Abstract
To analyse the differential indicators of COVID-19 in severe and mild cases and to study the factors affecting the immune function of patients and the time required for oropharyngeal swabs to become negative. Age, albumin (ALB) levels, prealbumin (PAB) levels, high-sensitivity C-reactive protein (hs-CRP) levels, platelet counts, lymphocyte counts, neutrophil counts, CD3+, CD4+, CD8+ T cell counts and the time for oropharyngeal swabs to become negative were collected from 37 patients with COVID-19; the neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR) were calculated as indicators of inflammation. An independent-sample t test was used to analyse differences between the severe and mild groups, and factors affecting the CD3+, CD4+ and CD8+ T cell counts and the time for the nucleic acid tests of oropharyngeal to convert to negative were identified by single-factor and multifactor analyses. Lymphocyte, ALB, PAB, CD3+, CD4+ and CD8+ T cell levels in the severe group were lower than those in the mild group, the
Introduction
Coronavirus disease 2019 (COVID-19) is an acute respiratory disease caused by a β virus, namely, severe acute respiratory coronavirus 2 (SARS-CoV-2). The World Health Organization (WHO) named the disease. In China, an autopsy was performed on a patient who died of COVID-19, and it was discovered that SARS-CoV-2 not only attacks the lung tissue and causes inflammation but also invades the immune system. Many clinical studies have also shown that patients with COVID-19 have obvious inflammatory responses, accompanied by a decrease in the absolute value of peripheral blood lymphocytes and a reduction in the numbers of T cell subsets. We collected clinical data from 37 patients with COVID-19 and analysed the differences between the severe group and the mild group. Then, we analysed the factors that affect immune function and negative oropharyngeal swab nucleic acid status to provide a reliable clinical reference for disease monitoring.
Subjects and methods
Subjects
This cross-sectional study was approved by the Ethics Committee of the Kunming Third People’s Hospital (approval No. 2020011513). All data were used only in clinical studies, and written consent was obtained from the patients or their guardians. According to the
Observed indicators
Based on existing reported variables that may affect clinical outcomes, combined with clinical findings, the final variables selected included patient age, albumin (ALB), prealbumin (PAB), high-sensitivity C-reactive protein (hs-CRP), platelet count, lymphocyte count, neutrophil count, CD3+ T cell count, CD4+ T cell count, CD8+ T cell count and time to a negative pharyngeal swab nucleic acid test (time from admission to penultimate predischarge negative oropharyngeal swab nucleic acid test). All patients admitted to the hospital underwent fasting venous blood analysis, and their routine blood parameters and levels of high-sensitivity C-reactive protein, ALB and PAB were measured. CD3+, CD4+ and CD8+ T cell counts were detected by flow cytometry. Inflammation was assessed by the NLR (neutrophil count/lymphocyte count) and PLR (platelet count/lymphocyte count).
Flow cytometry analysis
Venous blood samples (3 ml) were tested with a haematology autoanalyser (Sysmex, XG-550) to obtain the counts of the total WBCs, platelets, neutrophils and lymphocytes. Serum samples (4 ml) were tested for the levels of ALB, PAB and hs-CRP with an AU5400 automatic biochemical analyser. According to the manufacturer’s instructions, 20 μl of CD3/CD4/CD8/CD45 (FITC/PE/APC/PerCP) detection reagent (046-004831-00, Mindray, Shenzhen, China) and 50 μl of EDTA-anticoagulated venous blood were added to the bottom of the test tube and incubated at room temperature in the dark for 15 min. Then, 450 μl of flow haemolytic agent (105-004580-00, Mindray) was added, mixed gently and incubated at room temperature in the dark for 15 min. The stained cells were analysed using a flow cytometer (BD Company, New Jersey, USA), and the data were analysed with FlowJo software 2.8/3.0 (Treestar, Ashland, OR, USA).
Oropharyngeal swab viral nucleic acid detection by quantitative real-time PCR
Viral nucleic acid was extracted from the oropharyngeal swab specimens using a nucleic acid extraction kit (P732200119, Tianlong Biotechnology Co., Ltd., Suzhou, China). Nucleic acid was detected with the COVID-19 ORF1ab/N gene nucleic acid detection kit (Tianlong Biotechnology Co. Ltd., Suzhou, China). The system was prepared according to the kit instructions and placed on an ABI 7500 real-time quantitative PCR instrument for detection. The reaction procedure is as follows: 50℃ for 30 min, 95℃ for 10 min, 94℃ for 15 s, 50℃ for 30 s, 72℃ for 30 s, 94℃ for 10 s and 58℃ for 30 s, with 40 cycles. According to the cycle threshold (Ct) analysis, if the Ct values of the FAM channel and VIC channel are less than or equal to 37, and the curve is S-shaped with a significant exponential growth period, the test sample is positive; if the Ct value of one channel is ⩽ 37, the sample should be tested again. If the result is the same, then the sample is suspected of being positive. If the Ct values of both channels are > 37, and the internal standard channel test result is positive, then the test sample is negative.
Statistical processing
Measurement data are expressed as the (¯x ± s). Analysis was performed with the SPSS 22.0 statistical software package, and independent group t tests were used for comparisons between two groups.
Results
Compared with patients in the mild group, those in the severe group had significantly reduced lymphocyte counts, ALB levels, PAB levels, and CD3+, CD4+ and CD8+ T cell counts. The NLR and PLR were significantly higher in the severe group than in the mild group; there was no difference in the hs-CRP level or nucleic acid negative conversion time (Table 1). The CD3+, CD4+ and CD8+ T cell counts in all patients were measured by flow cytometry, and the data are shown as dot plots (Figure 1).
Comparison of inclusion indicators among the mild and severe groups.

