Abstract
Cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) negatively regulates the T cell activation and competes with CD28 in binding with B7.1/B7.2 molecules. Fusion of the extracellular region of CTLA-4 and a specific antigen is an effective method for improving the immune efficacy of DNA vaccines. This study aimed to investigate the effects of DNA vaccine of human epidermal growth factor receptor-2 (HER2) fused with CTLA-4 on the development of colon carcinoma in mice and to identify the potential immune mechanisms underlying its effects. We constructed recombinant plasmids corresponding to the control group, individual antigen group, and fusion antigen group. Then, mice were intramuscularly injected with the corresponding plasmids and exposed to electrical pulses. Immunogenicity was evaluated at 2 weeks after the last immunization. Furthermore, to investigate the antitumor immune effects of the recombinant plasmid, we established a mouse model of HER2 expression in transplanted tumors. Experimental results showed that the recombinant plasmids expressing fusion antigen induced a stronger cellular immune response. Inoculation of the HER2-CTLA-4 plasmid exerted the strongest inhibitory effect on HER2 expression-mediated tumor growth in mice. These results highlight the potential of the CTLA-4 fusion DNA vaccine as a therapeutic vaccine against colon cancer based on HER2 and CTLA-4.
Introduction
Colon cancer is one of the most common cancers and is an ideal target for immunotherapy.1,2 The majority of colon cancer patients undergo tumor resection and do not present clinical manifestations immediately after surgery. However, a large proportion of colon cancer patients eventually develop disease recurrence in the liver or stomach because disseminated metastasis often remains undetected. Recurrent and metastatic colon cancers exhibit relatively strong resistance against chemotherapy. Thus, there is an urgent need to develop novel therapies for colon cancer, such as immunotherapy. 3 The identification of antigen targets and improvements in gene expression techniques has increased our understanding of the immune system and has significantly advanced tumor immunotherapy. 4 DNA vaccines are highly effective immunotherapy treatments that can induce strong cellular immune response and have consequently attracted the attention of several researchers in the field of tumor immunotherapy.5,6
Carcino-embryonic antigen (CEA) and mucin 1 (MUC1) are currently two of the most widely studied targets for the development of colon cancer immunotherapy vaccines. However, human epidermal growth factor receptor-2 (HER2) is highly expressed in various cancers, including colon cancer, and has become increasingly recognized as a marker for poor prognosis. 7 Although the relationship between HER2 and colon cancer has not been completely elucidated, recent studies have indicated that HER2 plays a key role in the development and progression of colon cancer. 8 Therefore, HER2 shows promising potential as a novel gene therapy target.
DNA vaccines alone have limited efficacy because they usually induce weak cellular immune responses. Therefore, the use of complementary adjuvants may be required to improve the effectiveness of DNA vaccines. 9 Fusion of the extracellular region of cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) with the specific antigen is one of the most effective methods known to improve the immune efficacy of DNA vaccines, particularly in large animals.10–12
In this study, we established a DNA vaccine based on the fusion of the HER2 antigen with the CTLA-4 gene in the same reading frame. The immune and antitumor effects of the DNA vaccine on tumor-bearing mice model stably expressing HER2 were then evaluated.
Material and methods
Construction and expression of recombinant plasmids in mammalian cells
The coding region of the human HER2 extracellular gene (23-652aa; Genbank: X03363.1; UniProt: P04626) and the coding region of the mouse CTLA-4 extracellular gene (X05719.1) were fused with the Furin-2A gene to generate the fusion expression gene HER2-CTLA-4. The resulting HER2-CTLA-4 sequence was sent to Invitrogen for full gene synthesis and then directly cloned into the eukaryotic expression plasmid vector pVAX1 (Figure 1). The recombinant plasmid was designated pVAX1-HER2-CTLA-4. 293T cells were transiently transfected with the recombinant plasmid and the expression was verified. Western blotting was performed to confirm the expression of human HER2 and mouse CTLA-4 using mouse anti-HER-2 antibody (Abcam, ab106674, USA) and anti-mouse CTLA-4 antibody (R&D Systems; FAB434P).

