Abstract
Intestinal ischemia/reperfusion (I/R) is life-threatening and challenging in clinical practice. CDDO-imidazolide (CDDO-Im) is therapeutic in alleviating I/R injury. Nevertheless, there is a lack of investigation on the effects of CDDO-Im on intestinal I/R. Mice were randomly divided into four groups: (a) the sham group, (b) the CDDO-Im group, (c) the I/R group, and (d) the I/R + CDDO-Im group. Intestinal I/R was performed by clamping arteria mesenteric anterior for 45 min, followed by 24 h reperfusion. In addition, Kaplan–Meier method and the log-rank test were used to compare the survival rates among groups by observing for 24 h. Intestinal I/R in model group demonstrated severe injury of the intestinal mucosa, lung, kidney, and liver. The intestinal mucosal damage and intestinal barrier dysfunction were obviously attenuated in CDDO-Im-treated group compared with the model group. Also, preconditioning with CDDO-Im reduced pulmonary, hepatic and renal damage, and decreased oxidative stress (malondialdehyde (MDA), superoxide dismutase (SOD), and NO) and pro-inflammatory responses (tumor necrosis factor (TNF-α), interleukin 1β (IL-1β), and interleukin 6 (IL-6)) following I/R injury. Furthermore, we also observed that these protective properties of CDDO-Im were accomplished by the activation of nuclear factor E2-related factor 2 (Nrf2) signaling pathway and upregulation of its downstream antioxidant genes, including heme oxygenase (HO-1), NQO-1, and glutamate–cysteine ligase regulatory subunit (GCLM). Our data suggest that CDDO-Im exerts a beneficial effect on intestinal I/R-associated mucosal barrier dysfunction and distant organ injuries.
Keywords
Introduction
Intestinal ischemia/reperfusion (I/R) is a severe clinical event caused by acute mesenteric ischemia, trauma, sepsis, sepsis shock, and surgical procedure. 1 Increased intestinal permeability induced by intestinal I/R can provide an outlet for abundant bacteria and their antigens from the luminal environment into circulation and mesenteric lymph, leading to systemic response syndrome (SIRS) or multiple organ dysfunction syndrome (MODS).2,3 Although extensive investigation efforts have focused on characterizing the pathogenesis of distant intestinal injury and organ dysfunction, the underlying mechanisms remain the subject of debate, and no effective method prevents or controls the process.
Oxidative stress plays an important part during intestinal I/R injury. Reactive oxygen species (ROS) are excessively produced in injured tissues and cells following I/R injury, which could trigger apoptosis or necrosis, induce the amplification of pro-inflammatory response, and destroy the gut mucosal barrier function. 4 Nuclear factor E2-related factor 2 (Nrf2) signaling pathway is reported to exert a critical effect during the pathogenesis of I/R injury. 5 Nrf2 and its downstream target genes are widely regarded as a multiple-organ protector owing to its anti-oxidative and cyto-protective functions. 6 In addition, pharmacologic upregulation of Nrf2 pathway has suggested to protect against gut inflammation and epithelial barrier function.7,8
One of the synthetic oleanane triterpenoids, 2-cyano-3, 12 dioxooleana-1, 9 dien-28-imidazolide (CDDO-Im) is a potent factor in activating Nrf2 signaling and has been used extensively in various diseases in numerous disease models, such as acute kidney or lung injury. 9 Recent research has suggested that CDDO-Im exerted an antioxidant effect following I/R injury. 6 In addition, CDDO-Im has been reported to alleviate the inflammatory response in sepsis model. 10 However, the protective effects of CDDO-Im during intestinal I/R and the involved mechanisms remain illuminated. Therefore, the present study was performed to investigate the protective effects and potential mechanisms of CDDO-Im against intestinal I/R injury in mice.
Methods
Ethical statements
Male C57BL/6 J mice obtained from Model Animal Research Center of Nanjing University were kept under specific conditions in a temperature controlled room. Experimental mice included a mixture of males and females, 8–12 weeks of age (mice were matched for age and gender within each experiment). The animal study was designed and performed according to the principles of the Declaration of Helsinki and with the approval from the Institutional Animal Ethical Committee of Hainan Provincial People’s Hospital. Furthermore, all animal experiments should comply with the animal research: reporting of in vivo experiments (ARRIVE) guidelines (http://www.nc3rs.org.uk/arrive-guidelines).
