Abstract
Background and aim
Autoimmune hepatitis
Patients and methods
This observational study was conducted at the Pediatric Hepatology Unit at Cairo University Pediatric Hospital, Egypt. All children (<18 years of age) presenting from 2009 to 2016 with established diagnosis of AIH were included. Medical history, clinical examination, and results of investigations were retrieved from patients’ files. The main outcome measures included the rate of remission, relapses, and mortality.
Results
The study included 34 children with AIH. Twenty patients (58%) presented with chronic liver disease. There was a history of concomitant autoimmune diseases in 5 patients. Transaminases were elevated in all patients. There was synthetic dysfunction in 58%. Twenty-four patients (70.5%) had AIH-1, while nine patients (26.4%) had AIH-2 and one patient (2.9%) had autoantibody negative AIH. Piecemeal necrosis was observed in the liver biopsy of 79% of our cohort. Approximately 80% achieved biochemical remission (88% received combined therapy of prednisolone and azathioprine). About half of the patients developed relapses. One patient died of liver cell failure.
Conclusion
In children with liver disease, a diagnosis of AIH should be considered. In those patients, AIH-1 is more common than AIH-2. Prednisolone monotherapy or combined with azathioprine could achieve remission, but relapse is still common. Treatment non-adherence is the main risk factor for relapse.
Introduction
Autoimmune hepatitis (AIH) is a progressive inflammatory liver disorder of unknown etiology. If left untreated, it progresses to liver cirrhosis and liver failure. 1 Diagnosis of AIH relies on the exclusion of other causes of liver disease and the presence of positive clinical, biochemical, and histological criteria. 2
AIH has a very wide spectrum of clinical presentations ranging from being asymptomatic to an acute severe fulminant disease.3,4 It may be associated with other autoimmune disorders such as thyroiditis, type 1 diabetes, vitiligo, inflammatory bowel disease, or juvenile idiopathic arthritis.5,6
Biochemical features of AIH include elevation of serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), and immunoglobulin G (IgG) in addition to autoantibodies. 7 Liver biopsy is recommended in any patient with suspected AIH, 8 where interface hepatitis is the hallmark of the disease. 2
Immunosuppression is the mainstay of therapy in AIH. Prednisone is administered as the initial therapy either alone or in combination with azathioprine. 9 Liver transplantation is indicated in patients who develop fulminant hepatic failure that is unresponsive to corticosteroids and in patients who develop end-stage liver disease. 10
The aim of this study was to determine the clinical, biochemical, histopathological characteristics and treatment outcome of pediatric AIH, in a well-defined, large demographically diverse population of Egyptian children from a single center.
Methods
This retrospective observational study was conducted at the Pediatric Hepatology Unit at Cairo University Pediatric Hospital, Egypt. Children presenting from 2009–2016 with AIH and followed for at least 1 year were included. Data was retrieved from the patients’ files. The study protocol was approved by the Institutional Review Board and the Research Ethics Committee of Kasr Alainy Medical School. The written consent was waived by the Institutional Review Board due to the retrospective nature of the study as the data were collected from the patients’ files. Patients were excluded if they had associated hepatitis C virus (HCV) or de novo AIH after liver transplantation.