Schematic diagram of DNA vaccines containing the human HER2 and mouse CTLA-4 fusion gene. The coding region of human HER2 extracellular (23-652aa; Genbank: X03363.1; UniProt: P04626) and the coding region gene of mouse CTLA-4 extracellular (X05719.1) were connected via the Furin-2A gene to generate the fusion expression gene HER2-CTLA-4. The designed fusion gene was sent to Invitrogen for full gene synthesis and subsequently cloned into the eukaryotic expression plasmid vector pVAX1.
Experimental mice and tumor cell models
Female BALB/c mice (4–6 weeks old) were purchased from Beijing Vital River Laboratory Animal Technology Co., Ltd. The study design was approved by the Animal Ethics Committee of the Daqing Oilfield General Hospital, China. All experiments were carried out in accordance with the established guiding principles for animal research. The mouse CT26 cell line was purchased from Shanghai Cell Institute (Shanghai, China). To establish a cell model stably expressing human HER2, CT26 cells were transfected with the pcDNA3.1 (+)-HER2 plasmid and then screened using G418 to generate the CT26-HER2 cell line. Confocal microscopy was performed to confirm HER2 overexpression in the CT26-HER2 cells using anti-HER2 antibody as the primary antibody (Abcam, ab108371) and goat-anti-rabbit IgG (phycoerythrin (PE)-conjugated) as the secondary antibody.
Assignment of treatment groups and immunization of experimental animals with recombinant plasmids
Experimental animals were randomly divided into five treatment groups, namely, blank control group, empty vector group, individual antigen group, single adjuvant group, and fusion antigen group. Each treatment group consisted of five to eight mice. Mice were injected with 50 µg of the corresponding recombinant plasmid and immediately stimulated by electroporation. The detailed procedure is as follows. First, recombinant plasmids were intramuscularly injected into the quadriceps muscle of the rear leg, after which the electrode was plugged into the injection site of the muscle for discharge stimulation. The following parameters were used: distance between the electrodes of 5 mm, pulse voltage 60 V, pulse length 50 ms, and six pulse numbers (BTX 830 electroporator, Hollison, USA). 13 Immunization dosage was increased on days 10 and 20 after the first immunization. The immune boosting procedure was performed in full agreement with the primary immunization. Experimental mice were sacrificed on day 14 after the last immunization, and the appropriate immunological tests were performed. Three independent repeat experiments were conducted.
Investigation of humoral immune response
Enzyme-linked immunosorbent assay (ELISA) was performed to evaluate serum-specific antibody response. Briefly, 96-well microplates were first coated with recombinant HER2 antigen (10 μg/mL) and stored at 4°C overnight. After blocking with phosphate buffered saline (PBS) containing 5% skimmed milk powder, the serum was collected, diluted, and added to the 96-well microplates, followed by incubation at room temperature for 2 h. Afterward, samples were incubated with the second antibody horseradish peroxidase (HRP)-conjugated goat anti-mouse IgG; 1:1000 dilution at room temperature for 1 h. The reaction was terminated by addition of 2 M H2SO4, and absorbance at 450 nm was determined on a microplate reader.
Evaluation of cellular immune response
Splenic lymphocytes of mice from each experimental group were isolated on day 14 after the last immunization. Enzyme-Linked ImmunoSpot (ELISPOT) was performed according to the manufacturer’s instructions (Dakewei, China). Briefly, the well plate was coated with the anti-mouse interferon gamma (IFN-γ) mAb at 4°C overnight and subsequently blocked at 37°C for 1 h. Freshly isolated spleen cells (4 × 105 cells/well) were added to 96-well plates, added with antigen stimuli (5 μg/mL HER2), and incubated at 37°C for 48 h. After washing and performing the color reaction, the spots were counted and analyzed using the ELISPOT Reader. The experiment was repeated thrice with the same experimental conditions.
Tumor challenge experiments
A tumor challenge test was performed to investigate the potent antitumor effects of the recombinant plasmids. Briefly, each female BALB/c mouse (4–6 weeks old) was subcutaneously injected with 1.5 × 105 CT26-HER2 cells on the right side of the back. Mice were immunized with the appropriate recombinant plasmid at 3 days post-inoculation with the tumor cells. Immunization procedures were performed as described above. Antitumor activity was monitored by measuring the tumor volume two to three times a week using a vernier caliper. Tumor volume was estimated based on the following formula: tumor volume (mm3) = (length/2) × width2.