hs-CRP: C-reactive protein; ALB: albumin; PAB: prealbumin; NLR: neutrophil count/lymphocyte count; PLR: platelet count/lymphocyte count.
After conducting analysis with an independent t test, lymphocytes, neutrophils, leucocytes, ALB, PAB, CD3+ absolute value, CD4+ CD3+ absolute value, CD8+ CD3+ absolute value, PLR and age were significantly different among the groups.

CD3+, CD4+ and CD8+ T cell distribution in 37 cases of COVID-19.
Single-factor analysis of factors affecting CD3+, CD4+ and CD8+ T cell counts: Among the included variables, univariate analysis showed that age, ALB level, PAB level, hs-CRP level, platelet count, the NLR, the PLR and the nucleic acid negative conversion time were the main factors that influenced CD3+ T cells (Table 2). Age, PAB level, hs-CRP level, platelet count, the NLR and the PLR were the main factors that influenced CD4+ T cells (Table 3). Age, ALB level, PAB level, hs-CRP level, platelet count, the NLR, the PLR and nucleic acid negative conversion time were the main factors that influenced CD8+ T cells (Table 4).
Single-factor analysis of factors affecting CD3+ T cells.
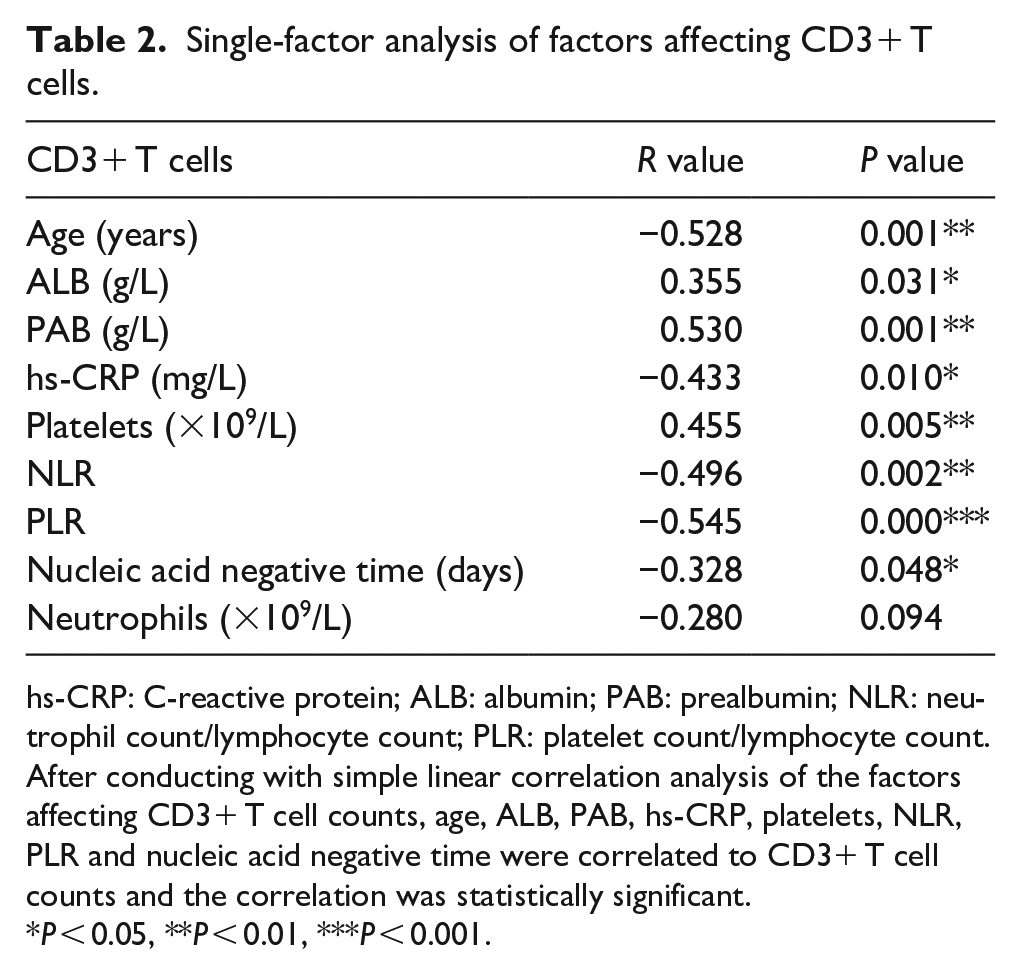
hs-CRP: C-reactive protein; ALB: albumin; PAB: prealbumin; NLR: neutrophil count/lymphocyte count; PLR: platelet count/lymphocyte count.
After conducting with simple linear correlation analysis of the factors affecting CD3+ T cell counts, age, ALB, PAB, hs-CRP, platelets, NLR, PLR and nucleic acid negative time were correlated to CD3+ T cell counts and the correlation was statistically significant.
Single-factor analysis of factors affecting CD4+ T cells.

