Abstract
In this study, we synthesized amino propyl ciprofloxacin (CPLX-NH2) as a ciprofloxacin (CPLX) derivative. Moreover, the immune antigen CPLX-NH2-BSA and coating antigen CPLX-NH2-OVA were prepared via CPLX-NH2 coupling with bovine serum albumin (BSA) and ovalbumin (OVA), respectively. Subsequently, the Kunming mice were immunized with immune antigen to obtain the polyclonal antibody with high titer. The regression equation of CPLX-NH2 antibody was y = –17.395x + 89.331 (R2 = 0.9961); IC50 and limit of detection (LOD) were 182.39 and 20.09 ng/mL, respectively. These results were superior to that of CPLX antibody. Meanwhile, the CPLX-NH2 antibody showed cross-reactivity to fluoroquinolones (FQNs) residues. The results of the study indicated that the proper modification of the drug, namely, the addition of a suitable spacer arm between the drug and the carrier protein will improve the efficacy of the antibody, which is a favorable concept for preparation of antibody.
Introduction
Fluoroquinolones (FQNs) is the third generation of quinolone antibiotics, which is derived from the quinolone nalidixic acid with the introduction of the fluorine atom at 6-position and the piperazine moiety at 7-position. 1 FQNs had excellent antibacterial activity to gram-negative bacteria, gram-positive bacteria, coccobacteria, and gonococci. Moreover, FQNs exhibited the powerfully inhibited effect for some of the chlamydia, sprirochete, mycoplasma, and anaerobic bacterium. Thus, FQNs can be applied to almost all the regular bacteria-infected diseases. However, widespread application of FQNs often accompanied drug-abuse and unreasonable application. Therefore, the adverse drug reaction (ADR) of FQNs had obtained more attention. Traditionally, drug residues analysis relied on chromatography, such as high-performance liquid chromatography (HPLC), 2 liquid chromatography/mass spectrometry (LC-MS), 3 and LC-MS/MS. 4 However, chromatography has many limitations such as time-consuming and instruments valuableness. Moreover, the sample pretreatment are complicated and difficult for field operations. Therefore, establishment of appropriate methods for rapid screening large numbers of samples of residues of FQNs are urgently needed. Among the methods of measurement, the immunoassays were widely used because of high sensitivity, rapidity, less sample dosage, and operation simplicity. 5
We all know, antibody plays important roles in immunoassays methods. However, antigen is the key factor for the preparation of antibody. FQNs have no immunogenicity as a small-molecule hapten. Meanwhile, it could not stimulate the body to produce specific antibodies against epitopes. As a consequence, to prepare antibody, artificial antigens were initially synthesized with conjugating FQNs to the carrier protein. Amazingly, antibodies usually have the excellent identification ability for antigens that are far removed from the binding site. 6 Therefore, increase of one spacer arm between small molecule of the drug and the carrier protein is an effective strategy to facilitate the production of specific antibodies against haptens.
In this work, we synthesized the compound amino propyl ciprofloxacin (CPLX-NH2) by introducing a amino propyl to the nitrogen-atom of piperazinyl moiety of ciprofloxacin (CPLX; Figure 1) as an attachment site to a carrier protein. We envisioned that this strategy with insertion of amino propyl chain can produce high-sensitivity polyclonal antibodies with broad-specificity toward FQNs residues. The antibodies showed satisfactory selectivity and measurement relationships for FQNs residues by enzyme-linked immunosorbent assay (ELISA; competitive indirect enzyme-linked immunosorbent assay (Ci-ELISA)). Thus, these results exhibited increase of one spacer arm between the drug molecule, and the carrier was able to prepare perfect antibody, which provided the possibility for rapid determination of FQNs residues.

Structures of ciprofloxacin (CPLX) and ciprofloxacin analog (CPLX-NH2).
