Abstract
Lipopolysaccharide (LPS) inhalation causes neutrophilic airway inflammation. We used LPS produced to Good Manufacturing Practice (GMP) standards to characterise the dose response. A total of 15 healthy non-smoking subjects inhaled 5-, 15- and 50-µg LPS. Whole blood cell counts and serum C-reactive protein (CRP) were measured at baseline and up to 24 h post challenge. Sputum was induced at baseline and 6 h post challenge for cell counts and quantification of myeloperoxidase (MPO), interleukin (IL)-1β, IL-6, IL-8 and tumour necrosis factor α (TNF-α) in supernatants. LPS inhalation was well tolerated. Blood neutrophil counts increased at 6 h post LPS with all doses. Serum CRP significantly increased with 15- and 50-µg LPS. All LPS doses significantly increased sputum neutrophil percentage (
Background
Lipopolysaccharide (LPS), also known as endotoxin, is a cell wall component of gram-negative bacteria that is recognised by the pattern recognition receptor toll-like receptor 4 (TLR-4). 1 TLR-4 stimulation activates transcription factors such as nuclear factor kappa-light-chain-enhancer of activated B cells (NF-кB) and activator protein 1 (AP-1), resulting in increased production of multiple pro-inflammatory cytokines. 1 LPS inhalation has been used as an experimental model of acute neutrophilic lung disease. Inhaled LPS challenge is generally well tolerated in healthy subjects, causing a rapid innate immune response with neutrophilic airway inflammation being apparent within a few hours.2–4 This has been used as a model to investigate the effects of novel drugs designed to inhibit neutrophilic airway inflammation.5–7 Induced sputum sampling is a practical method of studying inflammatory biomarkers in these studies.
The standardisation of inhaled challenge agents reduces potential sources of variation in the biological response observed. For LPS, such standardisation can ensure that the quantity and potency of inhaled material is tightly controlled. Previous challenge studies have not used standardised LPS, making it difficult to compare results across different studies due to potential variability in biological response. There is great variation in the doses of LPS used in previous healthy volunteer challenge studies,2,3,7 presumably attributable to differences in potency.
This healthy volunteer study used inhaled LPS produced according to good manufacturing practice (GMP) standards, which ensures standardisation of the potency and quality of the material for inhalation. The aim of this study was to characterise the dose response to this GMP-grade LPS. This information can be used to select an LPS dose for future clinical trials that is well tolerated and produces a robust inflammatory response.
Methods
Subjects
A total of 15 healthy subjects aged between 18 and 60 years with no history of smoking and normal lung function were recruited. Exclusion criteria were a history of cardiorespiratory or other chronic diseases that would make it unsafe for the volunteer to participate and a respiratory tract infection in the previous 6 weeks. Written informed consent was obtained, and the study was approved by a local ethics committee (Greater Manchester South 13/NW/0482).
Study design
For safety reasons, a dose ascending design was used. Subjects were required to produce an induced sputum sample at baseline. If a subject was unable to produce a sample, a repeat induction was performed. In order to be eligible for the study, subjects had to produce an adequate sputum sample at baseline. After an interval of at least 48 h, subjects were administered 5 µg of LPS from
LPS was reconstituted using 0.9% saline to produce 0.11, 0.33 and 0.83 mg/mL solutions for the 5-, 15- and 50-µg challenges. LPS was administered as an aerosol, using pre-calibrated nebuliser pots to deliver a set volume over five inhalations. A quantity of 9 µL per inhalation was used during 5- and 15-µg challenges and 12 µL per inhalation was used in 50-µg challenges. 7
Sputum was induced at 6 h post LPS challenge (following the administration of 200-µg salbutamol). 6 Blood samples were obtained prior to and at 3, 6 and 24 h post LPS challenge. Pulse, blood pressure, oxygen saturations, temperature and symptom recording were collected prior to LPS challenge and then post LPS challenge at 30 min, hourly for 8 h and at 24 h. Spirometry (using Micro-lab Spirometer, Cardinal Health, US) was performed prior to LPS challenge and then post LPS challenge at 5 and 30 min, hourly for 8 h and at 24 h. 7
Whole blood samples were analysed for differential cell counts in 13 subjects. Serum was stored at –80°C, and serum C-reactive protein (CRP) was measured using a high-sensitivity immunoturbidimetric assay with a limit of detection of 0.2 mg/L in 14 subjects.