Statistical analysis
Data are expressed as mean ± standard deviation (SD). Significant differences between experimental and control groups were analyzed by t-test. P < 0.05 was considered statistically significant.
Results
Expression of recombinant plasmids in eukaryotic cells and HER2 cells in tumor cell models
Expression of the pVAX1-HER2-CTLA-4 plasmid was confirmed in transfected 293T cells added to a six-well plate. Cells were collected at 48 h after transient transfection. Western blotting was performed to confirm the expression of HER2 and CTLA-4 using mouse anti-HER-2 antibody (Abcam, ab106674) and anti-mouse CTLA-4 antibody (R&D Systems; FAB434P). As shown in Figure 2, the cells successfully expressed HER2 and CTLA-4.
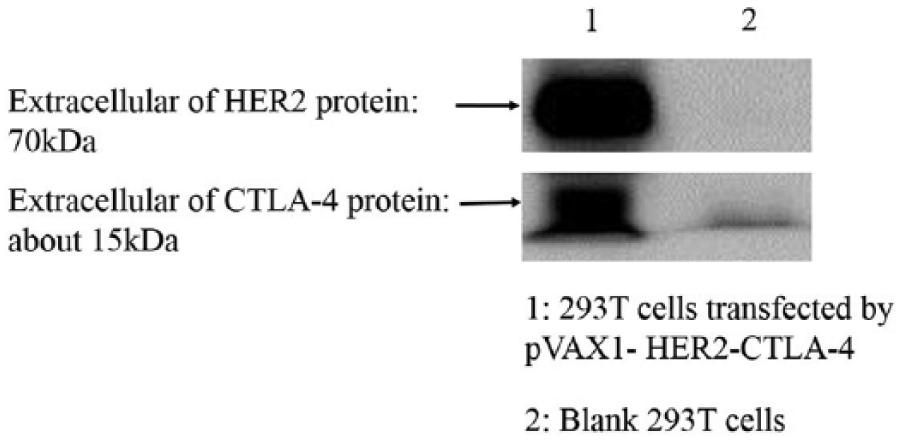
Expression of DNA vaccine in 293T cells evaluated via western blotting. Expression of the pVAX1-HER2-CTLA-4 plasmid was confirmed in 293T cells transfected with the DNA plasmid. Cells in the six-well plate were collected at 48 h after transient transfection. The expression of HER2 and CTLA-4 was confirmed via western blotting using the mouse anti-HER-2 antibody (Abcam, ab106674) and anti-mouse CTLA-4 antibody (R&D Systems; FAB434P).
Confocal microscopy and western blotting were performed to confirm HER2 overexpression in CT26-HER2 cells. As shown in Figure 3, HER2 was expressed on the surfaces of CT26-HER2 cells.
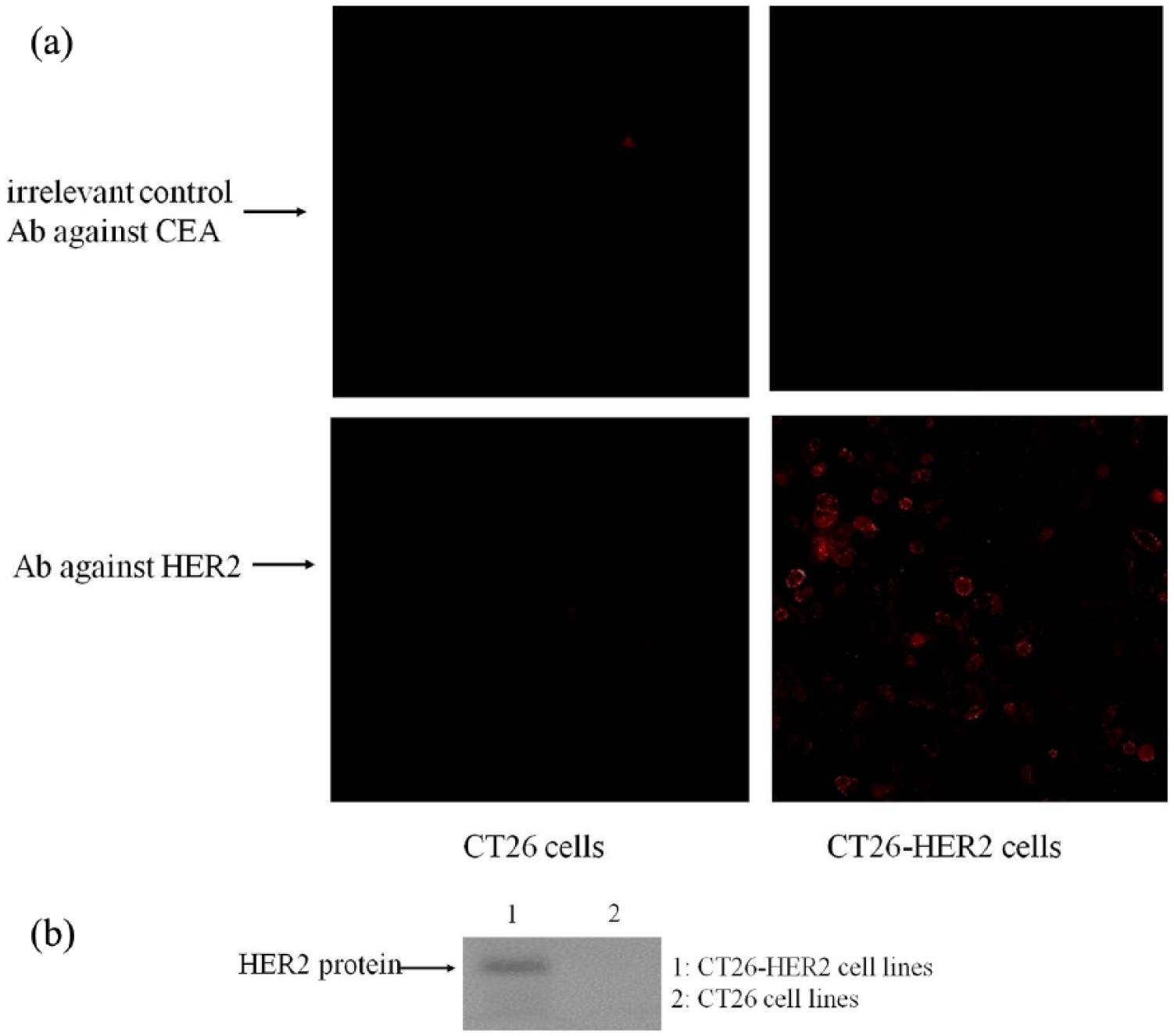