Collected data included the following: - Detailed medical history and clinical examination - Investigations: 1) Blood count and liver functions 2) Investigations to rule out other liver diseases included serum alpha 1 antitrypsin, hepatitis A virus immunoglobulin M (HAV IgM), cytomegalovirus (CMV) IgM, Epstein–Barr virus (EBV) IgM with acute presentation, hepatitis B surface antigen (HBsAg), hepatitis B core antibody (HBc Ab), HCV antibody, serum ceruloplasmin, 24 h urinary copper, and slit lamp examination for Kayser–Fleischer rings. 3) Investigations specific for AIH: serum total immunoglobulins and autoantibodies (antinuclear antibody [ANA], smooth muscle antibody [SMA], liver kidney microsomal [LKM-1], antimitochondrial antibody [AMA], and perinuclear antineutrophil cytoplasmic antibody [pANCA]). Autoantibodies were considered positive and clinically significant when present at a dilution ≥1:20 for ANA and SMA or ≥1:10 for anti-LKM-1. 4) Abdominal ultrasound 5) Liver biopsy was done if coagulation profile and platelet count allowed. Data collected included the hepatic architecture, presence of interface hepatitis, bridging necrosis, degree of fibrosis, and histological activity index. In addition, associated biliary involvement in the form of aggression towards bile ducts or picture of associated autoimmune sclerosing cholangitis (ASC) (substantial periductal “onion-skin” fibrosis) were documented. 6) International autoimmune hepatitis group (IAHG) revised diagnostic scoring,
11
simplified criteria for the diagnosis of autoimmune hepatitis,
12
and score criteria for the diagnosis of juvenile autoimmune hepatitis (JAIH)
13
were applied for all patients 7) Patients with evidence of biliary involvement (in the form of jaundice, pruritus, elevated serum bilirubin, gamma glutamyl transferase (GGT), and/or alkaline phosphatase (AP) or aggression towards bile duct in biopsy) were subjected to magnetic resonance cholangiopancreatography (MRCP) to rule out overlap syndrome (AIH/ASC). 8) Investigations to rule out associated autoimmune diseases were performed on patient-to-patient basis on clinical suspicion. - Therapy: Our treatment protocol is as follows: oral prednisolone is initiated in a dose of 2 mg/kg/day for 1 week, then prednisolone was withdrawn by weekly reduction of the dose 2.5–5 mg/week according to the patient’s weight to reach a dose of 7.5–15 mg/day over an average of 8-week period. Further withdrawal was tailored according to the patients’ liver function tests. In patients with inadequate response to steroids after 1 month of therapy, azathioprine was added as a steroid sparing agent in a dose 0.5 mg/kg/day with possible increments as needed up to 2.5 mg/kg/day. Close monitoring of treatment side effects was performed. In the presence of clinical, laboratory or imaging evidence of biliary involvement, ursodeoxycholic acid (UDCA), was administered in a dose of 10–15 mg/kg/day. This treatment protocol adopted in our center is one of treatment options for AIH in children as mentioned in the ESPGHAN Hepatology Committee Position Statement in 2018.
13
- Treatment response: Biochemical remission was defined as normalization of liver enzymes. Immunological remission was defined as normalization of immunoglobulin G and negative autoantibodies.
13
Duration needed to achieve remission was calculated for each patient. Relapse was defined as any elevation of ALT and/or AST after remission (as adopted from the ESPGHAN Hepatology Committee Position Statement in 2018).
13
Relapses were analyzed regarding the possible etiology (withdrawal of steroids or patient non-adherence to treatment), dose of immunosuppression at which relapse occurred, duration, frequency, management, and response to management. Treatment failure was defined as failure to achieve normal liver functions, and reasons for failure were analyzed including erroneous initial diagnosis, the presence of other associated autoimmune disease interfering with proper control of AIH as AIH/ASC overlap syndrome or non-adherence to treatment. - Outcome: Outcome was reported as remission, treatment failure, death, or liver transplantation.
Statistical analysis
Data were analyzed using Excel 2010 statistical package. Mean and standard deviation (SD) or median and interquartile range (IQR) were estimates of quantitative data, while frequency and percentage were estimates of qualitative data. Differences in biochemical characteristics were tested by Student’s paired and unpaired t-test, p values < 0.05 were considered significant.
Results
Over a period of 8 years (2009–2016), 5900 patients presented to the Pediatric Hepatology Unit, Cairo University; 43 of them were diagnosed as AIH, representing 0.73% of the total number of cases. Excluding causes of either acute or chronic liver disease other than AIH was pursued prior to the diagnosis of AIH. Nine patients were excluded from the study due to insufficient data in their files as they dropped out after a short period of follow-up. Therefore, 34 AIH patients were included in our study; 22 were females (64.7%) with a female: male ratio of 1.8:1. The mean age at disease onset was 7.2 ± 2.8 years (range: 2.5–14 years). The mean age at presentation to the Pediatric Hepatology Unit was 8 ± 3 years, ranging from 2.5 to 14 years. The median (IQR) duration of follow-up was 2.75 (3.2) years (range: 1–8 years).
