Abstract
Hypoxic–ischemic encephalopathy (HIE) in neonates can lead to severe chronic neurological deficit, including mental retardation, epilepsy, and sleep–wake cycle (SWC) disorder. Among these defects, little is known about the molecular mechanism of circadian rhythm disorder after HIE. Therefore, further study of sleep problems and its mechanism in HIE children will provide new ideas for clinical treatment of HIE children. For pediatric patients with cerebral ischemia, somnipathy often occurs due to visual and airway abnormalities. From May 2010 to August 2013, 128 newborns with history of HIE were followed up. Meanwhile, 88 normal full-term newborns in the same period were taken as the control group. The clinical data of the patients were collected and the sleep status was assessed by questionnaire. To establish the hypoxic–ischemic brain injury model of neonatal rats and analyze the mechanism of mir-182 in the circadian rhythm disorder caused by pineal function injury. The core clock genes during the regulation of the circadian clock were explored by bioinformatics methods. Patients’ sleep quality was affected by the circadian rhythm and respiratory problems; the pineal gland can regulate the core clock genes in the circadian clock during regulation. miR-182 was highly expressed in the pineal gland after hypoxic–ischemic brain damage (HIBD). Children with mild and moderate HIE showed significant sleep disorders in varying degrees, which provided a clinical basis for improving the long-term prognosis of children with HIE through targeted treatment of sleep disorders. MiR-182 is highly expressed in the pineal gland and is related to the expression of CLOCK protein. CLOCK gene is the target gene of miR-182, which provides a new target for the treatment of rhythm disorder related to the damage of pineal function caused by HIBD.
Introduction
Hypoxic–ischemic brain damage (HIBD) caused by hypoxic–ischemic encephalopathy (HIE) in neonates is a serious complication of perinatal asphyxia, which refers to hypoxic–ischemic brain lesions caused by neonatal hypoxia asphyxia in the perinatal period; this condition is characterized by neuropathological and pathophysiological changes and a range of clinical encephalopathy manifestations.1,2 According to statistics, the incidence of neonate asphyxiation in different regions of China is between 1.14% and 11.7%; the mortality rate of birth asphyxia of children under 5 years old reaches 221.3/100,000; and the mental disability rate is 0.9% among children aged 0–17 years, while some are caused by neonate asphyxia as the third leading cause of disability, accounting for 8.6%. 3 Among children with hearing disabilities aged 0–6 years, 6.34% are caused by neonatal asphyxia. This disease has become a major issue that jeopardizes the health and quality of life of children in China. As it is prevalent worldwide, 4 scholars at home and abroad have widely investigated this condition over many years.
The consequences of perinatal hypoxia and ischemia (HI) may vary according to the severity of the disease. Severe cases may cause death, while patients with mild symptoms may have relatively normal neurological function. 5 Although moderate-to-severe HI often causes serious motor impairments, such as cerebral pain (CP) and cognitive deficits, pediatric patients with mild HI are often found to have no obvious difference in both functions from normal and full-term neonates. However, recent studies have found that pediatric patients with mild HI have a high risk of low IQ, 6 but this is mostly mitigated due to the relatively normal motor function of these pediatric patients. In addition to cognition and dyskinesia, multiple types of neurological impairments can occur in pediatric patients with HI in the perinatal period. 7 For example, moderate-to-severe HIE can cause a delay in the initiation of the sleep–wake cycle (SWC). 8 For pediatric patients with cerebral ischemia, somnipathy often occurs due to visual and airway abnormalities. To date, the relationship between HI and biological rhythm disorder has not yet been studied by medical researchers.
To date, no systematic studies on the specific sleep disorders of pediatric patients with varying degrees of HI have been conducted. This study will expose the health hazards from hypoxic–ischemic impairment to the human body in a broad sense and provide a scientific basis for a more profound recognition of HI and its treatment.
Data and method
Ethical approval
The study was approved by the Institutional Ethics Committee of Wuhan Children’s Hospital (ethics no. 2010023), and surgical operations on all animals are approved by IACUC (approval no. 2010XS020), Wuhan Tongji Medical College Affiliated to Huazhong University of Science and Technology. Written informed consent was obtained from the legal guardian of the child before the study.
