Abstract
The study aimed to detect the expression level of interleukin-37 (IL-37) in patients with rheumatoid arthritis (RA) and explore its clinical significance. A total of 40 peripheral blood samples from active and stable RA patients were collected (40 patients with RA), and peripheral blood from 40 healthy volunteers was used as the control group. Peripheral blood serum and peripheral blood mononuclear cells (PBMCs) were isolated. The expression of IL-37 mRNA in PBMCs was detected by real-time fluorescence quantitative PCR. Serum levels of IL-37, rheumatoid factor (RF), and anticyclic citrullinated peptide antibody (CCP) were measured by enzyme-linked immunosorbent assay (ELISA). The results were then calculated and analyzed. The results showed that expression of IL-37 mRNA in the PBMCs of patients with RA was significantly higher than that in the control group (P < 0.05). Expression of IL-37 mRNA in the PBMCs of the active period group was significantly higher than that in the stable period group (P < 0.05). IL-37 levels in patients with RA were significantly higher than those of the control group (P < 0.05). IL-37 levels in the active period group were also significantly higher than those of the stable period group (P < 0.05). The comparative analysis of RF and anti-CCP antibody levels showed that IL-37 was positively correlated with RF and anti-CCP levels in patients with RA. In conclusion, the expression level of IL-37 in peripheral blood of RA patients was significantly higher than that of normal control group, and it was correlated with RF and CCP antibody levels, indicating that IL-37 plays an important role in the development of RA.
Introduction
Rheumatoid arthritis (RA) is an autoimmune disease. Characteristic joint lesions are mainly caused by the proliferation of fibroblast-like synoviocytes (FLS).1,2 Abnormally proliferating FLS and infiltrating inflammatory cells produce a variety of inflammatory cytokines (such as interleukin (IL)-1, IL-6, and TNF-α). These factors are integral in causing cartilage and bone destruction. 3 IL-37 anti-inflammatory cytokines have immunosuppressive functions. 4 They can reduce the ability of certain cells (such as macrophages, monocytes, and epithelial cells) to secrete pro-inflammatory factors and inhibit lipopolysaccharide-(LPS) induced inflammatory shock in mice. It has been demonstrated that IL-37 has a wide range of inhibitory effects on inflammation, 5 but its mechanism of action remains unknown. McNamee et al. 6 showed that IL-37 has a protective effect on mice with colitis. Bulau et al. 7 also showed that the expression of IL-37 in mice can reduce inflammation provoked by ConA-induced hepatitis. Boraschi et al. 8 found that IL-37 is highly expressed in the synovial tissue in RA. The principal cells constituting synovial tissue are synovial cells, which are mainly located in the synovial lining. This layer primarily includes macrophage-like synoviocytes (MLS) and FLS. 9 The expression of IL-37 in synovial tissue in RA is highly elevated in an inflammatory environment. The precise nature of changes to FLS expression of IL-37 resulting from inflammatory substances remains unclear. This study will investigate the expression of IL-37 in the peripheral blood of patients with RA in an inflammatory environment.
Materials and methods
Subjects
The subjects of this study were divided into two groups: the RA patient group and the healthy control group. For the healthy control group, 40 cases were selected from January 2016 to December 2017, for which health checkups were performed at the Tangshan Gongren Hospital physical examination center. Ages in this group ranged from approximately 25–72 years. The RA patient group was further divided into active period group and stable period group. The specific grouping was as follows:
Active period group: 40 patients with RA were selected from January 2016 to December 2017 in Tangshan Gongren Hospital. Ages ranged from approximately 23–71 years.
Stable period group: 40 patients with RA in remission were selected. Ages ranged from approximately 25–74 years.
Diagnosis met the diagnostic criteria for RA according to the RA disease activity score (DAS). The DAS score of patients in the active period group was calculated to be ⩾3.2, and the DAS score of patients in the stable period group was <3.2. At the same time, the patients’ clinical data were recorded. All subjects gave informed, signed consent to participate in the study and this study was approved by the ethics Committee of Tangshan Gongren Hospital.