Materials and methods
Identification of complete antigens
The CPLX-NH2, bovine serum albumin (BSA), ovalbumin (OVA), CPLX-NH2-BSA, and CPLX-NH2-OVA were diluted with phosphate-buffered saline (PBS) in a certain proportion, respectively (the conjugations were measured in the protein). According to the methods in references, PBS used as the blank control, each mixture was scanned in the 200–400 nm of ultraviolet region, respectively, and scanning patterns were combined to prove qualitatively whether conjugation is successful. Complete antigens of CPLX were confirmed by the same method.
Preparation of polyclonal antibodies
A total of 10 female Kunming mice were raised in the Experimental Animal Center of Hebei North University (SYXK (Hebei) 2014-0062), free drinking water and eating. After 1 week of adaptive feeding, mice were divided into two groups of five mice each: derivative group and control group. Five injections of immunogen at 2-week intervals were given to each mice.
The initial immunization were injected intraperitoneally at multiple points with a 1:1 (v/v, 200 μL per mouse) mixture of an immunogen CPLX-NH2-BSA (100 μg) in PBS and FCA (derivative group). The subsequent booster immunizations were given intraperitoneally with the same amount of immunogen emulsified with Freund’s incomplete adjuvant (FIA) at 2-week intervals after the initial immunization, and the fifth booster immunization was direct injection of immunogen. After 1 week from each booster injection, mice were tail-bled and the antisera were tested for antihapten antibody titer by indirect ELISA using a homologous coating antigen. A week after the final boost, the antisera were collected and aliquoted into two: one part for subsequent experiments and another frozen at −20°C for use.
Replace CPLX-NH2-BSA with CPLX-BSA, control mice (control group) were immunized with the same dose and method.
Optimized concentration of coating antigen/antibody (Ag/Ab) for ELISA procedure
Before development of the Ci-ELISA, the best working concentration of the immunoreagents (coating antigen and antibody) was determined using a checkerboard titration.
The coating antigen was coated transversely in microplate at the concentration of 8.00, 4.00, 2.00, 1.00, 0.50, 0.25, 0.125, and 0.0625 μg/mL and was added at 100 µL/well, respectively. The antibody was diluted with PBS from 1:2000 to 1:64000 times by double dilution, the longitudinal microplate plate was added (100 µL/well), and the rest is the same as indirect ELISA method. The coating antigen and the antibody with the optical density (OD) value of 1.00 and were selected as the optimal working concentration.
Development of the standard curve
Based on the result of checkerboard titration, a Ci-ELISA method was established under optimized conditions, and the calibration curve was fitted based on the average of three separate assays in triplicate. Reciprocal value of the inhibition rate (I) of different concentrations was taken as ordinate (the inhibition ratio (I) of antigen was determined by Ci-ELISA at the optimized conditions. I = B0/B, B is the OD value of the inhibition calculated as corresponding concentration of sample, B0 is the OD value of the corresponding concentration without antigen inhibition, that is, OD value of the blank control) and the standard concentration logarithm of serial dilution of CPLX-NH2 (or CPLX) was served as abscissa to calculate regression equation and correlation coefficient.
Identification of specificity
The specificity of antibody was evaluated according to the cross-reactivity (CR). CR is a phenomenon inherent to immunoassay, which can affect analytical results by either false positives or by increasing the concentration of the target compound when both the target and one or more analogs are present. CR of CPLX-NH2 (or CPLX) antibody with competitors, including CPLX-NH2, CPLX, ofloxacin, sarafloxacin (SALX), enrofloxacin (EFLX), difloxacin, streptomycin sulfate, ampicillin sodium, and penicillin sodium were assayed by Ci-ELISA, respectively. The 50% inhibition value (IC50) of each competitor was calculated by plotting their respective standard curves with serial dilution of the competitor solution as the tested antigen. CR of the antibody with each competitor was calculated according to the following formula

Assessment of the immunoassay precision for spiked samples
The chicken samples (Yonghui Supermarket, Zhangjiakou) were selected to assess the performance of immunoassay using two antibody for detection of FQNs. FQNs-free chicken samples were attrited and homogenized in a homogenizer, and the homogenates (3 g/sample) were spiked with FQNs at three concentration levels (20, 25, 30 ng/g), respectively. The spiked samples were allowed to stand at 4°C for 30 min. Each spiked sample was extracted with 2 mL PBS for 10 min on a Vortex shaker. The extract was heated for 10 min in a water bath at 100°C to degenerate the proteins in the spiked samples, and the extract was then centrifuged for 10 min at 5000 ×
Results
First, we synthesized the amino propyl derivative of CPLX by the classical nucleophilic substitution, and the compound structure was identified with nuclear magnetic resonance (NMR), infrared (IR), and electrospray ionization mass spectroscopy (ESI-MS) (Figure 2, for details see the Supporting Information).