hs-CRP: C-reactive protein; ALB: albumin; PAB: prealbumin; NLR: neutrophil count/lymphocyte count; PLR: platelet count/lymphocyte count.
After conducting with simple linear correlation analysis of the factors affecting CD4+ T cell counts, Age, PAB, hs-CRP, platelets, NLR and PLR were correlated to CD4+ T cell counts and the correlation was statistically significant.
Single-factor analysis of factors affecting CD8+ T cells.

hs-CRP: C-reactive protein; ALB: albumin; PAB: prealbumin; NLR: neutrophil count/lymphocyte count; PLR: platelet count/lymphocyte count.
After conducting with simple linear correlation analysis of the factors affecting CD8+ T cell counts, age, ALB, PAB, hs-CRP, platelets, NLR, PLR and nucleic acid negative time were correlated to CD8+ T cell counts and the correlation was statistically significant.
Among the included variables, univariate analysis showed that age, ALB level, PAB level, hs-CRP level, platelet count, the NLR, CD3+ T cell count and CD8+ T cell count were the main factors that influenced the nucleic acid negative conversion time (Table 5).
Single-factor analysis of nucleic acid negative time in oropharyngeal swabs.

hs-CRP: C-reactive protein; ALB: albumin; PAB: prealbumin; NLR: neutrophil count/lymphocyte count; PLR: platelet count/lymphocyte count.
After conducting with simple linear correlation analysis of the factors affecting nucleic acid negative time, age, ALB, PAB, hs-CRP, platelets, NLR, CD3 absolute value and CD8+ absolute value were correlated to nucleic acid negative time and the correlation was statistically significant.
Variables with
Multifactor analysis of factors affecting CD3+ T counts.

PLR: platelet count/lymphocyte count.
Factors with
Multifactor analysis of factors affecting CD4+ T counts.

PLR: platelet count/lymphocyte count.
Factors with
Multifactor analysis of factors affecting CD8+ T counts.

PLR: platelet count/lymphocyte count.
Factors with
Multifactor analysis of nucleic acid conversion in oropharyngeal swabs: The variables with
Analysis of multiple factors affecting nucleic acid conversion of oropharyngeal swabs.