Experimental equipment and reagents
Main equipment: TS100 inverted phase contrast microscope; low-temperature high-speed centrifuge; 80-2 centrifuge; SW-CJ-2F clean bench (Shanghai Wanheng Precision Instrument Co., Ltd., China); ES315 high-pressure steam sterilizer (China, Shandong Bolanda Machinery Co., Ltd., China); ultralow temperature refrigerator (Shandong Boke Biological Industry Co., Ltd., China); micropipette (Shanghai RongTai Biotechnology Co., Ltd.,China); AF200 ice making machine; ultrapure water meter (Shanghai Sheyan Instrument Co., Ltd.,China); Hitachi 7600 automatic biochemical analyzer (Hitachi, Japan); automatic electrochemiluminescence analyzer (Swiss Roche); 7500 real-time fluorescence quantitative PCR instrument (ABI, USA); microplate reader (BIO-RAD, USA).
Main reagents: IL-37 enzyme-linked immunosorbent assay (ELISA) test kit (E16185h; Huamei Biotechnology Co., Ltd., China); lymphocyte separation fluid; anhydrous ethanol; Roche anti-CCP kits (Tianjin Haoyang Biological Products Technology Co., Ltd., China); Hitachi RF Kit (Hitachi, Japan); Trizol reagent (Thermo Fisher Scientific, USA); RT-PCR kits; SYBR quantitative PCR kit (Shanghai Jima Pharmaceutical Technology Co., Ltd., China); DEPC water (Shanghai Harling Biotechnology Co., Ltd., China).
Research methods
Separation of PBMCs
The specific steps for separating PBMCs were as follows:
On an empty stomach, 2 mL of venous blood was collected in an ethylenediaminetetraacetic acid (EDTA)-2K anticoagulant tube. An equal amount of Hank’s buffer was added and mixed. 2 mL of lymphatic fluid was aspirated into a fresh centrifuge tube. Diluted blood was added to the supernatant and centrifuged at 2400 r/min for 20 min at room temperature. The turbid buffy coat at the junction of the separation fluid and the plasma comprised the mononuclear cell layer. Capillary pipettes were used to draw mononuclear cells and transfer them to new centrifuge tubes. 4 mL of Hank’s solution was added and centrifuged at 2400 r/min for 5 min, and the samples were washed twice. The supernatant was discarded, and the bottom layer contained mononuclear cells. The separated cells were counted, and the cells were adjusted to the desired concentration using Hank’s solution and stored at −80°C.
Extraction of total cellular RNA
L × 105 PBMCs were collected. LmL Trizol was added to the EP tube, which was placed at room temperature for 5 min to completely lyse the cells. The cell lysate was then transferred to a 1.5 mL EP tube. Trichloromethane was added at 0.2 mL/mL. The contents were mixed and allowed to stand at room temperature for 5 min at 4°C, after which the tube was centrifuged at 12,000 g for 15 min. The upper, colorless liquid was moved to a new EP tube. A volume of isopropanol equivalent to that of the supernatant was aspirated, mixed by inversion several times, and allowed to stand at room temperature for 10 min. This was then centrifuged at 12,000 g for 10 min at 4°C. The supernatant was subsequently filtered. 1 mL of DEPC (Diethypyrocarbonate) water was prepared and pre-chilled, and 75% ethanol was then added. The centrifuge tube was then shaken, and the pellet was suspended and washed. Then, the liquid was centrifuged at 7500 g for 5 min at 4°C. The ethanol was carefully discarded, and the RNA pellet was dried in air for 5 min. 50 μL of DEPC water was used to dissolve the RNA precipitate, and the solution was mixed. The solution was then dissolved in a water bath at 55–60°C for 10 min. A 5 μL RNA sample was collected. 95 μL DEPC water was added for dilution. An ultraviolet spectrophotometer was used to determine the sample concentration, A260 and A280. If A260/A280 was between 1.8 and 2.0, it indicated that the extracted RNA was of good purity. The remaining samples were stored at −80°C.
Synthesis of first strand cDNA
cDNA synthesis reaction reagent was added to a centrifuge tube in the absence of RNase enzyme. The steps were: total RNA 1 µg, 5×PrimeScnpt®RT Master Mix 4 µL, and add DEPC ddH20 to 20 µL.