Study subjects
This is a prospective study. In this study, some neonates, who were less than 1 week, born from May 2010 to August 2013 in the Wuhan Children’s Hospital, were chosen as primary screening subjects. Pass 11th software is used to calculate the profit sample size. 9 The inclusion criteria were as follows: (1) a history of abnormal obstetric diseases that can cause fetal distress in the uterus and symptoms of fetal intrauterine distress (fetus < 100 beats/min, last for more than 5 min); those with amniotic fluid grade III-contaminated areas had a clear history of asphyxia during delivery; (2) a history of asphyxia at birth, that is, the Apgar score is 1; (3) neurological symptoms soon after birth that continue to 24 h or above, such as altered state of consciousness (excessive excitement, drowsiness), change in muscle tone (high or slightly weak), or abnormality of primitive reflexes (normal or weak embrace reflexes); and (4) diseases caused by intracranial hemorrhage and birth injuries, as well as brain injuries caused by intrauterine infection, genetic metabolic diseases, and other congenital disorders. The exclusion criteria include the following: (1) severe Hypoxic–ischemic encephalopathy (HE); (2) acquired brain injury such as trauma; (3) other major lesions, such as neurological and psychiatric disorders after birth; (4) those whose parents/siblings are unable to provide population demography data and medical information and/or complete questionnaires; (5) those who often took anticonvulsant drugs, sedatives, and neuroleptic drugs during the past few months; and (6) those who have congenital development malformation that can cause upper airway obstruction. 9 The control group includes those born in that period, 35 weeks ⩽ gestational weeks ⩽ 42 weeks, 2500 g ⩽ birth weight ⩽ 4000 g, and the metaplasia weight is between the average weight P10–P90 at gestational ages; 5-min Apgar score > 8; and the medical records and surveys in clinical practices show that the perinatal children and infants have normal development.
Study method
We collected clinical data from the children’s patients, including age, gender, gestational age, birth weight, rescue history after birth, iconography data, and diagnosis and treatment process. For mild HIBD: patients presented alternate excitement and suppression, normal or slightly increased muscle tone, active embrace reflex, normal sucking reflex, visible myoclonus, no central respiratory failure, and normal or enlarged pupil diameter; the electroencephalography (EEG) examination was normal; the clinical symptoms could disappear within 72 h, and the prognosis was good. For moderate HIE: patients showed drowsiness, low muscle tone, weak cuddle and sucking reflexes and common convulsions, visible central respiratory failure, shrinking pupils, low EEG voltage, and visible epileptiform discharge; the clinical symptoms disappeared within 14 days, and sequelae may occur.
The Sleep Questionnaire form was developed with reference to the Brief Infant Sleep Questionnaire (BISQ) 10 and the Sleep Disorders Scale for Children (SDSC). Questionnaire variables include the following: (1) unfixed start time of nightly sleep (clock time when the pediatric patients fall asleep at night); (2) longer sleep latency (sleep time after going to bed at night >20 min); (3) lack of sleep (<11 h); (4) changes in daily bedtime routine (changes in start time and duration of sleep, prolonged sleep at night); (5) loud snoring; (6) poor sleep breathing (including shortness of breath, intermittent apneas during night sleep); and (7) awakening frequently at night (>two times/night). Each variable has an option of 0–2 levels: 0, never occurred in the past 6 months; 1, occurred ⩽ three times/week in the past 6 months; and 2, occurred > three times/week in the past 6 months. All seven variables were added to obtain a sleep quality score. In addition, sleep-relevant respiratory problems included the following symptoms: marked snoring, shortness of breath, and intermittent apnea. The physical examination excluded cases with congenital development malformation, if possible, that can cause upper airway obstruction.