Induced sputum was processed by a two-step method using phosphate-buffered saline (PBS) initially to obtain supernatant for protein analysis and then dithiothreitol (DTT) to obtain a cell pellet as previously described. 9 The supernatant was stored at –80°C for later analysis. Cytospin preparations (Cytospin 4, Shandon, Runcorn, UK) were stained with Rapi-Diff II (Atom Scientific, Hyde, UK). A total of 400 non-squamous cells were counted and differential cell counts obtained as a percentage of non-squamous cells. Cell viability was analysed by trypan blue exclusion. 10
Sputum supernatants were analysed for myeloperoxidase (MPO) and interleukin (IL)-8 by enzyme-linked immunosorbent assay (ELISA) (R&D Systems, Abingdon, UK), along with IL-1β, IL-6 and tumour necrosis factor α (TNF-α) by Luminex Assay (Merck Millipore, Massachusetts, USA). Assay quantitative ranges were 0.125–10 ng/mL for MPO, 34.215–2000 pg/mL for IL-8, 0.890–2000 pg/mL for IL-1β, 0.479–750 pg/mL for IL-6 and 0.967–1750 pg/mL for TNF-α. All immunoassays were performed in duplicate. The mean of the replicates was used for statistical analysis. All assays were required to demonstrate intra- and inter-assay precision with repeat measurements demonstrating co-efficient of variance ≤30%.
Clinical safety measurements (pulse, temperature, forced expiratory volume during the first second (FEV1) were compared to pre-dose (baseline) using paired t-tests with Bonferroni correction. Blood neutrophils and CRP data were analysed by repeated measures analysis of variance (ANOVA) with Dunnetts post hoc test against baseline. Parametric sputum data were analysed by repeated measures ANOVA with Bonferroni post hoc test and non-parametric data were analysed using the Friedman test with Dunns post hoc test. All analyses were performed using GraphPad Prism version 5.02 (San Diego, California; USA).
Results
Three females and 12 males with mean age of 45 years were recruited; body mass index (BMI) was within the range 20.5–30.3 kg/m2 with a mean FEV1 of 102.0% (standard deviation (SD): 12.3) at baseline. LPS challenges were performed safely in all subjects, with only mild symptoms reported. Inhalation of 5-µg LPS caused no symptoms. Three subjects experienced minor symptoms after inhalation of 15-µg LPS (mild fatigue, cough, and headache) and 50-µg LPS (cold symptoms and headache). All symptoms were resolved within 24 h. There was a significant decrease in FEV1 at 1 h after challenge with 5- and 50-µg LPS (2.5%,
Blood neutrophil differential counts were significantly increased compared to baseline at 6 h after challenge with all LPS doses (Figure 1(a);

Blood biomarkers following LPS challenge: (a) mean blood neutrophil counts at baseline, 3, 6 and 24 h following challenge with LPS 5, 15 or 50 µg (n = 13). Data shown as mean with SEM. Results analysed by repeated measures ANOVA with Dunnetts post hoc test. *
Sputum neutrophil percentage was significantly increased 6 h post inhalation of 5-, 15- and 50-µg LPS, with mean percentage changes of 16.9% (
Sputum differential cell counts.
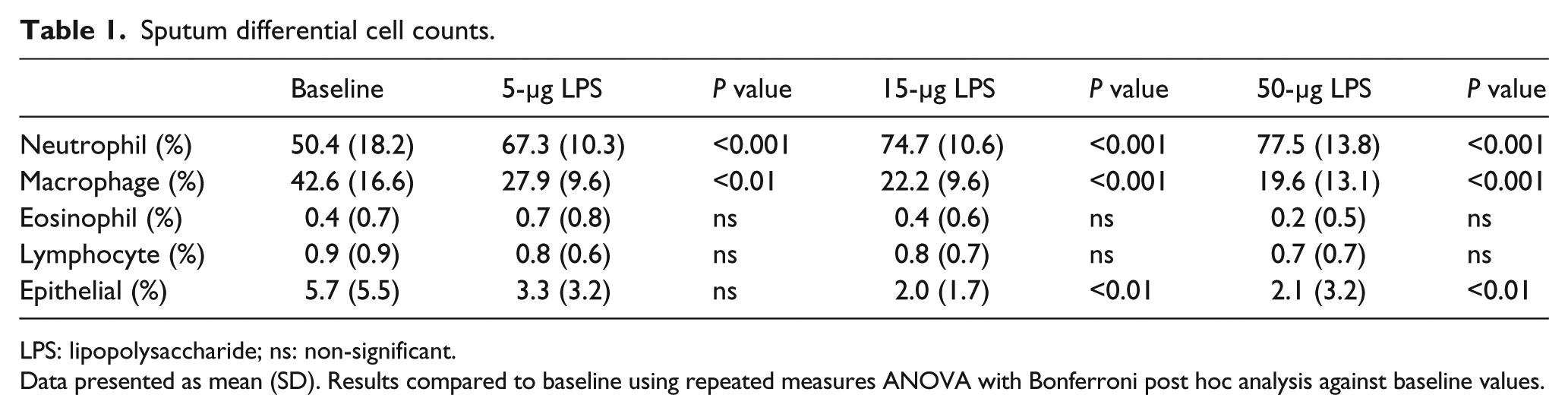
LPS: lipopolysaccharide; ns: non-significant.