The mixture was reacted at 37°C for 15 min, and the reaction was terminated by heating to 85°C for 5 s and subsequent quenching on ice. The reaction product was the template cDNA, which was then stored at −20°C.
Design and synthesis of primers
The IL-37-specific primer sequences are: IL-37, upstream primer: 5’-TTAGAAGACCCGGCTGGAAGCC-3’ (196bp), downstream primer: 5’-AGATCTCTGGGCGTATGTAGT-3’ (196bp); GAPDH, upstream primer: 5’-ACCCAGAAGACTGTGGATGG-3’ (204bp), downstream primer: 5’-TTCTAGACGGCAGGTCAGGT-3’ (204bp).
Real-time fluorescence quantitative PCR
Real time FQ-PCR detection reaction system is: cDNA template 2 µL, upstream primer (10 µM) 0.8 µL, downstream primer (10 µM) 0.8 µL, SYBR®Premix Ex TaqTM II (2×) 10 µL, Rox Reference Dye II (50×) 0.4 µL, RNase free water 6 µL. The total volume is 20 µL.
The reaction conditions were as follows:
The predenaturation condition was 95°C for 30 s, while the PCR amplification condition was 95°C for 30 s and 60°C for 34 s. The melting curve was 72°C–95°C. The above cycle was repeated 40 times.
ELISA detection of IL-37 levels
A total of 40 peripheral blood samples were collected from patients with RA during the active phase and stable phase, and 40 peripheral blood samples were obtained from the control group. The sera were centrifuged and stored in a −20°C freezer. IL-37 was detected using an ELISA test kit. The main steps were as follows.
The kit was taken out and equilibrated to room temperature (20–25°C) for 30 min. The number of plate wells for the antibody was determined. The 2000 pg/mL standard product was diluted to 100 μL per well and then added to a row of seven wells. To the first well, only a sample dilution was added as a control. 100 μL of test serum was added to each well. After adding the sample, the plate was attached with a sealing film. Then, it was placed in a 37°C water bath and incubated for 2 h. The liquid in the plate was filtered, and the moisture in the dry well was blotted up with absorbent paper. The prepared biotin-labeled anti-human IL-37 antibody working solution was added sequentially at 37°C for 1 h at 100 μL per well. The solution was washed thrice with 0.01 M TBS or 0.01 M PBS for 2 min each time. TMB coloring working solution was then added sequentially at 100 μL per well, and the reaction was completed at 37°C in the dark. The first three to four wells of the standard showed a distinct blue gradient. When the difference between the 3rd and 4th wells was not obvious, 50 μl of the TMB stop solution was added to each well. Absorbance values were measured at 450 nm with a microplate reader. Based on the absorbance of the sample, the corresponding concentration was found on the coordinates.
Detection of RF and CCP
The detection of RF was performed by immune transmission turbidimetry. Serum samples from patients with RA and those in the control group were collected. After the biochemical analyzer was self-tested, a standard product was placed for calibration. The specimen to be tested was then placed in the instrument, the detection instructions input, and the test performed. Test results were recorded for statistical analysis.
Anti-CCP antibodies were measured by electrochemiluminescence immunoassay. After self-inspection of the immunoassay analyzer, a standard product was placed, and the calibration test performed. The specimen to be tested was placed in the instrument, the detection instructions input, and the test performed. Test results were recorded for statistical analysis.
Statistical analysis
All data were analyzed using SPSS 19.0 software. Measured data were expressed as mean ± standard deviation. The t-test was used for comparison between the two groups. Comparison of the means between groups was performed using one-way analysis of variance (ANOVA). Pearson’s linear correlation analysis method was used to analyze the correlation between the two variables. P < 0.05 indicated that the difference was statistically significant.
Results
General clinical data
The general clinical data for statistical studies are shown in Table 1:
General clinical data.

RA: rheumatoid arthritis.
Detection of IL-37 mRNA expression in PBMC of patients with RA
Real-time FQ-PCR was used for detection. The results showed that the melting curves of the target gene and the reference gene had only a single peak, and only a single product was amplified. This demonstrated that the specificity of the primers designed in this study was suitable. The amplification curves of the target gene and the reference gene showed an exponential growth phase and a plateau phase from the maximum baseline, indicating that product amplification had occurred. As can be seen in Figure 1, the relative expression of IL-37 mRNA in the active period group increased by a factor of 3.26 compared with that of the control group (P < 0.05). The relative expression of IL-37 mRNA in PBMCs of the stable period group increased by a factor of 2.23 compared with those of control group (P < 0.05). The relative expression of IL-37 mRNA in the active period group was significantly lower than that of the stable period group (P < 0.05).

