology, Japan to TN, and the Private University Research Branding Project from the Ministry of Education, Culture, Sports, Science, and Technology, Japan to TN. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