Twenty patients (58.8%) presented with a picture of chronic liver disease, whereas 10 patients (29.4%) presented with acute hepatitic illness and 4 (11.7%) with acute liver failure. Jaundice was the main presenting symptom in 79.4%, followed by hematemesis (8.8%), abdominal distention (5.8%), disturbed conscious level (2.9%), and bleeding tendency (2.9%).
There was a positive history of concomitant autoimmune disease in 14.7%; 4 had systemic lupus erythematosus (one of them had ulcerative colitis) and one had thyroiditis. Family history of autoimmune diseases was reported in 5.8%.
Baseline blood counts and liver function tests in 34 children with autoimmune hepatitis.

AP: alkaline phosphatase, ALT: alanine aminotransferase, AST: aspartate aminotransferase, GGT: gamma glutamyl transpeptidase, INR: international normalized ratio, IQR: interquartile range, IU/L: international unit/liter, Max: maximum, Min: minimum, SD: standard deviation, TLC: total leucocytic count, ULN: upper limit of normal.
HAV IgM and EBV IgM were positive in 2 patients each; their serum aminotransferases did not normalize after 6 months of close follow-up, and the diagnosis of AIH was made in the 4 patients. IgG was elevated in all patients with a mean value of 3002 ± 1122 mg/dl (range: 1148–6520). IgA deficiency was reported in 2 patients (5.9%). SMA was detected in 24 patients (70.6%), ANA in 61.8%, pANCA in 35.3%, LKM in 26.5%, and AMA in 8.8%. Based on autoantibodies, 70.5% had AIH-1, 26.4% had AIH-2. and one patient (2.9%) had autoantibody negative AIH.
By abdominal ultrasound, hepatomegaly was detected in 41.1%. Liver was coarse in 52.9% and bright in 11.7%. In one patient with SLE, there was grade I increase in renal echogenicity. MRCP revealed sclerosing cholangitis in one patient.
Liver biopsy was performed to 19 patients (55.8%). Hepatic architecture was distorted in 16 patients (84.2%), piecemeal necrosis was observed in 15 patients (79%), and bridging necrosis in 6 patients (31.5%). Biliary involvement in the form of inflammatory cells with aggression towards the biliary epithelium was present in 4 patients (21%). According to Ishak score, 14 fibrosis index ranged from 2–5 out of 6 and the histological activity index ranged from 6–13 out of 18.
Autoimmune hepatitis scores in 34 children with autoimmune hepatitis.

AIH: autoimmune hepatitis, ASC: Autoimmune sclerosing cholangitis, IAHG: international autoimmune hepatitis group, JAIH: juvenile autoimmune hepatitis.
aOne patient who was classified as definite ASC was not included in the table.