Relative expression of IL-37 mRNA in PBMCs of rheumatoid arthritis patients and control group (the active period group compared with the control group *P < 0.05; the stable period group compared with the control group and P < 0.05; the active period group compared with the stable period group #P < 0.05.).
Results of IL-37 ELISA detection in patients with RA
The standard curve was constructed. The linear regression equation was y = 482.94x2 − 84.86x + 0.62, r2 = 0.9989. The IL-37 concentration in all samples was calculated. The results showed that the IL-37 levels (70.94 ± 21.72 pg/mL) in patients with RA was significantly higher than those of the control group (17.35 ± 4.59 pg/mL; P < 0.05). The IL-37 levels in the active phase group (86.55 ± 20.15 pg/mL) was significantly higher than that in the stable phase group (54.89 ± 14.36 pg/mL; P < 0.05). The results are shown in Figure 2.
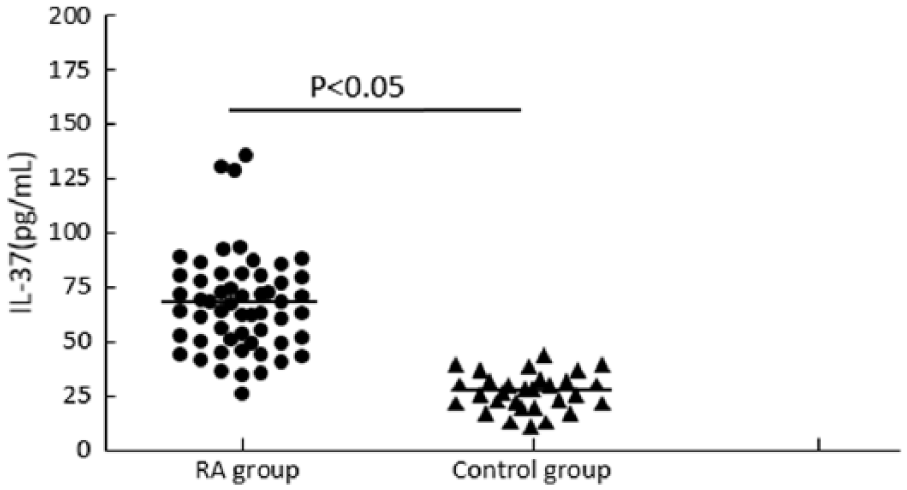
Comparison of IL-37 levels (A: IL-37 level in rheumatoid arthritis patients and control group, B: IL-37 level in active phase group and stable phase group).
Correlation analysis of IL-37 level with anti CCP antibody and RF
As depicted in Table 7, anti-CCP antibody (314.15 ± 98.15 U/mL) and RF (317.06 ± 142.23IU/mL) concentrations in the active period group were significantly higher than those of the stable period group (CCP: 221.35 ± 63.83U/mL, RF: 199.16 ± 97.36IU/mL; P < 0.05). Moreover, IL-37 levels in patients with RA were positively correlated with levels of both anti-CCP (r = 0.473, P = 0.001) and RF antibody (r = 0.379, P = 0.003; P < 0.05). The results are shown in Figure 3.