HER2 expression in CT26-HER2 cells. The pcDNA3.1 (+)-HER2 plasmid was transfected into CT26 cells and then screened by G418 to establish a cell model stably expressing HER2. The generated cell line was designated CT26-HER2. (a) Confocal microscopy was performed to confirm HER2 overexpression in CT26-HER2 cells using anti-HER2 antibody as a primary antibody (Abcam, ab108371) and goat-anti-rabbit IgG (PE-conjugated) as the secondary antibodies. (b) Western blotting was performed to confirm HER2 overexpression in CT26-HER2 cells using anti-HER2 antibody as a primary antibody (Abcam, ab108371) and goat-anti-rabbit IgG (HPR-conjugated) as the secondary antibodies.
Investigation of the immune response induced by recombinant plasmids
At 14 days after the last immunization, serum samples were collected from mice in each experimental group and analyzed via ELISA. Results showed that recombinant plasmids harboring the specific antigens could induce the production of target-specific antibodies in laboratory animals (Figure 4(a)).

The DNA vaccine induced antibody and T cell responses in mice. (a) Mice were immunized with the pVAX1, pVAX1-HER2, pVAX1-CTLA-4, or pVAX1-HER2- CTLA-4 plasmids. Anti-HER2 and anti-CTLA-4 antibodies were detected by ELISA. (b) Analysis of the spot frequencies of HER2-specific IFN-γ-producing T cells. Data are expressed as mean ± SD.
ELISPOT assay was conducted to evaluate cellular immune responses. Mice were euthanized at 14 days after the last immunization. Spleen cells were isolated and restimulated with HER2 proteins. As shown in Figure 4(b), mice immunized with pVAX1-HER2-CTLA-4 or pVAX1-HER2 showed higher secretion of HER2-specific IFN-γ, relative to mice in the control groups. Immunization with the recombinant plasmid containing the fusion antigen was found to induce a stronger cellular immune response and resulted in a significantly higher number of spots.