Data presented as mean (SD). Results compared to baseline using repeated measures ANOVA with Bonferroni post hoc analysis against baseline values.
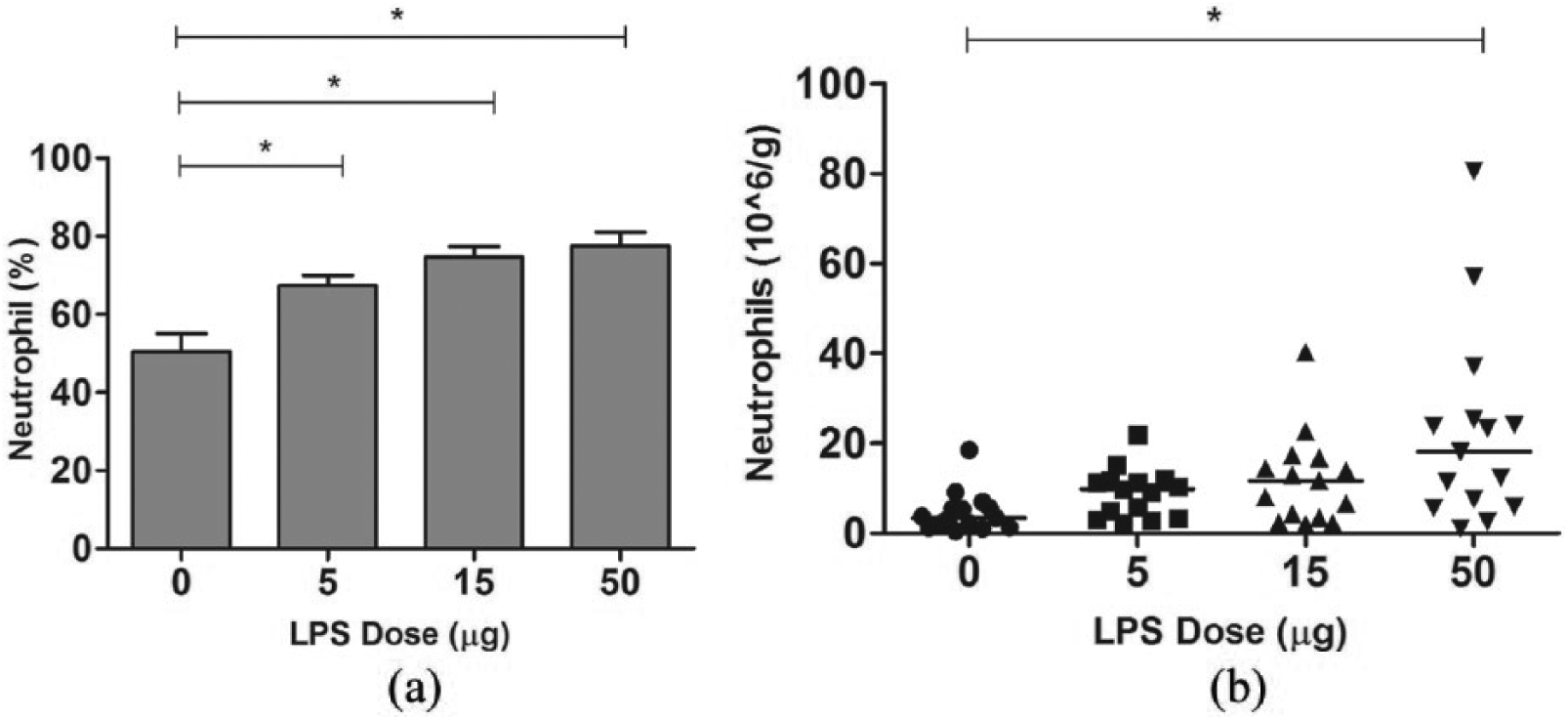
Effect of LPS challenge on Sputum Neutrophils: (a) mean sputum neutrophil levels at baseline and 6 h following challenge with LPS 5, 15 or 50 µg (n = 15). Data are shown as mean with SEM. Results analysed by repeated measures ANOVA with Bonferroni post hoc test; *
Inhalation of 50-µg LPS caused statistically significant increases in IL-1β, IL-6 and TNF-α levels (Figure 3), while lower LPS doses caused numerical increases in cytokine levels that were not statistically significant. LPS challenge did not affect levels of MPO or IL-8 (only the highest LPS dose was analysed).