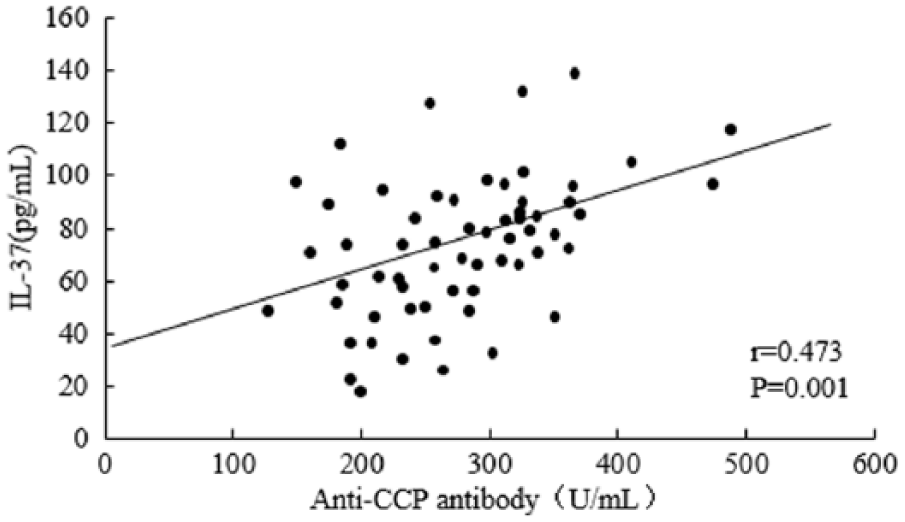
Correlation analysis chart (A: Correlation between IL-37 levels and anti-CCP antibodies in patients with rheumatoid arthritis, B: Correlation between IL-37 levels and RF in patients with rheumatoid arthritis).
Discussion
IL-37 is an anti-inflammatory cytokine with a protective role in many diseases,10,11 such as Crohn’s disease12,13 and LPS-induced septic shock. 14 Studies by Nold et al. 15 showed that IL-37 is highly expressed in the synovium of patients with active RA, but not in healthy people. As an anti-inflammatory factor, IL-37 may inhibit the overexpression of inflammation by mediating a negative feedback mechanism. It has been thus suggested that IL-37 is involved in the pathogenesis of RA.
This study found that the expression of IL-37 mRNA in the group with RA was significantly higher than in the control group, suggesting that IL-37 plays a role in the pathogenesis of RA. IL-37 may be involved in the regulation of inflammatory responses in RA. ELISA results showed that IL-37 levels in the RA group were significantly higher than in the control group (P < 0.05). In addition, in patients at different clinical stages of RA, IL-37 levels were also different. IL-37 levels in the active period group were significantly higher than those in the stable period group (P < 0.05). The correlation analysis between measured IL-37 levels and clinical laboratory indicators of RA showed that IL-37 levels in patients with RA were significantly correlated with RF and anti-CCP antibody levels. This suggests that IL-37 is closely related to the production of specific autoantibodies in RA. Abnormal levels of RF and anti-CCP antibodies indicate that the patient has a severe condition with a rapid progression, as well as severe bone destruction. 16 This indicates that IL-37 levels have a distinct correlation with the severity of RA.
High expression of IL-37 in RA patients can exert anti-inflammatory effects through the following aspects: a large number of CD4+T Th1 cells are present in the synovial tissue of RA patients. It produces cytokines such as IL-1α, IL-1β, and IL-18, which have a strong pro-inflammatory effect. IL-18 can induce IFN-γ production, leading to increased inflammation in RA. Studies have shown that IL-37 can inhibit IL-18 signaling pathways and reduce IFN-γ synthesis. 17 It has been suggested that IL-37 can reduce the inflammatory response in RA by inhibiting the synthesis of IFN-γ. IL-37 achieves its anti-inflammatory effect by inhibiting the activity of dendritic cells (DCs). 18 DCs can activate autoreactive T cells of the RA synovium, which leads to the persistence of inflammation. This experiment’s results show that increased IL-37 levels in patients with RA can inhibit the activity of DCs and reduce the innate immune response induced by DCs. The combination of IL-37 and Smad3 proteins exerts an inhibitory effect on inflammation. Smad3 can bind to TGF-β and migrate to the nucleus to affect gene transcription. The incorporation of DNA in the nucleus inhibits the activation of DCs and macrophages, makes T cells tolerant, and inhibits cytotoxicity, thus reducing inflammatory responses. TGF-β is highly expressed in RA patients. IL-37 can promote TGF-β’s inhibitory properties through Smad3, 19 thereby exerting its anti-inflammatory effects.
In sum, IL-37 is involved in the pathogenesis of RA. IL-37 levels in patients with RA were positively correlated with autoantibodies, including RF and anti-CCP antibodies, in RA. This suggests that IL-37 can be used as an index to assess the severity of illness.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