Establishment of animal models
Surgical operations on all animals must be approved by IACUC, the Medical College of Soochow University. The newborn HIBD animal model was built as reported by Sun et al. 14 Neonatal Sprague-Dawley (SD) animals were selected, and after 7 days, they inhaled ether for anesthesia. The animals were secured on a surgical table in a supine position, a longitudinal incision of approximately 1 cm was made in the center of the neck, and the subcutaneous tissues and muscles were separated. Then, the left common carotid artery was found and freed from between the deep side of the left sternocleidomastoid muscle and the trachea and permanently ligated with a 4-0 silk doubly, and the incision was sutured. Each animal was operated for 5–10 min at room temperature (24°C–26°C). After the surgery, the SD was placed in a 37°C incubator for 1 h and then moved into a closed, atmospheric hypoxia chamber previously placed in a 32°C incubator, where the humidified 8% nitrogen–oxygen mixture was supplied for 2 h at a flow rate of 1.5 L/min. An oxygen meter monitored the oxygen concentration. After the model was established, the SD was returned to the mother rats. For the control group, the left common carotid artery was isolated but not ligated after suture. The skin should be sutured.
SDs in the experimental group (n = 7) and the control group (n = 6) were randomized. All SDs in the same litter were randomly assigned to the following two groups and sacrificed by HIBD or 12 h after the sham operation. The Kuszak method removes the pineal gland. The animal, Rl, was prostrate on a homemade ice bench soon after ether anesthesia. The skin between the ears of the head was cut to expose the sagittal and lambdoidal sutures. The skull sagittal bone and endocranium were sheared with the sagittal suture as the vertical axis, an eyelid forceps was used to gently lift up the joint sinus and sagittal sinus veins and make them form a 45°–60° angle to the brain plane, and another eyelid forceps was used to remove the pineal gland from below the joint sinus with another eyelid, pulverize it, and place it in a 1 mL Eppendorf tube with TRIzol. In the process, the pipette was repeatedly used evenly, and then the samples were stored in a −20°C freezer.
Bioinformatic prediction of the miR-182 target gene
Three kinds of bioinformatic prediction analysis software—miRanda, TargetScan, and PicTar—were applied to retrieve the possible target gene of miR-182 and screen out the genes with a high conservation degree and low binding free energy, which are related to the regulatory system of the circadian rhythm biological clock. In this study, miRanda, TargetScan, and PicTar were employed to predict the possible target genes downstream of miR-182. The intersection of the three was taken, and we found that Clock in the clock gene family might be a potential target gene.
Real-time fluorogenic quantitative polymerase chain reaction detection of miR-182 and Clock gene expression in pineal gland tissues of an animal model after HIBD
Total RNA from pineal gland tissues of the animal model was extracted with TRIzol reagent according to the manufacturer’s instructions. An ultraviolet spectrophotometer was used to detect the total RNA concentration, and RNA was reverse transcribed into cDNA. U6 was used as the reference gene, and the sequence of the target gene was acquired from miRBase. Then, Primer 5.0 primer design software was employed to design polymerase chain reaction (PCR) primers. The target gene was amplified with SYBR Green PCR MasterMix, with a total reaction system of 20 μL. The reaction conditions are as follows: 5°C, 10 min; 95°C, 15 s; 60°C, 15 s; 72°C, 30 s, 45 cycles in total. U6 served as the standardized data for internal reference, and 2-ΔΔCt was adopted for calculation. Replicate wells were used for each experiment, and the experiment was repeated three times.
Western blot detection of Clock protein expression in pineal gland tissues of the animal model after HIBD
1 mL of lysis buffer was added to 10 μL of protease inhibitor, and the mixture was placed on ice for 30 min to fully split and decompose pineal gland tissues from the animal model. Then, it was centrifuged for 20 min at 4°C and 12,000 r/min. The supernatant was collected and quantified using the bicinchoninic acid (BCA) assay method.The protein was separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE), and the protein on the gel was electrically transferred to a nitrocellulose membrane. Next, 5% skim milk powder was used for sealing overnight, and the primary antibody was added. After incubation for 3 h at room temperature, the secondary antibody was added. Finally, electrochemiluminescence (ECL) luminous fluid was adopted for exposure and development. The experiment was repeated three times. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) protein was used as a housekeeping protein, and ImageJ image analysis software was applied to analyze the exposed strips and calculate the optical density value (OD value) of the target strip.
Statistical analysis
The data are expressed as x ± s, and statistical software SPSS 17.0 was used for statistical analysis. One-way analysis of variance (ANOVA) was employed for intergroup data comparison. P < 0.05 indicates a significant difference, and P < 0.01 indicates a very significant difference.
