Abstract
Objectives
Chronic ulcerative stomatitis (CUS) is a chronic, ulcerative condition of the oral cavity, clinically and histologically similar to oral lichen planus (OLP), first described as a new disease entity in 1990 by Parodi et al. In this review, 30 years after our first description of CUS, we aimed to systematically review the literature of CUS cases reported ever since.
Methods
We present a systematic review of CUS literature cases, performed in compliance with the PRISMA statement.
Results
Of 125 retrieved articles, 20 satisfied inclusion criteria. These described 76 CUS cases, all presenting orally evident disease: erosions (55%), white lesions (49%), erythema (49%), ulcerations (34%) were the most frequent signs; 54% experienced discomfort/pain. Topographically, buccal mucosa (68%) and gingiva (54%) were the most affected locations, followed by tongue (42%), hard palate (27%), labial mucosa (22%), and widespread involvement (15%). Great diagnostic delay (6.3 years) was evidenced highlighting CUS is an entity too often misdiagnosed. Histopathology found lichenoid features (46%) and non-specific inflammation (54%). Extra-oral involvement was reported in 21%, especially as LP (69%). Of DIF, 97% were positive; 3% negative, compensated by positive IIF, permitting diagnosis. Of patients on steroids, only 12% reported therapeutic success; most steroid-non-responsive patients passed to antimalarials, with 91.66% success when used alone, 100% success in combination therapy.
Conclusion
Dermatologists should suspect CUS in chronic steroid-unresponsive erosive/ulcerative stomatitis. In these cases, to diagnose CUS, the presence of stratified epithelium–specific antinuclear antibodies (SES-ANA) should be investigated through immunofluorescence. Once diagnosed, CUS can be treated with antimalarials, which are an effective treatment contrarily to corticosteroids.
Keywords
Introduction
Chronic ulcerative stomatitis (CUS) is a chronic, ulcerative condition of the oral cavity, first described as a new disease entity in 1990 by Parodi et al. as well as by Jaremko et al.1,2 Both clinically and histologically similar to oral lichen planus (OLP), CUS is defined by the association of chronic oral ulcers and erosions, sometimes surrounded by white striae, with a particular type of antinuclear antibodies (ANA), termed stratified epithelium–specific antinuclear antibodies (SES-ANA), the identification of which, through immunofluorescence, permits to diagnose CUS. Also, characteristic is the low response to corticosteroid therapy, offset by the good response to antimalarials, as well as the frequent association with lichen planus (LP) (-like) cutaneous lesions.1,2
As a result of the clinical similarity to more diffuse and characterized chronic ulcerative mucosal conditions, such as OLP, pemphigus vulgaris, cicatricial pemphigoid, and bullous lupus erythematosus, the diagnosis of CUS is very often significantly delayed.1–3
Also histologically the diagnosis of CUS is challenging: CUS presents often as non-specific or lichenoid mucositis, hardly differentiable from OLP. 3
The diagnostic hallmark of CUS, permitting to differentiate it from the other similar entities, is the presence of SES-ANA, at direct immunofluorescence (DIF) and/or indirect immunofluorescence (IIF) that should be always investigated in chronic, recalcitrant, poorly steroid-responsive oral mucosal ulcerations, to detect a possible CUS. 3
Indeed, CUS should always be suspected in chronic steroid-unresponsive erosive/ulcerative stomatitis and the presence of SES-ANA should be investigated through immunofluorescence permitting diagnosis. Once diagnosed, CUS can successfully be treated with antimalarials, which are an effective treatment contrarily to corticosteroids. Therefore, the need to recognize CUS, diagnose it and treat it correctly.
Historically, since the first description of CUS in 1990, 1 the epithelial antigen involved in its pathogenesis has been investigated: Parodi et al. performed an analysis of the sera from 2 (1990) and 5 (1998) CUS patients discovering circulating antibodies against a mammalian epithelial antigen. As the antigen’s activity resulted affected by DNA-breaking and protein-hydrolyzing-enzymes, it was postulated to be a multimolecular, non-histonic DNA-protein complex.1,4
Meanwhile, Jaremko et al. 2 were the first to refer to CUS-associated ANA, as stratified epithelium–specific ANA (SES-ANA), which they found both in vivo, binding to oral mucosa and skin (DIF), and in serum, binding to epithelial substrates only (IIF).