I/R model
The model of intestinal I/R injury was performed as described previously. 11 In brief, mice were anesthetized intraperitoneally with chloral hydrate. After midline laparotomy, the superior mesenteric artery (SMA) of I/R mice was identified and occluded with an atraumatic microvascular clamp for 45 mins followed by 24 h of reperfusion. Intestinal tissues and plasma were extracted and kept at −80°C for further analysis. The mice were divided into following 4 groups: (a) sham operation, (b) CDDO-Im group, (c) I/R group, and (d) I/R + CDDO-Im group. CDDO-Im (2 mg/kg) was administrated intraperitoneally 3 h before beginning intestinal ischemia. 6 CDDO-Im was prepared freshly just before administration by dissolving the frozen powder in phosphate-buffered saline containing 10% dimethyl sulfoxide (DMSO) and 10% cremphor. Control mice received the same volume of vehicle. The mice that failed to survive were excluded from analysis following I/R procedure. After reperfusion, small intestine without adipose tissue was taken from the same place at the distal end of ileum (2–6 cm proximal to the distal ileum) for further analysis.
Histopathological assessment and apoptosis detection
Paraffin section (4 μm thickness) of intestinal tissue was stained by hematoxylin and eosin (H&E) for light microscopy. Chiu et al. 12 described histological severity of intestinal tissue according to the methods with following modifications: no damage, score 0; subepithelial space at villous tip, score 1; loss of mucosal lining of the villous tip, score 2; loss of less than half of the villous structure, score 3; loss of more than half of the villous structure, score 4; and transmural necrosis, score 5. Sections were blindly assessed. Histological severity of pulmonary tissue was also evaluated using H&E examination. Lung injury was assessed on the basis of the presence of exudates, hyperemia/congestion, neutrophil infiltration, and intra-alveolar hemorrhage/debris. 11
Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) was used to detect apoptosis in intestine on the basis of the manufacturer’s instructions. The proportion of TUNEL-positive cell helps to quantify the apoptotic index, as previously described. 13
Biochemical assay
The oxidative stress in the intestinal tissues was measured using malondialdehyde (MDA), superoxide dismutase (SOD) and glutathione peroxidase (GSH-Px) and KeyGen Biotech based on the manufacturer’s recommendations. Serum and tissue inflammatory cytokines (tumor necrosis factor (TNF-α), interleukin 1β (IL-1β), and interleukin 6) in mice were evaluated by enzyme-linked immunosorbent assay (ELISA) kit (R&D System). The contents of alanine transaminase (ALT), glutamic oxalacetic transaminase (AST), and creatinine were (Nanjing Jiancheng, China) detected by using the commercial kits on the basis of the manufacturer’s recommendations.
Measurement of intestinal permeability
The mice were starved for 4 h and then administered fluorescein isothiocyanate (FITC)-dextran at a dose of 600 mg kg-1 by gavage. After 4 h, the mice were killed and bled by cardiac puncture. Fluorometry was used to detect the serum FD-40 levels. Ussing chamber analyses were performed to evaluate the ex vivo permeability of intestines in mice, as described previously. 14
Bacterial translocation
The caudal lymph nodes (CLN) and the mesenteric lymph nodes (MLN) were taken for bacteriological cultures using aseptic techniques, and each node (0.1 g) was homogenized with 0.9 mL of sterile saline in a tissue grinder. In addition, blood (obtained by cardiac puncture) was obtained for bacterial colony counts by diluting and plating on Luria broth (LB) agar, as previously described. 13 Bacterial growth in the plates was expressed as colony-forming units per gram of tissue. When over 102 colonies/g of tissue were found, the result of cultures was considered to be positive.13,14
Real-time quantitative polymerase chain reaction
Total RNA was extracted from intestine using the TRIZOL reagent (Life Technologies Inc., Carlsbad, CA, USA), and the oligo (dT)-primed complementary DNA was used for reverse transcription of the purified RNA. The transcript levels of interest genes were evaluated by quantitative polymerase chain reaction (PCR) using SYBR Green detection. Levels of each gene were quantified as a relative value to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) and then normalized to the mean value of the control group. The following primers were used to determine messenger RNA (mRNA) expression levels: Nrf2, GCLM, HO-1, NAD(P)H, quinone oxidoreductase-1 (NQO-1), and glyceraldehyde phosphate dehydrogenase (GAPDH). Primers for quantitative polymerase chain reaction (qPCR) analyses of the relevant sequences are listed in Supplementary Table 1.