Results
General information of research objects
There were 16 children included in the study, 128 of whom were diagnosed with HIE, including 44 with moderate HIE and 84 with mild HIE. 88 normal children born in the same period were used as control (for details, see Table 1).
Basic information of research object (x ± s).
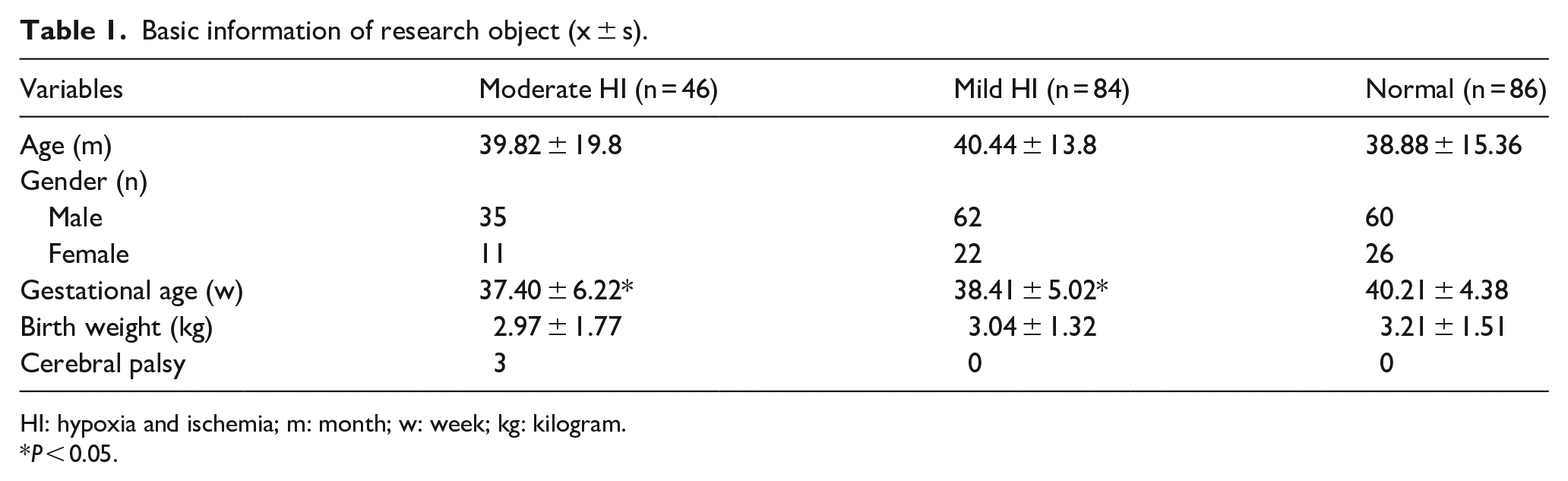
HI: hypoxia and ischemia; m: month; w: week; kg: kilogram.
P < 0.05.
Various sleep disorders of pediatric patients with HIE
Compared to pediatric patients with mild HIE, those with moderate HIE had a higher risk of breathing-relevant sleep disorders, including apparent snoring and other symptoms. These findings coincide with a previous conclusion that there is a positive correlation between the occurrence of cerebral palsy and sleep-relevant respiratory problems, 12 possibly because, compared to mild HIE, moderate HIE can cause extensive pallium and subcortical injuries, which may have a significant impact on the neuromuscular control of the upper respiratory tract. Although previous studies have suggested that mild hypothermia therapy may delay the formation of the SWC of pediatric patients with moderate-to-severe HIE, the relationship between the maintenances of HIE and SWC remains unclear. 5 Therefore, this study showed for the first time that the maintenance of regular SWCs during childhood development may also be vulnerable to the history of HI disease. Melatonin was shown to play a protective role in HIE pathophysiology (see Table 2).
Various sleep problems in children with HIE.

HI: hypoxia and ischemia; HIE: hypoxic–ischemic encephalopathy.
There was significant difference between moderate HIE group and mild HIE group.
There is significant difference between moderate HIE group and normal group.
There was significant difference between mild HIE group and normal group.
Changes in miR-182 and Clock mRNA expression in pineal gland tissues after HIBD
The miR-182 mRNA expression level in the HIE group was higher than that in the control group at the corresponding time point after HIE model setup (P < 0.05, # in Figure 1). The Clock mRNA expression level was lower than that of the control group at the corresponding time point (P < 0.05, * in Figure 1).