In 1999, Lee et al. 5 identified the main autoantigen of CUS, a 70 kDa epithelial nuclear protein which they defined “chronic ulcerative stomatitis protein” (CUSP).
Shortly thereafter, Parodi et al. 6 confirmed that antibodies precipitating the 70 kDa molecule were the same antibodies binding to nuclei of epithelial cells.
Ebrahimi et al. identified CUSP as an isoform of p63 protein, namely ΔNp63α. The p63 gene is located on chromosome 3q27-29, encoding six p53-homologous proteins. ΔNp63α is restricted to the epithelium, playing a crucial role in the normal development of oral epithelium and skin. 7
Solomon et al. confirmed CUS patients’ antibodies are directed towards ΔNp63α, with 52% of cases having circulating IgA antibodies, in addition to IgG, though with equal clinical manifestations. Also, they found the immunodominant regions of ΔNp63α are the N-terminal and DNA-binding domains, and antibody cross-reactivity with p53-, p63-and p73-isoforms is limited. 8
Recently, Carlson et al. demonstrated the pathogenicity of SES-ANA in CUS, using 3D human skin equivalent (HSE). 9 They added CUS patients’ sera to HSE, replicating in vivo localization of CUS autoantibodies in nuclei of keratinocytes of basal epithelial layers, obtaining epithelial detachment, the hallmark of CUS. The same result was obtained adding a monoclonal ΔNp63α autoantibody to the HSE, giving double confirmation of the pathogenic role of the autoantibodies in CUS. 9
In short, over time, the autoimmune pathogenesis of CUS has been described: autoantibodies (SES-ANA), mainly IgG, bind to the nuclear protein ΔNp63α (CUSP) located in basal and parabasal layers of stratified squamous stratified epithelium in a speckled pattern, causing a separation of keratinocytes from the basal membrane and from each other, resulting in a clinical ulcer, that is the hallmark of CUS.1,2,8,9
In this review, after 30 years from the first description of CUS, we present a systematic review of the CUS cases reported in literature ever since.
Materials and methods
A systematic review of the literature on published CUS cases was performed in compliance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis, through PubMed and Google Scholar. 10
Combinations of the following MeSH terms and keywords were used to retrieve all the relevant articles: chronic ulcerative stomatitis; chronic stomatitis, and ulcerative stomatitis. To identify eligible articles, we screened the titles and abstracts, and when necessary, full texts. Also, the references of the identified articles were manually screened to include eventually left-over articles.
Inclusion criteria were: articles in English, articles focusing on CUS, case reports, case series, commentaries, and reviews reporting new CUS cases. Conversely, non-English articles, studies reporting a definitive diagnosis other than CUS, and literature reviews without new CUS reports were excluded. Moreover, further analyzing the described cases of initially included articles, we only eventually included only CUS cases with definite diagnosis, not reporting cases with casual SES-ANA positivity and no mucous manifestations.
The series of information extrapolated from the reviewed articles were: year of publication; study site; number of studied patients; gender, age, ethnicity of patients; time for diagnosis; oral cavity involvement site, symptoms and signs; skin manifestations site, symptoms and signs; oral mucosal histology; DIF; IIF; administered therapy; and response to therapy.
Results
A total of 125 articles were retrieved from the literature search, since 1990 up to date. Figure 1 presents a schematic illustration of literature search, study selection criteria and articles selected for the present review (Figure 1). “PRISMA flow-chart.”
“Clinical, histological, DIF and IIF features of studied patients in literature.”

A: asian; B: black; BM: buccal mucosa, BMZ: basal membrane zone, C:caucasian, F: female, G: gingiva, H: hispanic, HP: hard palate, L: lichenoid, LFB: linear fibrinogen at BMZ, LM: labial mucosa, M: male, NSM: nonspecific mucositis, T: tongue, W: oral cavity widespread, x: present,/: not applicable, -: negative.