Data analysis
The results were expressed as means ± standard deviation (SD), and were analyzed by SPSS 17.0 (Chicago, IL, USA) and GraphPad Prism software 6.0 (La Jolla, CA, USA). One-way analysis of variance or Student’s t-test was performed to compare the continuous variables between groups. In all cases, P < 0.05 was considered as significant.
Results
Pretreatment with CDDO-Im protects against intestinal I/R injury and improves survival
In the Figure 1, we demonstrated the protective effects of CDDO-Im on the intestines. Intestinal I/R group was characterized by severe intestinal mucosal damage, thin and short villi, wider villi spacing, collapse of intestinal mucosal epithelial cells, capillary hemorrhage, and infiltration by inflammatory cells (Figure 1(c)). Compared with the model group (I/R group), mice pretreated with CDDO-Im obviously reduced intestinal I/R injury, and had a markedly decrease in Chiu’s score (Figure 1(d)). In addition, 24 out of the 30 mice were dead 24 h post-intestinal I/R, while 18 of the 30 mice injected with CDDO-Im were still alive 24 h after intestinal I/R. In general, the survival rate in the I/R + CDDO-Im group was significantly higher than that of the I/R group (Figure 1(f)).
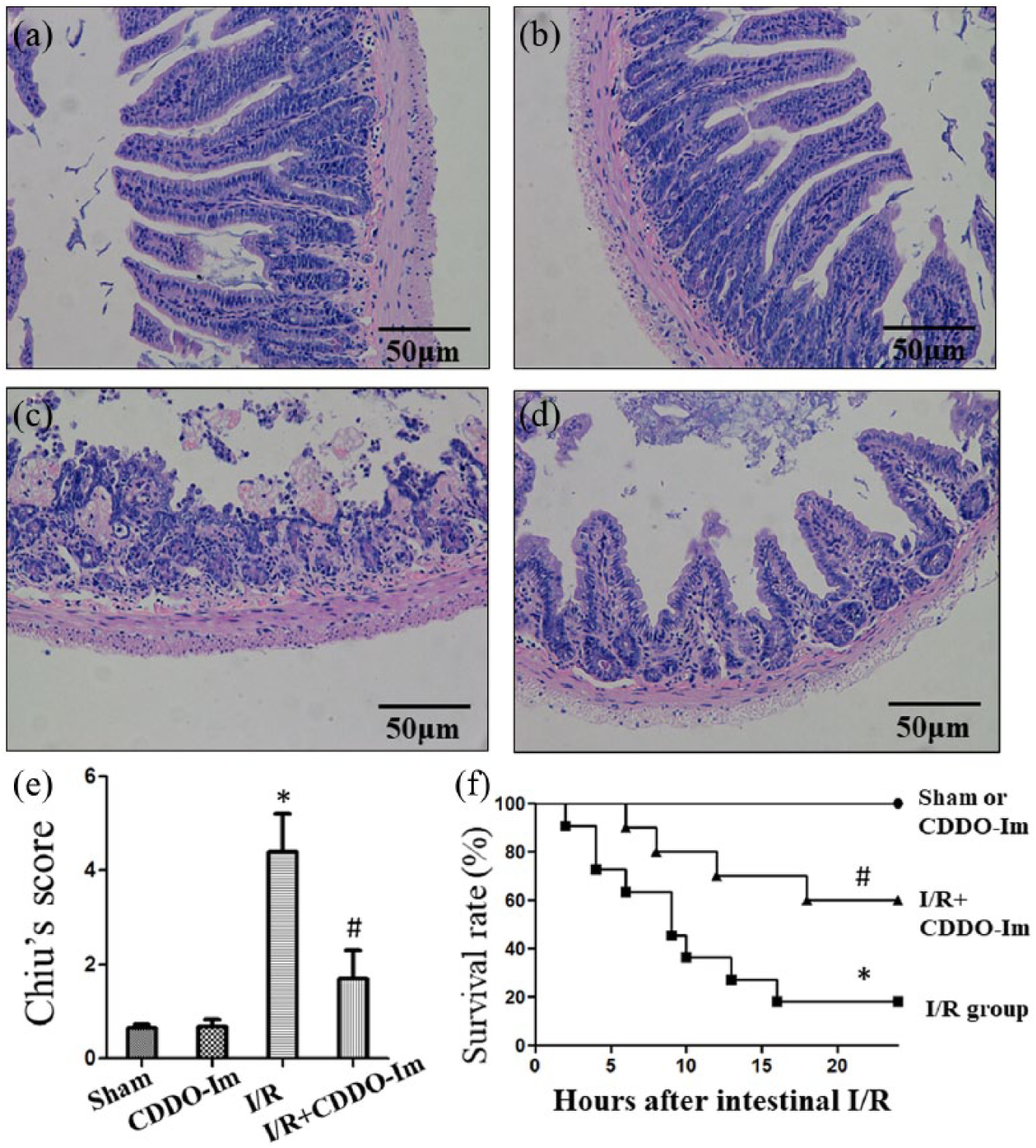
CDDO-Im protects against intestinal injury after intestinal ischemia reperfusion (I/R) injury. Representative photomicrographs of (a) sham-operated group, (b) CDDO-Im, (c) I/R group, and (d) I/R + CDDO-Im group. (e) Intestinal histology was evaluated using the Chiu’s score. (f) 24 h survival analysis of the CDDO-Im and I/R treated mice after intestinal I/R. The survival rate was compared by Kaplan-Meier method and the log-rank test. *P < 0.05 versus Sham or CDDO-Im group, #P < 0.05 versus I/R group.
CDDO-Im preserves intestinal permeability and prevents bacterial translocation