Factors with
Discussion
The immune system is the most important defence against a virus. Studying the factors that affect immune changes is important for monitoring the disease and predicting prognosis. In this study, we analysed the differences in inflammation and immune function in patients with different degrees of disease severity in an attempt to identify the factors that affect the body’s defence system and to further analyse the factors that affect nucleic acid conversion. Finally, the relationship between the immune system and inflammation in COVID-19 was partly clarified.
Through this research, we found that leucocyte count, neutrophil count, and PLR were significantly higher and the lymphocyte and T cell subpopulation counts were lower in severe patients than in mild patients. Lymphocytes are important cells for the body’s immune response when inflammation occurs. 3 The decrease in the number of lymphocytes indirectly reflects immune dysfunction or suppression. 4 Existing clinical analysis and our research have shown that peripheral blood lymphocyte counts and T cell subpopulation counts are reduced in COVID-19 patients,5,6 suggesting that patients with COVID-19 experience immune dysfunction after infection with SARS-CoV-2. This dysfunction is related to the severity of the disease. Platelets serve as sentinels of the vascular system, regulating and participating in the host’s immune response. 7 In the severe group, albumin and prealbumin levels were also significantly lower in patients with severe COVID-19 than in those with mild COVID-19. The difference may be related to infection-related metabolic changes and insufficient liver synthesis, but it cannot be ruled out that SARS-CoV-2 affects the liver in addition to affecting lung tissue.
One of the main ways for the body to defend against the invasion of foreign pathogens is through lymphocyte-mediated cellular immunity. It has been reported that in community-acquired pneumonia, the lymphocyte count decreases, and severe inflammatory reactions occur at the same time. 8 The depletion of T cells is related to the severity of disease and prognosis. Based on recent autopsy findings in patients with COVID-19, 9 the number of lymphocytes is significantly reduced, immunohistochemical staining showed that CD4+ and CD8+ T cell counts were reduced in the spleen and lymph nodes, and bone marrow cells decreased. Our results are consistent with the autopsy results; T cell subset counts decreased in more than half of the patients. The PLR has been reported to predict the severity and prognosis of pneumonia. 10 Our research also shows that the PLR was a main factor influencing the T cell subsets; fighting SARS-CoV-2 exhausts the T cells, 11 and monitoring changes in platelet counts and the PLR can help reveal the body’s immune function status to a certain extent. In the current epidemic period, isolated observation areas and fever clinics are usually unable to test CD3+, CD4+ and CD8+ T cell counts. Even so, the immune status of the body can be indirectly evaluated by routinely testing venous blood to monitor platelet counts and calculate the PLR.
The breakdown of the immune balance is a turning point in disease progression. CD8+ T cells, as the foremost sentinel of the immune system, play an important role in the process of removing the virus. CD8+ T cells were important factors in the multifactorial analysis of nucleic acid negative conversion. CD8+ T cell counts are reduced in COVID-19 patients, and we consider it possible that infection with SARS-CoV-2 exerts cytotoxic effects on T cells. CD8+ T cells mobilize naive CD8+ T cells in the spleen and lymph nodes, resulting in a reduction in the number of CD8+ T cells in secondary lymphoid organs. Studies have reported that CD8+ T cells can also secrete the immunosuppressive factor IL-10 to prevent excessive inflammatory responses. 12 In addition, CD8+ T cells affect the clearing of the virus, so the role of CD8+ T cells in COVID-19 should not be ignored. The current discharge criteria in the latest edition of the COVID-19 Diagnosis and Treatment Guidelines include two consecutive negative oropharyngeal swabs that are obtained and tested more than 24 h apart. Frequent throat swab sampling will definitely increase the risk of infection for medical staff. In our study, when the level of CD8+ T cells recovered, the probability of a negative oropharyngeal swab nucleic acid test was higher. Reducing unnecessary sampling thereby reduces the chance of infection for medical staff and avoids wasting medical resources.
This study also has some limitations. This is a single-centre retrospective study, and although we included all cases that met the criteria, there were only 37 patients. Most COVID-19 patients were imported cases, with fewer severe cases of COVID-19, which may have affected the research results. Moreover, as COVID-19 is a novel infectious disease, many detection methods still have limitations. We could not ensure the accuracy of the antibody test kit, so antibody detection data were not analysed.
Conclusion
The PLR, PAB level, ALB level and T cell counts were different between severe and mild COVID-19 patients. The platelet count and PLR may affect the immune function of patients with COVID-19, and CD8+ T cells may affect the negative nucleic acid conversion time in oropharyngeal swabs.
Footnotes
Author contributions
H.-Y. F. and X.L. wrote the draft of the manuscript; H.-J.L., Y.-R.D., Y.-J.Z., X.-Q.T. and G.-P.J. participated in COVID-19 patient management; Y.L. and J.-P.G. revised the manuscript. All authors have read and approved the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from Ethics Committee of the Kunming Third People’s Hospital (approval No. 2020011513).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from all subjects before the study.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