Changes in miR-182 and Clock mRNA expression in each group after HIE.
Clock protein expression changes in pineal gland tissues after HIBD
The Clock protein expression level in the HIE group was lower than that of the control group at the corresponding time point after the HIBD model setup (P < 0.05).
miR-182 binds the 3′-UTR of the CLOCK gene in vitro
In this experiment, luciferase gene detection was used to validate the interaction between miR-182 and the CLOCK gene. HeLa cells were transfected with a reporter gene carrying the predicted miR-182 targeting sequence in the CLOCK gene 3′-UTR, and then the miR-182 mimics were added. The luciferase activity declined by 55% (the luciferase activity in the target sequence is shown in Figure 2). However, when the targeting sequence was substituted by a mutant sequence, the decrease in activity disappeared. Thus, miR-182 interacts with the targeting sequence in the CLOCK gene 3′-UTR (the luciferase activity in the mutant sequence is shown in Figure 3).
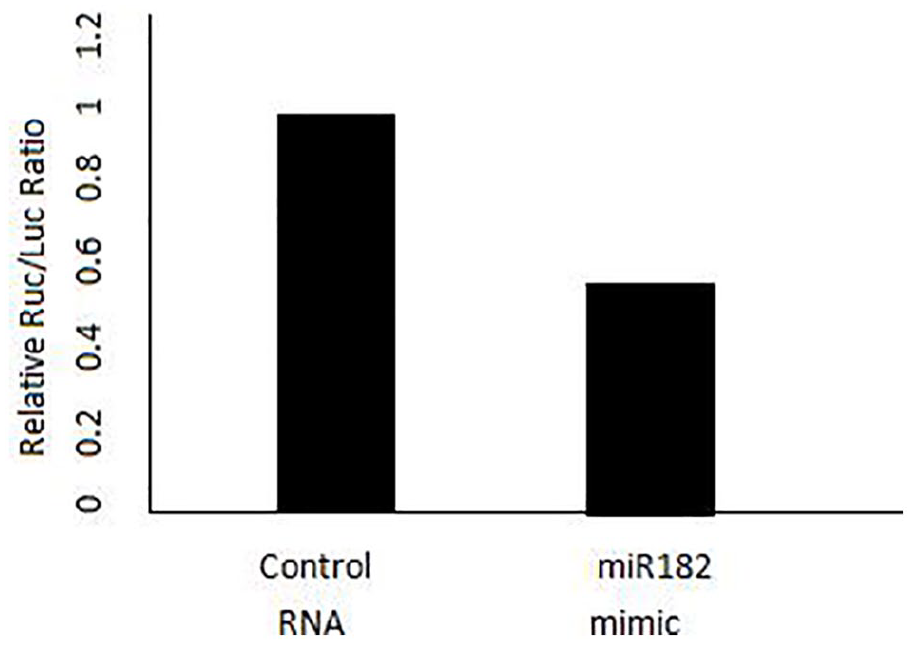
Luciferase activity in the target sequence.
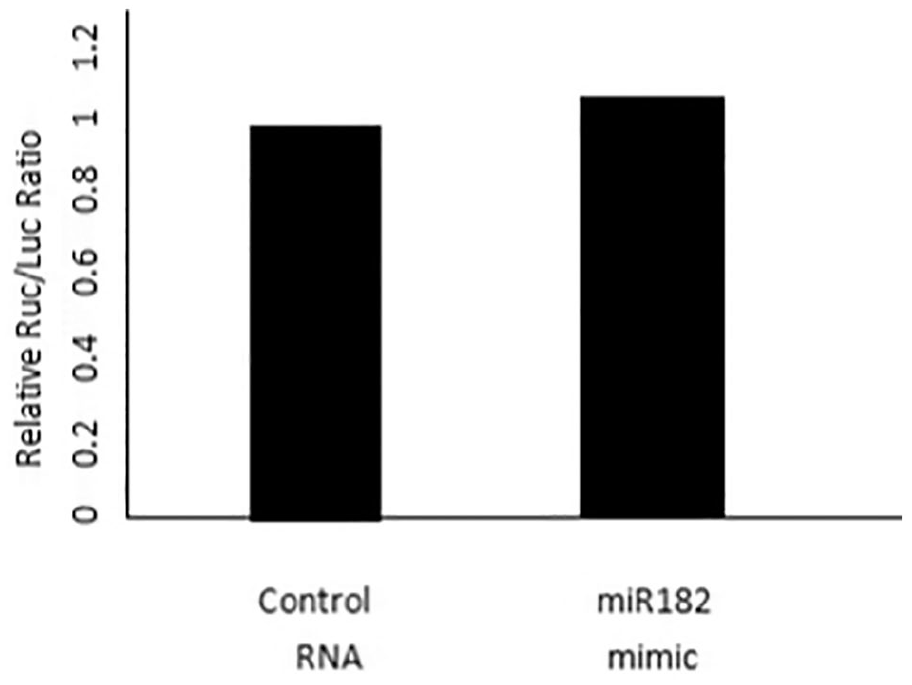
Luciferase activity in the mutant sequence.