Tumor challenge experiments
A tumor challenge test was performed to investigate whether the recombinant plasmid can induce potent antitumor activity. The antitumor effects of the recombinant plasmids were evaluated primarily based on the size of the transplanted tumors in mice. Tumor growth curves were generated and tumor inhibition rates were calculated.
As shown in Figure 5(a), recombinant plasmids containing the fusion antigen more effectively inhibited tumor growth. In addition, mice immunized with recombinant plasmids containing fusion antigens had the lowest tumor weights, relative to those of mice in the individual antigen group or the adjuvant group (Figure 5(b)). Furthermore, results showed that the recombinant plasmid pVAX1-HER2-CTLA-4 group had the highest tumor inhibitory rate relative to the control group and showed statistically significant difference relatives to those of the control groups.

Antitumor efficacy of DNA vaccines in the tumor mouse model following immunization. At 3 days after injection with tumor cells, mice were immunized with the pVAX1, pVAX1-HER2, pVAX1-CTLA-4, or pVAX1-HER2- CTLA-4 plasmids, respectively. (a) Growth curve of HER2
Discussion
Immunotherapy has recently emerged as a highly active area of research and it plays an important role in the treatment of various diseases, especially tumors. 13 The Food and Drug Administration (FDA) approval of Provenge in 2012 for use in the treatment of prostate cancer represents a milestone in the field of immunotherapy. 14 However, multiple studies have demonstrated that vaccines containing only a single antigenic component are ineffective and do not sufficiently induce strong antitumor immune responses.
This study focuses on improving the immune effects of vaccines using molecular adjuvants. In particular, CTLA-4 and CD152 were mainly expressed on the surface of helper T cells (TH). CTLA-4 is a receptor of B7 molecules, which are primarily expressed on the surface of antigen-presenting cells (APCs). CTLA-4 is known as a negative regulator of T cell activation and competes with CD28 in binding affinity with B7.1/B7.2 molecules. Fusion of the extracellular region of CTLA-4 with a specific antigen is an effective approach for enhancing the immune efficacy of DNA vaccines.12,15
In this study, the mouse CTLA-4 domain was fused with the target antigen to bind murine B7 molecules. This study aimed to determine the effects of the DNA vaccine produced by fusing HER2 with CTLA-4 on the development of colon carcinoma in mice and to elucidate the potential immune mechanisms involved. Therefore, several recombinant plasmids were constructed and intramuscularly injected into the mice, followed by electrical pulses. Immunogenicities of the plasmids were then evaluated. Meanwhile, we established a mouse model of HER2 expression in transplanted tumors to investigate the antitumor immune effects induced by the recombinant plasmids. Experimental results showed that the recombinant plasmid harboring the fusion antigen induced a stronger cellular immune response. HER2-CTLA-4 plasmids exerted the strongest inhibitory effect on tumor growth induced by HER2 expression in mice.
The mechanisms underlying the antitumor immunity induced by therapeutic vaccines have been studied for many years. Currently, the most widely accepted theory is that T cell response (detected via ELISPOT herein) is the body’s primary antitumor defense mechanism, while humoral immune responses play a secondary role (evaluated by antibody detection herein). 16 In addition, tumor growth may be suppressed by either DNA vaccines against CTLA-4 or purified specific anti-CTLA-4 monoclonal antibody. 17 Recent studies have focused on monoclonal antibodies, and an experimental study has combined monoclonal antibodies with PD1 or PDL1 monoclonal antibody. 18 Therefore, our current experimental results indicate that the primary mechanism responsible for regulating HER2 tumor growth is the specific response of anti-HER2T cells. Furthermore, treatment with CTLA-4 DNA vaccine suppressed tumor growth by inducing the production of specific anti-CTLA-4 antibodies. Further in-depth studies will be conducted to examine the effects of DNA vaccines on tumor growth. For example, we aimed to identify which T cell types, particularly CD4 + T cells or CD8 + T cells, can induce the most effective antitumor immune response. Moreover, cytokine secretion will be analyzed. These results highlight the potential of the specific DNA vaccine based on the fusion of HER2 with CTLA-4 as a therapeutic vaccine against colon cancer.
Footnotes
Acknowledgements
S.H. and X.Y. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