As shown in Figure 2(a) and (b), serum FD-40 and intestinal permeability were higher in the I/R injured-mice compared with the sham group, which were markedly decreased via CDDO-Im pretreatment. In addition, mice in I/R group decreased small intestinal bowel permeability resistance compared with the sham group; however, the resistance was decreased in the CDDO-Im group (Figure 2(c)). Intestinal barrier integrity was usually evaluated by detecting bacterial translocation to MLN, central lymph nodes (CLN), and blood. Our results demonstrated that the I/R mice showed significant bacterial translocation to MLN, CLN, and blood compared with the sham group. However, pretreatment with CDDO-Im obviously decreased the bacterial translocation to distant organs during intestinal I/R injury (Figure 2(d)).

The effects of CDDO-Im on intestinal permeability and bacterial translocation. (a) Serum FD-40 was measured to evaluate in vivo permeability. (b–c) The in vitro permeability of the mouse intestines was measured by performing Ussing chamber analyses. The MLN and CLN were collected and cultured at 37ºC for 24 h. (d) The culture results of bacterial growth were considered positive when more than 102 colonies/g of tissue were found *P < 0.05 versus sham or CDDO-Im group; #P < 0.05 versus I/R group.
Effects of CDDO-Im on I/R-associated oxidative stress and inflammatory response
Oxidative stress is a critical event in the process of intestinal I/R injury. 4 Figure 3(a) showed that the expression of SOD and GSH-Px were dramatically reduced and MDA expression was significantly elevated in the I/R group compared with the sham group. CDDO-Im pretreatment markedly increased the expression of SOD and GSH-Px, and decreased the elevated MDA expression compared to the I/R group. In addition, given inflammatory activation plays an important role in accelerating intestinal I/R injury, 1 thus we evaluated the tissue expression of pro-inflammatory cytokines (including IL-1β, IL-6, TNF-α) and neutrophil infiltration (myeloperoxidase (MPO)) by ELISA. Moreover, serum inflammatory mediators were significantly improved by CDDO-Im treatment followed by intestinal I/R injury (Supplementary Table 2). Figure 3(b) and (c) showed that CDDO-Im preconditioning significantly attenuated the increased inflammatory cytokines and MPO expression following intestinal I/R injury. In addition, CDDO-Im preconditioning obviously down-regulated the increased NO level following I/R-injured intestines (Figure 3(d)).