”Administered therapy and response to therapy of patients in literature.”

BD: betamethasone dipropionate, C: chloroquine, CB: clobetasol, D: dexamethasone F: fluocinonide, FA: fluocinolone acetonide HC: hydroxychloroquine, SLE: systemic lupus erythematosus; MP: methylprednisolone,/:
Articles reported in previous literature reviews not satisfying the above mentioned inclusion criteria were excluded.
Overall, selected articles described 76 CUS patients, with female: male ratio of 14:1, mean age at diagnosis of 61 years, median age of 63 years (86 and 28 years being the extremes).
Ethnic origin was reported for 46 patients: 91.3% were Caucasian, 4.3% African-(American), 2.2% Hispanic, and 2.2% Asian. Interestingly, 13 studies were conducted in the USA, of which 46% in New York; 5 in Europe and only 2 in South Africa.
Time for diagnosis was reported for 40 patients, with a mean diagnostic delay of 6.3 years, median delay of 4 years (0 and 31 years delay being the extremes). Two patients had oral symptoms for an unspecified period of time. Interestingly, this diagnostic delay hints toward a frequent misdiagnosis of CUS, often underdiagnosed and consequently underreported.
Orally, all reviewed CUS cases presented clinically evident disease with at least one clinical sign of CUS. Mostly, more than one clinical sign was present. The most frequently reported signs of disease were: erosions (55%), white lesions (49%), erythema (49%), and ulcerations (34%). We want to remark, however, the difficulty to distinguish between oral erosion, ulceration, and sometimes erythema. The term is most likely used depending on the clinician’s personal interpretation and must be intended as mostly interchangeable, at least for the binomial erosions-ulceration. Of note, 54% of ulcerations and 31% of erosions were associated to white lesions, especially Wickham’s striae-like. Notably, the pattern ulceration/erosion with surrounding white striae seems to be very common for CUS (Figure 2). “Chronic Ulcerative Stomatitis presenting with a typical ulcer surrounded by white striae, on the buccal mucosa (before treatment).”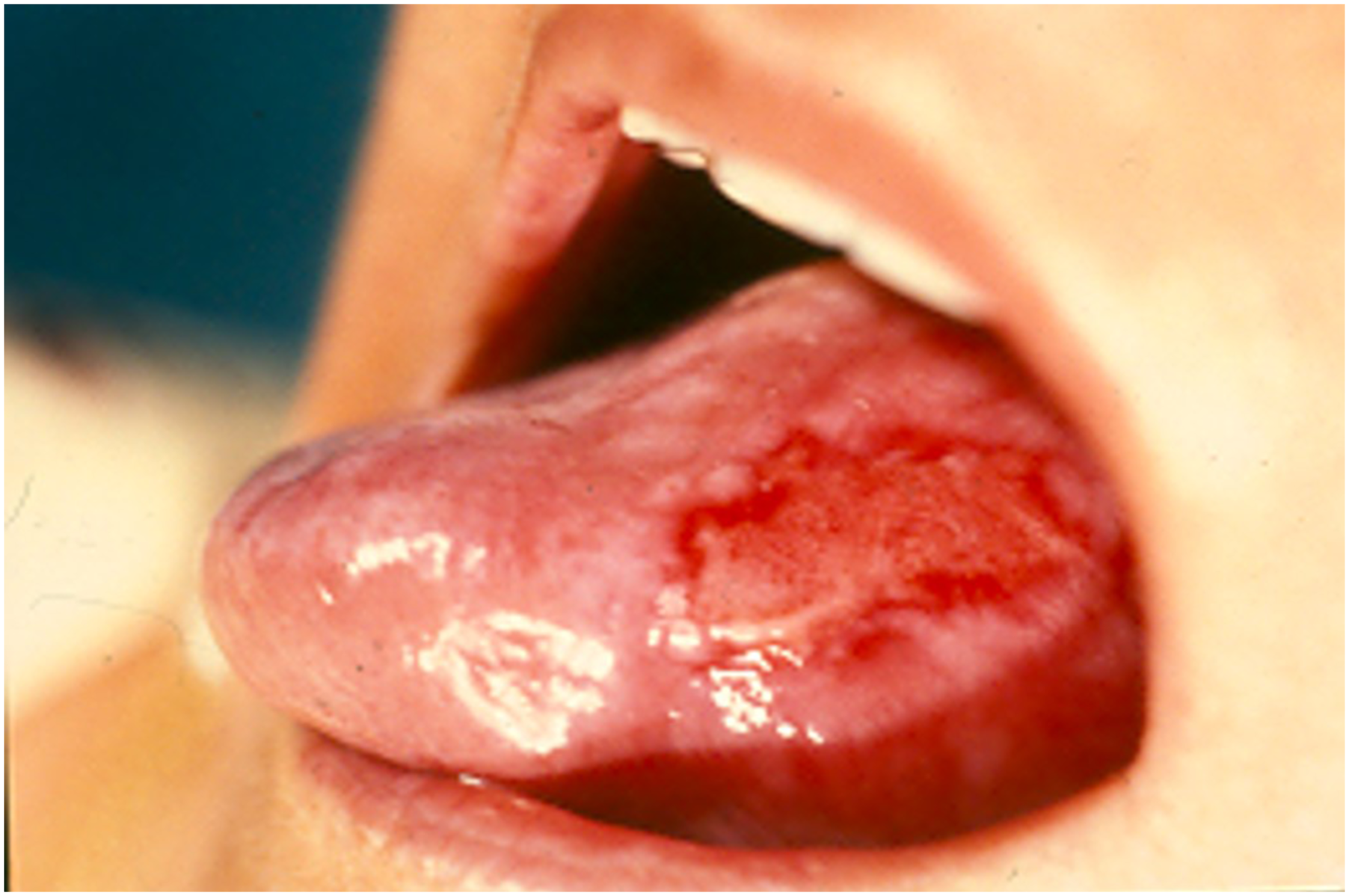
Other reported signs of CUS were: desquamation (11%), xerostomia (4%), vesiculation (4%), and positive gingival Nikolsky’s sign (7%), evidencing the difficulty differentiating CUS clinically from autoimmune bullous disease.