Synthesis of amino propyl derivative of ciprofloxacin (CPLX-NH2).
Keeping 3-amino propyl derivative of CPLX in hand, preparation of complete antigen was completed based on previous methods. 7 As seen in Figure 3(a), the maximum absorption peak of CPLX-NH2 appear at 271.00 nm and BSA at 278.40 nm; the spectrum of CPLX-NH2-BSA was different from either of them with its maximum absorption peak at 271.60 nm. These results indicated that the small target molecule CPLX-NH2 was attached to the carrier protein (BSA). Therefore, the conjugation between CPLX-NH2 and BSA was successful, and the conjugates of CPLX-NH2-BSA could be used as animal immune antigen. Meanwhile, to explore the influence of 3-amino propyl chain for function of immune antibodies, we made the molecule CPLX to attach the carrier protein (BSA) as control with the same method (Figure 3(b)).

UV-vis spectra of complete antigen: (a) CPLX-NH2-BSA and (b) CPLX-BSA.
Furthermore, we prepared the coating antigen, namely, the CPLX-NH2 was loaded onto another protein (OVA). As seen in Figure 4(a), the maximum absorption peak of OVA was at 279.60 nm, the spectrum of CPLX-NH2-OVA was different from either of them with its maximum absorption peak at 274.00 nm. These results indicated that the target small-molecule CPLX-NH2 was attached to the carrier protein (OVA). 8 Hence, the conjugation between CPLX-NH2 and OVA was successful, and the conjugates of CPLX-NH2-OVA could be used as coating antigen. On the other hand, the same principle can be proved that the conjugation between CPLX and the carrier protein (OVA) was successful (Figure 4(b)).

UV-vis spectra of coating antigen (a) (CPLX-NH2-OVA)/ (b) (CPLX-OVA).
With the completed antigen (CPLX-NH2-BSA) in hand, the serum was obtained from one selected mouse, and we measured the antibody titer by indirect ELISA within 7 days after the third immunization. The results displayed the titer of CPLX-NH2 serum was up to 1:64000, but CPLX serum was up to 1:16000. First, this result showed that antibody against the antigen was produced and the immunization could continue. More importantly, the efficiency of antibody production was increased with adding short chain of the small drug molecules. 9
Inspired by the above results, we further explored the condition for practical application. The blood was gained from orbit of each mouse with 7 days after the fifth immunization. The results of checkerboard titration showed that the titer of CPLX-NH2 antibodies can be up to 1:128000, the optimal working concentration of the antibody was 1:32000, and the optimal concentration of coating antigen was 0.125 μg/mL. In addition, the titer of CPLX antibodies can be up to 1:64000, the optimal working concentration of the antibody was 1:16000, and the optimal concentration of coating antigen was 0.25 μg/mL.