CDDO-Im alleviated oxidative stress and inflammatory response during I/R injury. Effects of (a) CDDO-Im on oxidative stress, (b) tissue inflammation, (c) MPO activity, and (d) NO levels in intestinal tissue after I/R. P*< 0.05 versus sham or CDDO-Im group; #P < 0.05 versus I/R group.
Pretreatment of CDDO-Im reduces intestinal apoptosis after I/R
Apoptosis of intestinal epithelial cells is considered to be of the important events during the pathophysiology of intestinal I/R injury. Therefore, intestinal apoptosis levels were evaluated in both CDDO-Im treated and untreated group by TUNEL assays. As seen in the Figure 4, the apoptosis index decreased from 45.6% ± 10.7% in the I/R group to 21.3% ± 8.7% with CDDO-Im preconditioning (P < 0.05). Thus, pretreatment of CDDO-Im reduces apoptosis following intestinal I/R injury.
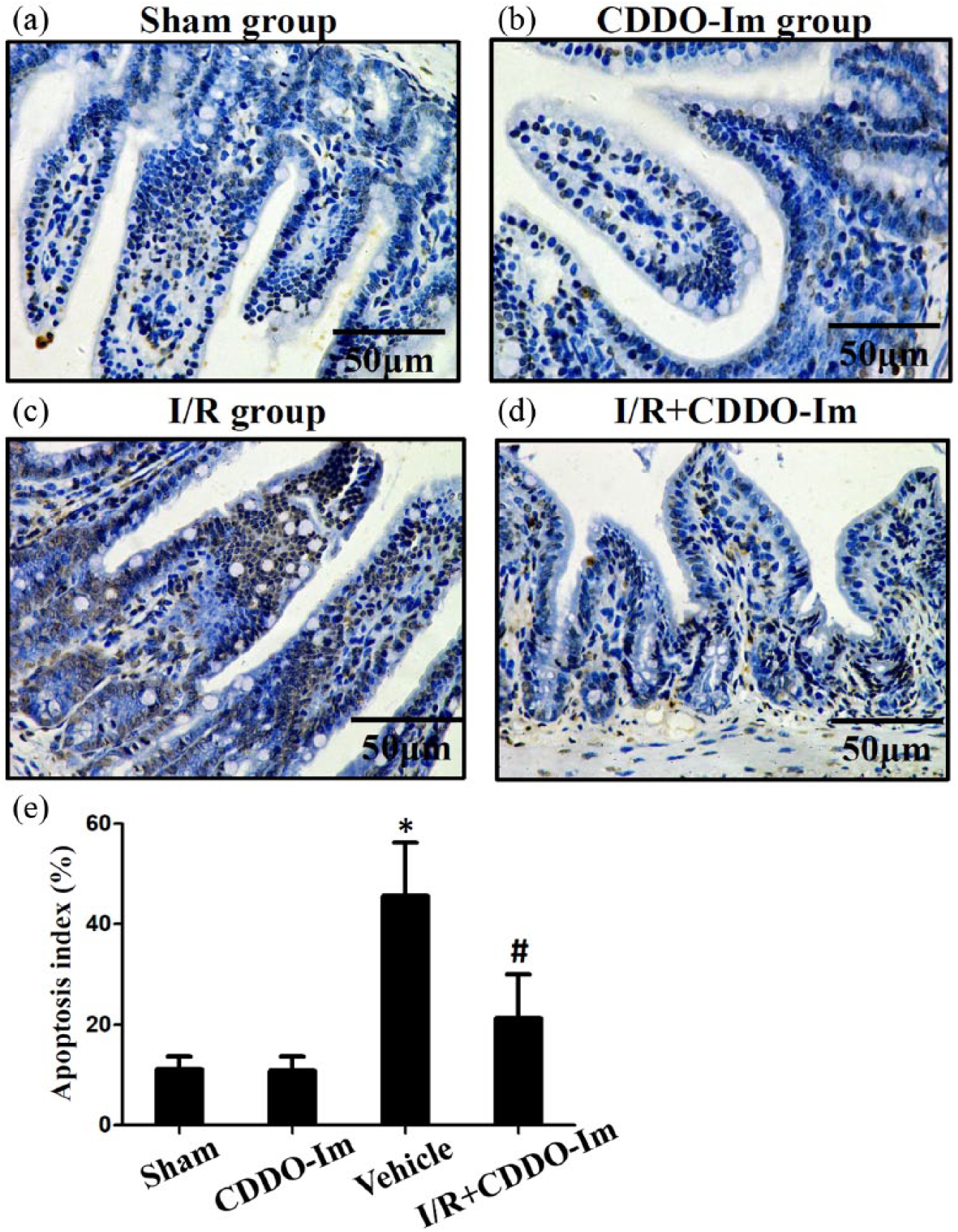
CDDO-Im protects against apoptosis after intestinal I/R injury. Representative photomicrographs illustrating apoptotic nuclei (TUNEL fluorescence staining) in the intestine of the (a) sham group, (b) CDDO-Im group, (c) I/R group, and (d)–(e) I/R + CDDO-Im group. Histogram of the apoptosis index in the ileum of mice. *P < 0.05 versus sham or CDDO-Im group, #P < 0.05 versus I/R group.
CDDO-Im upregulates antioxidant genes via Nrf2 activation
The effects of CDDO-Im on intestinal I/R-mediated Nrf2, GCLM, NQO-1 and HO-1 levels were detected using real-time PCR assay. The results show significant upregulation (P < 0.01) of GCLM, NQO-1 and HO-1 during CDDO-Im or I/R treatment when compared to the control group. The CDDO-Im pretreatment following I/R injury showed the most (P < 0.001) expression of Nrf2, GCLM, NQO-1 and HO-1 (P < 0.001) (Figure 5(a) and Supplementary Figure 1). Immunofluorescence was also used to verify the effect of CDDO-Im pretreatment on I/R-injured intestines via Nrf2 signaling pathway. The fluorescence intensity of Nrf2 was markedly increased in the I/R + CDDO-Im group (Figure 5(b) and (c)).