Regarding symptoms of CUS, the review evidenced the majority of patients (54%) experienced discomfort, reported as pain/soreness/tenderness/burning sensation/stinging sensation, and 3% even experienced difficulties eating. Though we expected an even greater percentage of patients experiencing pain or similar symptoms, we cannot totally exclude that some authors did not report the symptoms, focalizing mainly on eye-catching assessable signs of disease.
All cases presented involvement of the oral mucosa: the precise topographic distribution of lesions was reported for 74 patients. The review showed an involvement of the buccal mucosa (68%) and gingiva (54%) as most characteristic. Of 74, 15% had widespread mucosal involvement of oral cavity. Other affected sites were: tongue (42%), hard palate (27%), and labial mucosa (22%); these were mostly (97%, 100%, and 95%, respectively) associated to other topographical lesions, eventually involving the oral cavity mucosa widely. Meanwhile, lesions of the most frequent locations (buccal mucosa and gingiva) were less frequently associated to lesions in other locations (62% and 60%, respectively).
Though the definition of the disease itself implies a predominant oral involvement, also extra-oral cutaneous and mucous involvement was reported in 21% (16/76) of patients, presenting especially as LP. Of these, 69% (11/16) had a confirmed diagnosis of LP or LP-like manifestations. Extraoral involvement consisted especially of disorders of skin, nails, hair, or other mucous membranes such as the conjunctiva.
Oral mucosal histopathologic findings have been reported in 71 cases. Almost half (46%) presented lichenoid features or even diagnosis of OLP; alternatively, bioptic specimens presented non-specific inflammation defined “non-specific mucositis” (54%). It emerges that CUS is histopathologically hardly differentiable from OLP. In fact, a parakeratotic/atrophic epithelium, vacuolar degeneration of basal cells, cytoid bodies, band-like interface infiltrate, saw-tooth rete ridges are typical features of OLP that do, however, overlap with CUS histopathologic features. Only few histologic features may help differentiating CUS from OLP: OLP often presents a sharply defined band-like inflammatory infiltrate, limited to the superficial lamina propria; though cases of uniform infiltrate extending to the whole lamina propria have been described.