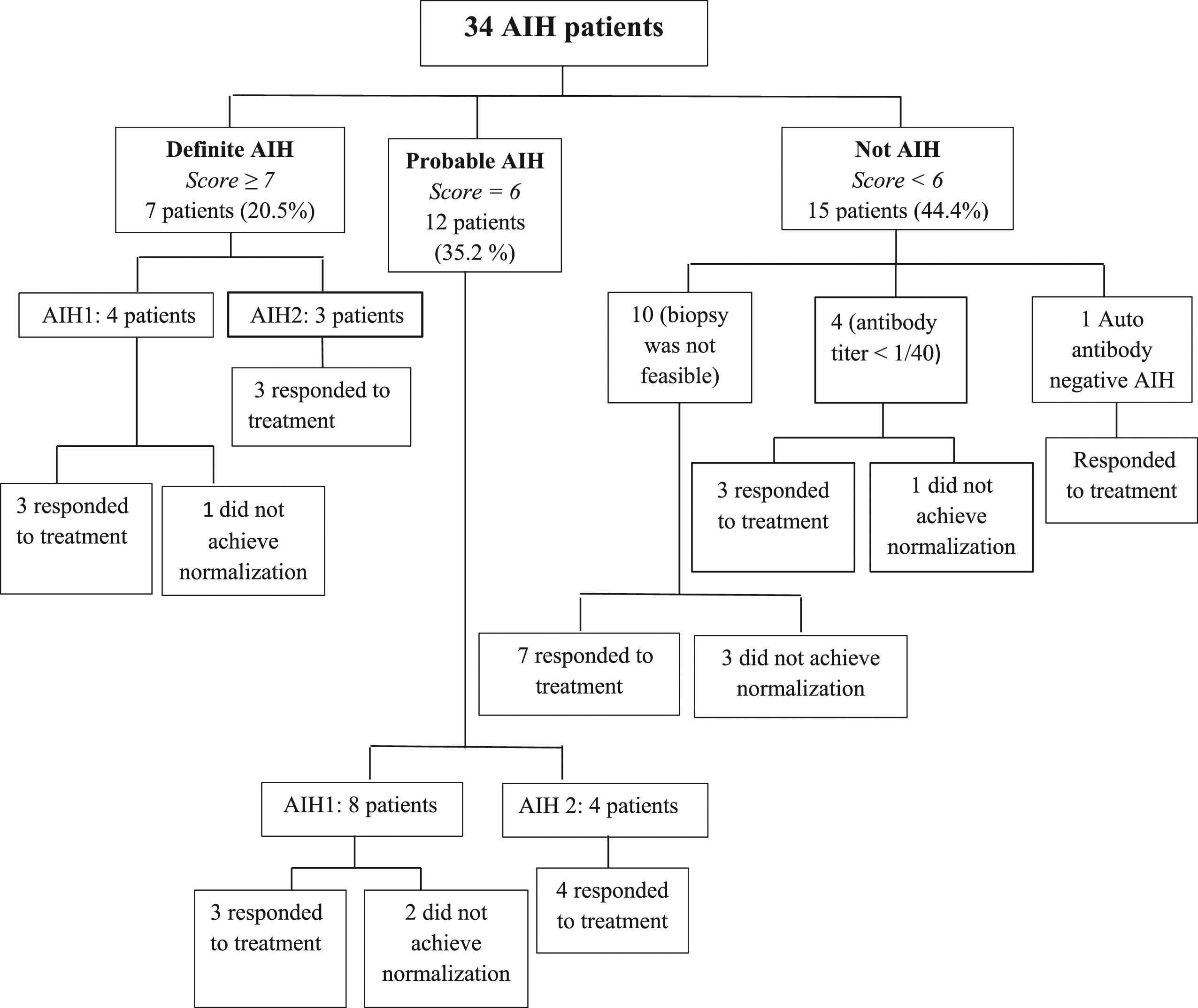
Interpretation of the results of 34 children with AIH according to the simplified criteria for the diagnosis of AIH. AIH: autoimmune hepatitis; AIH-1: autoimmune hepatitis type 1; AIH-2: autoimmune hepatitis type 2.
The initial treatment for our patients with AIH was oral prednisolone. Azathioprine was added to prednisolone in the treatment of 30 patients (88.2%). The duration between initiation of steroids and start of azathioprine ranged from 1–11.5 months. In one patient, steroids were withdrawn completely and he was controlled on azathioprine only without relapse for 1.5 years of follow-up. In patients with elevated biliary enzymes with failure of normalization of transaminases by both prednisolone and azathioprine, UDCA was added. This was the case in 12 patients (35.2%), 8 of them achieved normalization of their liver enzymes after addition of UDCA.
Liver function tests before and 1 month after initiation of immunosuppressive therapy in 34 children with autoimmune hepatitis.

AP: alkaline phosphatase, ALT: alanine aminotransferase, AST: aspartate aminotransferase, GGT: gamma glutamyl transpeptidase, INR: international normalized ratio, IQR: interquartile range, Max: maximum, Min: minimum, PT: prothrombin time, PC: prothrombin concentration, SD: standard deviation, ULN: upper limit of normal range.