We had tested the lowest detection limit with I = 1.5 in view of the lowest detection limit of ELISA is determined with I ⩾ 1.5–2. 10 The Ci-ELISA standard curve of CPLX-NH2 antibody was shown in Figure 5(a). The results showed that there were excellent linear relationship between I value (B/B0) and the logarithm of CPLX-NH2 concentration (R2 = 0.9961). Thus, the limit of detection (LOD) was 20.09 ng/mL as I = 1.5. 11 Moreover, IC50 of the antibody was 182.39 ng/mL. However, the linear relationship (R2 = 0.9875), IC50 (225.94 ng/mL) and LOD (31.39 ng/mL) of CPLX antibody (Figure 5(b)) were relatively worse than that of CPLX-NH2 antibody. Therefore, these results mean that the strategy of short chain is efficient for improving the linear relationship and specificity, and reducing LOD of antibody.
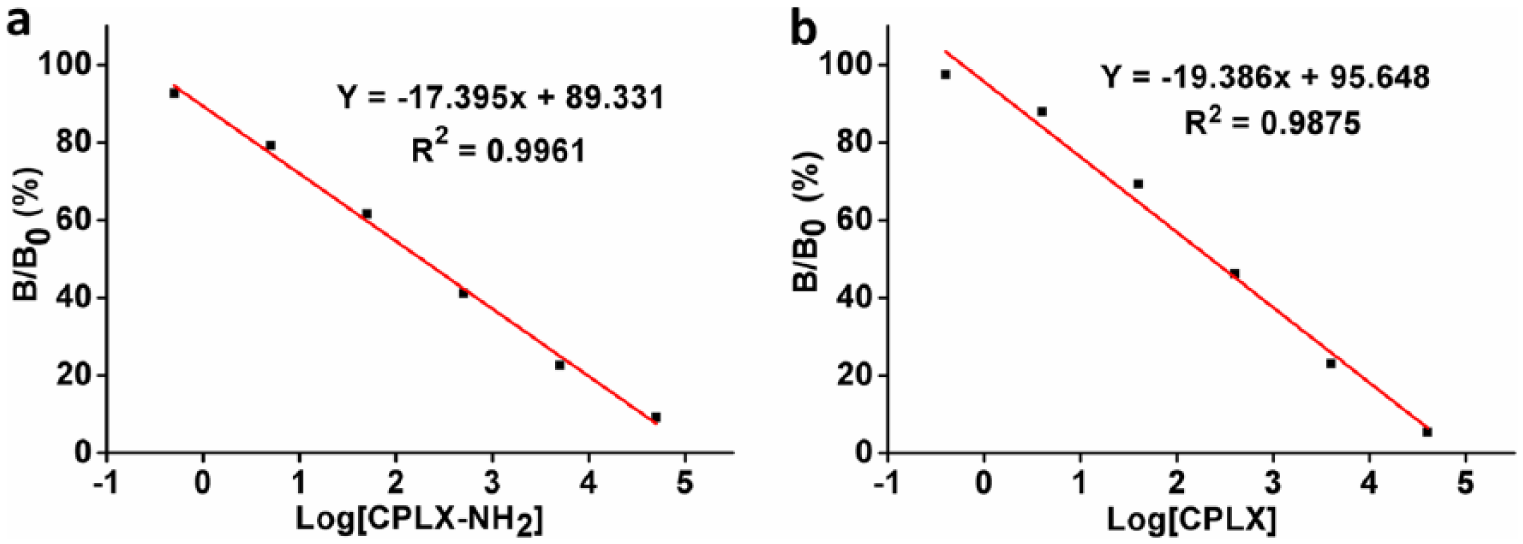
Standard curve for detection of (a) CPLX-NH2 and (b) CPLX antibody by Ci-ELISA.
We also researched the CR of antibody, and the results were shown in Table 1. The investigation demonstrated that the CPLX antibody and CPLX-NH2 antibody exhibited a higher CR for various FQNs. However, the antibodies do not possess CR to streptomycin sulfate, ampicillin sodium, and penicillin sodium. Thus, the antibody has broad-spectrum property to the same type of drugs and specificity to the different kind of drugs. Based on broad-spectrum specificity and practicability of the method, the antibody could be used to detect the residues of CPLX, danofloxacin, and EFLX.
The cross-reactivity (CR) of antibody for various FQNs.