Also, a mixed infiltrate of T-lymphocytes and plasma cells is more specific for CUS, whereas OLP typically has a pure T-lymphocytic infiltrate and lacks plasma cells. However, an overlap of the lymphocytic subsets is commonly observed and differentiating CUS from OLP only histologically is not considered a reliable diagnostic method.3,27
The gold standard for diagnosing CUS is DIF: 97% (63/65) of the reviewed DIF tests yielded a positive result, showing the typical SES-ANA IgG speckled pattern in the lower third of the epithelium (Figure 3). “Direct immunofluorescence of CUS showing typical SES-ANA IgG speckled pattern in the lower third of the epithelium.”
Also, 40% (25/63) presented fibrinogen deposition along the basement membrane zone (BMZ), often with extension into the superficial lamina propria, defined as “shaggy appearance.”
Adjunctive DIF signal was observed for IgA (13%, 8/63), IgM (6%, 4/63), and C3 (13%, 8/63). However, most studies did not report whether adjunctive components were analyzed but yielded negative results or whether they were not analyzed at all. Further studies are needed to analyze if these components are of relevance or not.
Of DIF analyzed specimens, 3% (2/65) were negative, but in these cases, IIF was positive, as in Kapinska’s article, who interpreted the result as false negative, because it was taken directly from the lesion without any surrounding unaltered mucosa.14,20 DIF, as all other laboratory analyses, is susceptible of errors. Negative results may be interpreted as false negatives, especially considering that IIF yielded a positive result, confirming the diagnosis of CUS. 20
Also for the 11 cases where DIF was not performed, IIF was positive.
Overall, IIF with patients’serum samples was performed in 36 cases: 97% were positive, 3% negative, in line with the point stated about false negatives and DIF. Notably, IIF was always performed on specific epithelial substrates such as human esophagus/guinea pig esophagus/monkey esophagus/normal human skin.
Regarding therapy, CUS is well known for its poor response to corticosteroids, contrarily to other autoimmune oral stomatitis. 18
In fact, while reviewing literature, it emerged that, of 26 patients initially on steroid therapy, only 12% (3/26) reported therapeutic success (one received steroids and dapsone).
More in detail, administered therapy was reported for 48 patients. Overall, 26/48 (54%) patients were initially on steroid therapy. Of these, one reported inefficacy; nine received secondarily antimalarials; 16 did not receive further antimalarial therapy, though only 2 reported therapeutic success.
CUS is known since the first disease reports for its response to antimalarials. 2
Indeed, patients unsuccessfully treated with steroids, were passed to antimalarials, mostly obtained benefit/complete clearance when used alone (91.66% success) and in combination therapy (100% success).
Antimalarials, mostly chloroquine and hydroxychloroquine in doses of 200 mg/die and 200–800 g/die, respectively, were administered to 67% (32/48) of patients. Overall, 56% (18/32) of patients on antimalarials also received a combination therapy: mostly corticosteroids (89%), with benefit or complete clearance (44%); benefit but slight relapses when tapering (56%). Relapses were mostly controlled by reintroducing therapy. Other combination therapies used were tacrolimus (5.5%) and cyclosporine (5.5%) both obtaining therapeutic success.
Fourteen patients were treated with antimalarials without combination therapy: 58.33% (7/12) achieved good therapeutic success; 33.33% (4/12) obtained remission but relapsed when tapering, mostly controlled by reintroducing therapy; 8.33% (1/12) were unsuccessful; two patients were lost at follow-up.
Tapering therapy emerged as a problem; reintroducing therapy to control disease relapses was needed in 56% of patients on antimalarials in combination therapy and in 33.3% on antimalarials alone. We suppose the latter have a milder disease, which can be controlled only with antimalarials, and relapses less frequently when tapering.