*p value is significant.
Biochemical remission was achieved in 79.4%. Patients with AIH-2 achieved a higher rate of remission: 8 out of 9 patients (88.9%) compared to 18 out of 24 patients with AIH-1 (75%); however, the difference was not statistically significant (p = 0.7). The single patient who was seronegative for autoantibodies achieved biochemical remission. In 4 patients who had AIH triggered by HAV and EBV infections, one of each achieved biochemical remission.
The median (IQR) time from start of treatment till normalization of liver enzymes was 6.5 (6.75) months. It was 4.5 (21.5) months in patients with AIH-1 and 7 (148) months in patients with AIH-2. Although patients with AIH-2 needed longer time to achieve normalization of their serum aminotransferases, the difference was not statistically significant (p = 0.38). At time of normalization of liver enzymes, 9 patients (33.3%) were receiving prednisolone only in a dose ranging from 0.2 to 2 mg/kg/day. Ten patients (37.1%) were receiving prednisolone and azathioprine and 8 patients (29.6%) were on prednisolone, azathioprine, and UDCA. Two patients had normalization of transaminases on a dose of 2 mg/kg of prednisolone and achieved a rapid response with normalization of enzymes after 2 weeks of starting induction with steroid monotherapy.
Seven patients (20.5%) did not achieve normalization of their serum aminotransferases (6 with AIH-1 and 1 with AIH-2). Reasons behind treatment failure included treatment non-adherence in 6 patients (85.7%) and one patient had overlap syndrome and ulcerative colitis.
Out of the 27 patients who achieved biochemical remission, 13 patients (48%) developed relapses. The mean age of patients who developed relapses was 10.8 ± 3.7 ranging from 4.5–18 years. Patients who developed relapses were significantly older than patients who did not (p = 0.009). The total number of relapses was 34. The median (IQR) number of relapses per patient was one (2.5) (min: 1, max: 5). The duration between normalization of serum aminotransferases and the first relapse ranged between 1.75 months and 3.75 years. Non-adherence to therapy was the main cause of relapse in 41%. Nine relapses (26.4%) developed secondary to withdrawal of steroids, and in one patient, relapse was secondary to discontinuation of azathioprine as the patient developed bicytopenia.
Regarding patients’ outcome (Figure 2), 27 patients (79.4%) achieved biochemical remission and 4 (11.4%) achieved immunological remission. One patient who had associated SLE died from bleeding esophageal varices, liver failure, and septic shock. None of the patients underwent liver transplantation. All patients who presented with acute liver failure achieved biochemical remission. Outcome of 34 children with autoimmune hepatitis.
Discussion
AIH is a challenging disease. Most studies on AIH in children are predominantly from Caucasian cohorts. Herein, we describe the disease characteristics in Egyptian children.
AIH commonly shows female predominance; females represented 64% of our cohort. Similar results were reported in Canadian and Brazilian populations.15,16 In our study, the age at disease onset ranged from 2.5–14 years, which is younger than that reported in other studies.17,18
There are racial disparities in presentation and outcomes of pediatric AIH. African Americans are more likely to present with end-stage liver disease. 19 Chinese and Brazilian children mostly present with acute hepatitic picture,16,20 while in Pakistani children, the most common presenting complaint was jaundice. 21 In our cohort, about 60% of the patients presented with a picture of chronic liver disease and the rest presented with acute hepatitic illness and acute liver failure. Variable presentations should alert pediatric hepatologists to consider AIH in the differential diagnosis of any of these clinical situations.
Because nearly half of pediatric AIH cases present with symptoms of acute hepatitis mimicking viral hepatitis, 15 exclusion of viral hepatitis is crucial. Autoimmune serology is frequently positive in viral hepatitis, 22 and AIH can be triggered by viral infection. 23 This occurred in 4 of our patients in whom AIH was triggered by HAV and EBV.
History of autoimmune disease in the patient or in a family member can be a clue for the diagnosis of AIH. Up to 40% of patients with AIH could have a family history of autoimmune disorders. 24 Among our cohort, 14.7% had associated autoimmune diseases and 5.8% had a family member with autoimmune disorder. Compared to other studies,15,16 our lower reported percentage may be attributed to the lack of routine screening for associated autoimmune diseases.