Mean sputum biomarker levels at baseline and 6 h following challenge with LPS 5, 15 or 50 µg. Data are shown as mean with SEM. (a) IL-1β, (b) IL-6, (c) TNF-α, (d) MPO and (e) IL-8. Results analysed by repeated measures ANOVA with Bonferroni post hoc test, *
Discussion
Inhaled LPS caused a dose response effect in healthy human volunteers, with biomarker measurements showing that the 50-µg dose consistently caused significant inflammation in the lungs and circulation. The lower LPS doses caused neutrophilic inflammation, but generally the effects on a range of inflammatory biomarkers including serum CRP and sputum supernatant cytokines were much less consistent. The GMP-grade LPS material contributed to the clear demonstration of a dose response effect. Future studies using this material can be performed using 50-µg LPS, with confidence that both systemic and lung inflammation will be observed.
The sputum neutrophil percentage was significantly increased, compared to baseline, by all LPS doses. Although there was a numerical dose response, there was no statistically significant difference between doses. We have previously observed an apparent “ceiling effect” for sputum neutrophil percentage after LPS challenge 10 at approximately 80%–85%. In contrast, sputum neutrophil absolute count and the sputum supernatant biomarkers did not suffer with a “ceiling effect” and were able to demonstrate differences between doses.
We have previously reported a significant increase in sputum supernatant IL-6 levels following LPS challenge3,7 and now show increased IL-1β, IL-6 and TNF-α after LPS administration. In contrast, we observed no change in IL-8 levels following LPS challenge. Increased IL-8 levels have been reported following LPS challenge using a Meso Scale Discovery (MSD) analysis platform, 3 although another study reported no increase. 2 Different analysis methods could explain these differences. MPO is stored in neutrophils and released upon cell activation. 11 It has been reported that LPS challenge significantly increases levels of MPO in sputum supernatant.7,12 We observed a non-significant trend towards increased levels after 50-µg LPS. Again, sputum processing methods (PBS vs DTT) and different analytical platforms can cause different results. We focussed mainly on airway biomarkers, but various blood biomarkers can also be measured after LPS challenge.
Overall, LPS challenge using GMP-grade material was well tolerated in healthy subjects, and a dose response effect was observed. The 50-µg LPS challenge caused consistent neutrophilic airway inflammation, and is a suitable dose for future LPS challenge studies.
Footnotes
Acknowledgements
N.J., A.M. and D.S. were involved in study concept and design. N.J., A.M., C.P. and S.W. were involved in data acquisition. N.J. and A.M. drafted the manuscript. All authors were involved in data interpretation, manuscript revision and manuscript approval.
Availability of data and materials
The data sets used and analysed during this study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
D.S. has received sponsorship to attend international meetings, honoraria for lecturing or attending advisory boards and research grants from various pharmaceutical companies including Apellis, AstraZeneca, Boehringer Ingelheim, Chiesi, Cipla, Genentech, GlaxoSmithKline, Glenmark, Johnson and Johnson, Menarini, Mundipharma, Novartis, Peptinnovate Pfizer, Pulmatrix, Skypharma, Teva, Therevance and Verona. The remaining authors have no competing interests to declare.
Ethics approval and consent to participate
All patients provided written informed consent using a protocol approved by Greater Manchester South Ethics Committee, reference 13/NW/0482.
Funding
This study was funded by GlaxoSmithKline, Middlesex, UK.