Discussion and prospective
More and more pediatricians pay attention to the possible sequelae of perinatal children.10–13 Through the long-term follow-up of HIE patients, we found that there were different degrees of sleep disorders in mild HIE and moderate HIE patients compared with normal children. Moderate HIE children have a higher risk of respiratory related sleep disorders than mild HIE children, consistent with previous reports. The reason may be: compared with mild HIE, moderate HIE can cause extensive cortical and subcortical damage, which may have a significant impact on the neuromuscular control of the upper respiratory tract. At the same time, this study found that the maintenance of regular SWC was affected by the history of HI during the development of children.
Modern biological research shows that although there is no circadian pacemaker in the rat pineal gland, all clock genes are still expressed in the pineal gland, and their functions in the pineal gland are similar to those of central circadian oscillators. 14 It is well known that pineal gland has an effect on endocrine output. Therefore, it is considered as a neuroendocrine effector of circadian system. Therefore, pineal gland is one of the best models to study the biological clock of vertebrates.15,16
In this study, we screened microRNA (miRNA) in the pineal gland of HIBD newborn rats, and identified a miRNA called miR-182. The expression of this miRNA was significantly reduced in HIBD, combined with the 3′-UTR of CLOCK gene and regulated its expression. These findings may provide clues to elucidate the mechanism of miRNA regulation of circadian abnormalities in neonates with HIBD. MiR-182 has been known to play an important role in promoting tumor metastasis in vivo, and to maintain the function of the outer segment of the cone receptor and vision in adults.17,18 Previous studies have shown that abnormal miR-182 precursors may lead to circadian dysrhythmia in patients with severe depression accompanied by insomnia.19,20 Our results are consistent with the above results. This study shows that miR-182 is obviously enriched in the pineal gland, and plays a major role in regulating the abnormal increase of CLOCK protein after HIBD in newborn rats. However, the downregulation of miR-182 did not seem to have a significant effect on the overall expression of CLOCK gene, because the addition of miR-182 analog did not affect the expression of brain and muscle Arnt-like 1 (BMAL1) protein. Future research will focus on identifying more downstream targets of miR-182, thus providing more evidence to explain how altered expression causes sleep and wakefulness disorders in neonates with brain injury.
Limitations of the study
The study needs to be further confirmed by large-scale prospective control study, and at the same time, sleep rhythm problems of children with different degrees of HIE in adulthood still need further follow-up.
Conclusion
Children with mild and moderate HIE showed significant sleep disorders in varying degrees, which provided a clinical basis for improving the long-term prognosis of children with HIE through targeted treatment of sleep disorders. MiR-182 is highly expressed in the pineal gland and is related to the expression of CLOCK protein. CLOCK gene is the target gene of miR-182, which provides a new target for the treatment of rhythm disorder related to the damage of pineal function caused by HIBD.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study was approved by the Institutional Ethics Committee of Wuhan Children’s Hospital (ethics no. 2010023), and surgical operations on all animals are approved by IACUC (approval no. 2010XS020), Wuhan Tongji Medical College Affiliated to Huazhong University of Science and Technology. Written informed consent was obtained from the legal guardian of the child before the study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the legal guardian of the child before the study.
Trial registration
It is not applicable for the study.