Synthetic functions were impaired in more than 50%. We observed a significant improvement of synthetic functions within 1 month of treatment. Synthetic functions can be a guide for response to therapy supporting the diagnosis of AIH especially in the setting of unfeasible liver biopsy.
AIH-1 is more common than type 2.15,16 AIH-1 represented 70.5% of our patients. Seronegative AIH was present in only one patient (2.9%). Seronegative AIH needs a high index of suspicion because in the setting of negative autoantibodies, start of corticosteroids is challenging. 25,26
Scoring systems for diagnosis of AIH are continuously evolving especially in pediatric populations. According to the IAHG revised diagnostic score; 50% of our patients fulfilled a score for definite AIH and 50% for probable AIH. These percentages were similar to that reported by Somroo et al. 21 Regarding the simplified diagnostic criteria, 44.4% of our patients had a score <6; however, most of them responded to treatment. This percentage was higher than what was reported in the study of Arcos-Machancoses et al. 27 (28%) as they included patients with available liver biopsy only. By applying JAIH score to our patients, 53% were not identified as AIH due to the lack of cholangiography. However, 88% of patients whose score <7 responded to treatment.
Frequency of definite AIH among our patients using different scoring systems was less than that reported in other studies and can be explained by several factors. First, HLA is not done on a routine basis in our center due to high costs. Second, some autoantibodies were not available for all patients (SLA, anti-LC-1, and pANCA). Third, extrahepatic autoimmunity and cholangiographic examination were not properly screened in outpatients. Fourth, liver biopsy could not be performed in 44.2% of the patients at initial presentation due to impaired coagulation profile. In addition, typical histological features for AIH as described by Hennes et al.
12
(particularly; emperipolesis and hepatic rosette formation
Biochemical and immunological remission were achieved in 79.4% and 11.7% of our patients (75% of AIH-1 and 88.9% of AIH-2). This was in concordance with the study conducted by Porta et al. 16 (74.7% of AIH-1 and 89.4% of AIH-2).
Relapse is one of the frequent challenges in AIH affecting approximately 40% of patients. 1 Non-adherence to treatment was the main cause of treatment failure as well as relapse among our patients. There are important psychological implications related to the immature age at which the diagnosis of AIH is made and the rebellious attitude of adolescents. Non-adherence is prominent in this age group, with deleterious effects on disease control. 30
Relapse occurred in half of our patients. In the study conducted by Karakoyun, 18 relapse developed in only 9% of patients. This variation is based upon the definition of relapse, where relapse was defined in our study as any elevation of transaminases above ULN (as defined in the ESPGHAN guidelines); meanwhile, it was defined by Karakoyun et al. 18 as a flare of transaminases >two times ULN and EASL guidelines (2015) as ALT elevation>3 times ULN. 31
In spite of the significant findings the current study provides, it contained some limitations. The first study limitation was the insufficiency of some data and diagnostic tests (e.g., cholangiogram) that was mainly related to the retrospective nature of the study. In addition, the unavailability of a liver biopsy to all patients limited its use as a scoring factor in the initial diagnosis, and moreover, histological remission was not assessed in any patient after therapy. Moreover, the small sample size was an additional limitation, which needs further larger study to do regression analysis to confirm some findings such as the relation of non-adherence to the rate of relapses.
Conclusion
AIH is a challenging disease in the pediatric population with diverse clinical characteristics. In children with liver disease, a diagnosis of AIH should be considered. In those patients, AIH-1 is more common than AIH-2. Prednisolone with or without azathioprine could achieve improvement, but relapse is inevitable. Treatment non-adherence is the main risk factor for relapse.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
The study protocol was approved by the Institutional Review Board and the Research Ethics Committee of Kasr Alainy Medical School.
Informed consent
The written consent was waived by the Institutional Review Board due to the retrospective nature of the study as the data were collected from the patients’ files.