Effects of CDDO-Im on the activation of the Nrf2 antioxidative pathway. qPCR of total Nrf2, GCLM, NQO-1 and HO-1 (a) among four groups. (b) Immunofluorescence analysis of the effect of CDDO-Im preconditioning on the expression of Nrf2 after intestinal I/R injury. *P < 0.05 versus sham or CDDO-Im group, #P < 0.05 versus I/R group.
CDDO-Im alleviates acute pulmonary injury and protects against hepatic and renal injuries
Lung is one of the most seriously injured distant organs caused by intestinal I/R injury. 15 By H&E staining, histological changes of lung tissue in the I/R-injured mice were severe compared with the sham group, including alveolar congestion, intracellular hemorrhage, exudates and inflammatory infiltration (Figure 6(a)). Nevertheless, CDDO-Im pretreatment significantly alleviated these acute pulmonary injury, as seen in the markedly decreased lung injury scores. In addition, in the kidney, the CDDO-Im pretreatment markedly decreased the creatinine level compared with the I/R group (Figure 6(b)), and in the liver tissue, CDDO-Im preconditioning significantly reduced plasma ALT and AST following intestinal I/R injury (Figure 6(c)).

CDDO-Im reduced distant organ injury after intestinal I/R injury. (a) Representative photomicrography of the lung section, and lung tissue injury in mice submitted to sham, I/R, and CDDO-Im-treated groups. Plasma creatinine (b) ALT and (c) AST levels were measured after intestinal I/R injury. The data represent the mean ± SD. *P < 0.05 versus sham or CDDO-Im group, #P < 0.05 versus I/R group.