Discussion
Though meanwhile well-characterized, CUS still remains a very rarely reported entity: disease reports do not reach 100 cases worldwide. We suppose the disease is highly underdiagnosed because probably confused with clinically and/or histologically similar entities such as LP, pemphigus vulgaris, cicatricial pemphigoid, and lichenoid mucositis. 3
Especially OLP is often confused with CUS: both entities may present LP (-like-) cutaneous manifestations in addition to the similar oral and histological presentation. 3
Some authors even consider CUS as a variant of OLP, rather than a distinct entity: the ΔNp63α-directed autoimmunity of CUS might be an additional immunopathogenic mechanism involved in LP-epithelial cell damage, defining CUS as LP variant.7–9
Indeed, Cozzani et al. 28 found circulating SES-ANA in LP patients, demonstrating SES-ANA are not a distinctive marker for CUS; however, negative DIF findings suggest SES-ANA are probably an epiphenomenon in LP, irrelevant for its pathogenesis.
In LP, cytotoxic T-cell immune response causing epithelial cell damage is the main pathogenetic mechanisms. Causing probably normally sequestrated nuclear proteins as ΔNp63α to become exposed to the immune system, stimulating an immune reaction towards the self and producing SES-ANA autoantibodies.29,30
We believe CUS is a distinct entity. Not so much for its clinical and histological presentation, that is very similar if not almost indistinguishable from OLP,31,32 but for its characteristic immunopathological characteristics, evidenced in DIF and IIF and for its peculiar response to therapy.
Clinically, CUS is prevalently characterized by erosions and ulcerations, whereas the most characteristic feature of OLP is actually represented by white striae.26,31,32
Histologically, OLP presents sharply defined, band-like, pure T-lymphocytic infiltrate, limited to the superficial lamina propria.31-34 On the contrary, a mixed infiltrate characterizes CUS.3,27 Clinical and histological features are however often overlapping, leading to a blurred distinction of the two entities.
More characteristically, DIF of CUS shows typically speckled SES-ANA in the lower epithelium versus shaggy fibrinogen along the BMZ, which is typical of OLP; only a minority of CUS presents also shaggy fibrinogen along the BMZ.3,27
Above all, CUS must be differentiated from OLP for its low response to corticosteroids,35,36 counterbalanced by the good response to antimalarials.2,3 We believe this is the most relevant point, as relieving patients from disease should be the physician’s main goal, regardless whether CUS can be considered as a distinct entity or as OLP variant. Only diagnosing CUS correctly we may actually treat it, using the antimalarials to which it is responsive.
It emerges from our review that in cases with high CUS suspect, where DIF was negative or not performed, IIF was considered of choice to make diagnosis and always yielded a positive result, permitting thereby the diagnosis of CUS.
Regarding the limitations of this study, they encompass the low number of included CUS cases, probably due to an underdiagnosis of CUS that is only rarely reported in literature.
Also, the wide time period considered for retrieving articles from the literature is a limitation of the study, as CUS was first mentioned as a disease entity more than 30 years ago, in 1990.
Furthermore, it must be mentioned that this review is based on case series and case reports that are articles with a low level of evidence (IV).
Also, no prospective study on the therapeutic effect of antimalarials on CUS is provided, but only retrospective studies, as these are the only studies that are reported in the literature.
Conclusion
We highlight therefore the need to educate dermatologists on CUS, an often misdiagnosed entity, which we believe must be considered in cases of highly steroid unresponsive erosive or ulcerative stomatitis. Indeed, CUS is often mistaken for more common and well-known chronic ulcerative mucosal conditions, such as OLP, 34 as a result of the clinical and histological similarity of these entities.
However, a low response to corticosteroid therapy characterizes CUS, offset by the good response to antimalarials. It is therefore important to recognize CUS cases: only diagnosing CUS correctly, it may actually be treated, using antimalarials to which it is responsive.
The presence of (SES-ANA) at immunofluorescence is diagnostic of CUS. Importantly, only once correctly identified and diagnosed, ulcerations and erosions of CUS can be correctly treated, as CUS can be treated with is highly responsive to antimalarials, but not steroids typically used for oral erosions.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