CPLX-NH2: amino propyl ciprofloxacin; CPLX: ciprofloxacin.
Finally, two antibodies were applied to analyze chicken samples with three concentrations of three different FQNs. As shown in Table 2, The detections of CPLX-NH2 antibody were more approximate to HPLC than detections of CPLX antibody. The results demonstrated that the immunoassay using CPLX-NH2 antibody can be used in the determination of FQNs in agricultural samples.
Recovery of FQNs from spiked chicken samples by immunoassays of two antibody (n = 3).

CPLX-NH2: amino propyl ciprofloxacin; CPLX: ciprofloxacin; HPLC: high-performance liquid chromatography.
Discussion
FQNs can be applied to almost all the regular bacteria-infected diseases. However, widespread application of FQNs often accompanied drug-abuse and unreasonable application. To reduce the risk and preserve the damage of FQNs to humans, the maximum residue limits (MRLs) were restricted. For example, the MRLs is 100 µg/L for the sum of CPLX and EFLX in milk and 30 µg/kg in chicken muscle; tolerance limits for SALX, norfloxacin (NFLX), CPLX, and other FQNs is in a range of 10–200 µg/kg for various foods in Japan. 12 Therefore, it is of crucial importance to build effective methods of detecting the residues of FQNs in animal-based food.
Among the methods of measurement, the immunoassays were widely used. We all know, antibody plays important roles in immunoassays methods. The length and location of spacer arm were pivotal to manufacture antigen. Increase of one spacer arm between small molecule of the drug and the carrier protein is an effective strategy to facilitate the production of specific antibodies against haptens. We all know, if the length is shorter, the characteristic structure of hapten molecule will be masked by carrier proteins, but if longer, configuration of hapten molecule is easy to be folded so that hapten molecules closed to the protein surface are shielded. Thus, the length of 3–6 carbons chain is relatively appropriate. In addition, antibody can recognize hapten moiety away from the binding site, so the location of conjugating hapten with carrier protein is preferably remoted from the important antigenic determinant. Therefore, one side chain is linked to the piperazine of CPLX structure based on the importance of F atom at 6-position, carbonyl at 4-position, and carboxyl at 3-position for antibody recognition.
In this study, we synthesized CPLX-NH2 as a CPLX derivative. Moreover, the immune antigen CPLX-NH2-BSA and coating antigen CPLX-NH2-OVA were prepared via CPLX-NH2 coupling with BSA and OVA, respectively. Subsequently, the Kunming mice were immunized with immune antigen to obtain the polyclonal antibody with high titer. The regression equation of CPLX-NH2 antibody was y = –17.395x + 89.331 (R2 = 0.9961); IC50 and LOD were 182.39 ng/mL and 20.09 ng/mL, respectively. These results were superior to that of CPLX antibody. Meanwhile, the CPLX-NH2 antibody showed CR to FQNs residues. The results of the study indicated that the proper modification of the drug, namely, the addition of a suitable spacer arm between the drug and the carrier protein will improve the efficacy of the antibody, which is a favorable concept for preparation of antibody.
Supplemental Material
Supporting_Information – Supplemental material for An effective strategy for preparation of a polyclonal antibody with an addition of carbon chain of ciprofloxacin
Supplemental material, Supporting_Information for An effective strategy for preparation of a polyclonal antibody with an addition of carbon chain of ciprofloxacin by Dong Wei, Guozhen Fang and Shuo Wang in European Journal of Inflammation
Footnotes
Authors’ Contribution
The project was conceived by D.W. and S.W. The experiments were carried out by D.W. and G.F. under the supervision of S.W., and the manuscript was written by D.W.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Hebei province of China (grants no. C2016405007), the 100 Outstanding Innovative Talents Support Program of Hebei Higher Education Institutions of China (III) (grant no. SLRC2017019), and the Doctoral Fund of Hebei North University of China (grant no. BSJJ1405).
Supplemental material
All additional information pertaining to experimental methods are given in the supporting information, available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