Discussion
Intestinal I/R injury is a severe clinical challenge with a high rate of mortality, despite surgical intervention. 16 Ischemia induces tissue disruption because of dysfunction of oxygen delivery. Accumulation of toxin and depletion of cellular energy leads to apoptosis or necrosis. Paradoxically, restoration of the blood supply induces additional cell damage and amplifies inflammatory response via excess oxidative stress, thereby aggravating organs injuries and cell death.3,17 Therefore, pharmacological intervention is a critical method for protecting against intestinal ischemic injury following I/R injury. 18
CDDO-Im has been suggested to have a cytoprotective and anti-apoptotic effect on mice I/R model as a novel activator of the Nfr2 pathway, 19 and these protective effects are related to the inhibition of the oxidative stress and the inflammatory response. Thimmulappa et al. 10 demonstrated that pretreatment with CDDO-Im alleviated lipopolysaccharides (LPS)-induced inflammatory cytokine secretion and mortality. Apoptosis of intestinal epithelial cells is considered as one of the critical accelerants of gut epithelial integrity disruption. 20 Xu et al. 6 demonstrated that CDDO-Im treatment helped to protect against cell death (such as apoptosis and necrosis) following I/R injury, and protection mediated by CDDO-Im on apoptosis maybe attributed to the up-regulation of anti-apoptotic genes, including B-cell lymphoma-2 (Bcl2) and B-cell lymphoma-extra large (Bcl-xl). Nevertheless, the specific signaling mechanisms involved in the preventive effects of CDDO-Im against intestinal I/R injury remain unclear. Furthermore, how CDDO-Im protects against systemic inflammation and distant organs injuries, including the lung, liver and kidney, following intestinal I/R injury has not been investigated. In our study, we aimed to investigate the effects of CDDO-Im on reducing intestinal I/R injury, including pulmonary, hepatic and renal injury, and the possible signaling pathway involved.
Regarding the distant organs injuries with the etiology of I/R, occlusion of the SMA induces local intestinal damage and the reperfusion induces amplifying systemic pro-inflammatory responses, which could further induce the damage of multiple organs, such as lung, kidney and liver. 21 As far as we know, we first investigate the protective effect of CDDO-Im on intestinal I/R-associated organs injuries. In the present study, CDDO-Im pretreatment reduced pulmonary, hepatic, and, renal injury during I/R injury, as evidence by improved histological findings and biomarkers of organ functions (serum ALT, AST, and creatinine).
Inflammatory response is an important effector to the aggravation of I/R injury, and uncontrolled inflammation gives rise to advanced tissue damage. 22 An I/R injury challenge leads to the translocation of bacteria and toxins, amplifying systemic inflammation and apoptosis, and results in the occurrence of SIRS and MODS. The effects of CDDO-Im on reducing expression of TNF-α, IL-1β, and IL-6 have not been previously explored during intestinal I/R injury. Our results showed that CDDO-Im markedly reduced systemic and local TNF-α, IL-1β, and IL-6 and expression, suggesting that CDDO-Im could not only protect the organ injury caused by the ischemic insult, but inhibit the increased pro-inflammatory cytokines. Therefore, CDDO-Im pretreatment could be a potent drug in alleviating the systemic inflammatory response of intestinal I/R injury.
Oxidative stress is the other crucial event in the mechanisms of I/R injury. In order to explore the effectivity of CDDO-Im during intestinal I/R injury, oxidative biomarkers, such as MDA, SOD, and NO, were applied to detected the oxidative stress levels. Under normal condition, NO reduces the release of neutrophil and the accumulation of platelet. However, the NO levels in the tissue are markedly up-regulated following I/R injury, contributing to mitochondria disruption and dysfunction of energy metabolism. 23 In our study, the decreased NO expression of intestines in the CDDO-Im pretreatment group corresponds with the alleviative morphologic manifestations of the intestinal tissue. Hence, the protective effect of CDDO-Im in I/R-induced mucosal injury could be partly due to its anti-oxidative potential.
The Nrf2 signaling pathway has a crucial role in antioxidant and anti-inflammatory cellular responses. Under normal conditions, Nrf2 is retained in the cytoplasm by Keap1 and is degraded through ubiquitination. Upon activation of oxidative stress, Nrf2 translocates to the nucleus and initiates the transcription of antioxidant genes. 24 A recent study indicated that the Nrf2 pathway may also serve as an anti-inflammatory modulator. 25 Nrf2 activation has also been reported to reduce organ damage and prevent inflammation from hemolysis induction in sickle cell disease, and its activation significantly reduces immune cell infiltration and reduces the expression of the pro-inflammatory cytokines IL-1β, IL-6, and TNF-α through inhibiting the NF-κB signaling pathway. 26 In addition, Meng et al. 17 investigated the possible mechanisms underlying intestinal I/R-induced apoptosis. They suggested that increased Nrf2 expression helped to decrease intestinal IR-induced apoptosis and that the protective effects of Nrf2 signaling could be dependent on its anti-inflammatory effects via the inhibition of the NF-κB signaling. Our study indicated that intestinal Nrf2 expression was markedly increased after CDDO-Im treatment, and CDDO-Im significantly mediated nuclear translocation of Nrf2. Our results indicated that CDDO-Im preconditioning following intestinal I/R injury dramatically mitigated pathological changes within the intestines and reduced the inflammatory response and apoptosis by activation of Nrf2 signaling pathway.
There are some limitations of this study. First, although the CDDO-Im preconditioning significantly improved survival rate using the 24 h survival analysis, life-sustaining operations for mice were not performed within the 24 h, which were conventionally conducted in clinical resuscitations. Hence, transitional research is needed to verify whether the CDDO-Im treatment can decrease mortality induced by intestinal I/R injury. However, we consider that CDDO-Im can serve as a bridge between intestinal I/R injury and clinical practice. Second, because only TUNEL analysis was used to evaluate apoptosis in intestinal tissue, whether CDDO-Im exerts an antiapoptotic role in intestinal I/R needs to be further explored. However, we demonstrated that CDDO-Im significantly decreased TNF-α production following I/R injury, and TNF-α has been verified to activate the apoptotic cascade through the death receptor/caspase pathway. 15 We therefore conclude that CDDO-Im attenuates apoptosis following intestinal I/R injury, at least in part, through an indirect manner.
In conclusion, the results clearly suggest that CDDO-Im pretreatment leads to a significant improvement of survival rate in intestinal I/R mice. CDDO-Im can alleviate intestinal mucosal damage and intestinal epithelial apoptosis following intestinal I/R, as well as the injury to the lung, liver, and kidney. Altogether, CDDO-Im may be a novel treatment that helps to protect against intestinal I-R injury.
Supplemental Material
Supplementary_Figure_1 – Supplemental material for Protective effect of CDDO-imidazolide against intestinal ischemia/reperfusion injury in mice
Supplemental material, Supplementary_Figure_1 for Protective effect of CDDO-imidazolide against intestinal ischemia/reperfusion injury in mice by Youqun Huang, Mulin Ye, Chunlin Wang, Zhenfen Wang and Weiping Zhou in European Journal of Inflammation
Supplemental Material
Supplementary_Table_1 – Supplemental material for Protective effect of CDDO-imidazolide against intestinal ischemia/reperfusion injury in mice
Supplemental material, Supplementary_Table_1 for Protective effect of CDDO-imidazolide against intestinal ischemia/reperfusion injury in mice by Youqun Huang, Mulin Ye, Chunlin Wang, Zhenfen Wang and Weiping Zhou in European Journal of Inflammation
Supplemental Material
Supplementary_Table_2 – Supplemental material for Protective effect of CDDO-imidazolide against intestinal ischemia/reperfusion injury in mice
Supplemental material, Supplementary_Table_2 for Protective effect of CDDO-imidazolide against intestinal ischemia/reperfusion injury in mice by Youqun Huang, Mulin Ye, Chunlin Wang, Zhenfen Wang and Weiping Zhou in European Journal of Inflammation
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from Natural Science Foundation of Hainan Province (813203).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
