Abstract
Although a number of studies have shown that the occurrence and progression of osteoarthritis (OA) is related to endocrine system dysfunction, there is limited evidence about what roles sex hormones play. The aim of the present study was to examine the capacity of 17β-estradiol (ED) and follicle stimulating hormone (FSH) to alter the differentiation of bone marrow (BM) cells in arthritic mice. The experiments were conducted in collagenase-induced osteoarthritis in mice. Cartilage degradation was observed by safranin and toluidine blue staining. Flow cytometry was used to define different BM and synovial cell populations. The influence of FSH and ED on osteoclastogenesis was studied in BM cultures and on the osteoblastogenesis in primary calvarial cultures. The levels of IL-8, TNF-α, FSH, and osteocalcin were estimated by ELISA. FSH increased cartilage degradation and serum osteocalcin levels, while ED abolished it and lowered serum osteocalcin. FSH elevated the percentage of monocytoid CD14+/RANK+ and B cell CD19+/RANK+ cells in contrast to ED which inhibited the accumulation of these osteogenic populations. Also, ED changed the percentage of CD105+/F4/80+ and CD11c+ cells in the synovium. FSH augmented and ED suppressed macrophage colony-stimulating factor (M-CSF) + receptor activator of nuclear factor-κB ligand (RANKL)-induced osteoclast (OC) formation, and this correlated with a respective increase and decrease of IL-8 secretion. FSH did not influence osteoblast (OB) formation while ED enhanced this process in association with changes of TNF-α, IL-8, and osteocalcin production. ED reduced osteoclast generation in bone. The key outcome of the current study is that both hormones influenced BM cell differentiation, with FSH favoring osteoclast formation and ED favoring osteoblast accumulation.
Keywords
Introduction
Osteoarthritis is a progressive complex disease associated with chronic pain and frequently leading to loss of joint functions.1–3 Degeneration of cartilage is regarded as its general sign, but more recently, development of moderate synovial inflammation, affection, and alterations in periarticular tissues has also been found.4,5 Estrogen deficiency, in addition to being one of the most frequent causes of osteoporosis in women, has been reported to increase the risk of OA. 6 This finding implies prevention of OA by estrogens, though the mechanisms involved are not well understood. The prevention of OA by estrogens seems to be due not only to a direct effect on osteoblastic activity, but also to an indirect influence on osteoclasts by downregulation of inflammatory cytokines.7–9 IL-1, IL-6, and TNF-α are released by osteoblasts and participate in pathologic resorptive events.10,11 Studies indicate that estrogens might have a direct and indirect influence on osteoblast formation and on the induction of osteoblast resistance to apoptosis. 12 Osteocalcin is a molecule expressed by osteoblasts that is relevant to the rate of bone remodeling. In patients with OA high osteocalcin serum levels correlate with disease severity and might be used as a specific marker. 13 Osteoblasts are not only responsible for bone formation but also cause bone resorption, through influence on the proliferation, differentiation and fusion of osteoclast progenitors. 14 In vitro studies show inconsistent results, since estrogen has been reported to inhibit, to stimulate or not to have effect on osteoblast proliferation. 15 In many arthritic conditions, the balance between the removal of bone by osteoclasts and the restoration by osteoblasts is shifted to resorptive processes. 16 The influence of FSH on osteoclast function was investigated in vitro and in animal models and the results showed that FSH levels increased bone loss independently of estrogen concentrations. 17 FSH gradually increased 5–6 years before menopause and preceded estrogen decline. 18 While FSH triggered osteoclast differentiation and survival, it had a negligible effect on osteoblast differentiation. FSH receptor was expressed on osteoclast precursors, mature osteoclasts, and mesenchymal stem cells but was not detected on osteoblasts. The blocking of FSH receptor with antibodies inhibited the differentiation of bone marrow macrophages into osteoclasts, an effect that was observed in FSHR knockout mice. 19 FSH influenced the proliferation of osteoclast precursors indirectly via inflammatory cytokines, and directly through the signaling pathways.20,21 Osteoclast generation also required the binding of osteoclast surface receptor activator of nuclear factor-κB (RANK) to RANK ligand secreted by osteoblasts. Depending on the serum FSH concentration in perimenopausal and postmenopausal women it was found that CD14+ monocytes expressed RANK. 22 Ritter et al. 23 showed that FSH and RANKL could enhance the formation of TRAP-positive cells from human mononuclear cells.
Previously, we have established that the administration of 4 μg estradiol/mouse for 30 days in mice with collagenase-induced osteoarthritis resulted in reduced synovitis and cartilage destruction which correlated with attenuated glycosaminoglycan and proteoglycan loss, and osteophyte formation. 24 In the present study we investigated in parallel the capacity of ED and FSH to alter osteoblast/osteoclast balance in arthritic mice.
Materials and methods
Model of collagenase-induced osteoarthritis (CIOA) and treatment
Outbred ICR (CD-2) female mice, 8-10-weeks of age, average body weight 20–24 g (total number = 120), were fed with standard diet and tap water ad libitum. For induction of OA, mice were injected intra-articularly (i.a.) with 2U/10 μl of collagenase from Clostridium histolyticum (Sigma-Aldrich, Germany) assigned as day 0. The incidence of OA was between 80% and 90%. The sample size was calculated by the formula n = logβ / logp where 1-β is the chosen power of 0.05 and p represents the proportion of the animals that do not develop CIOA (20%). A total of nine animals should be examined to have a 95% chance of detecting CIOA that has affected 80% of the animals in the group.
Control groups of mice were i.a. injected with phosphate buffer saline (PBS) and were either untreated (sham, n = 10) or treated with FSH or ED (n = 10 in each group). Four groups of mice were injected with collagenase. One of them was injected i.p. with 0.2 ml PBS for 10 days (CIOA mice, n = 10 in two experiments). Follicle stimulating hormone (FSH, Sigma-Aldrich) was dissolved in sterile phosphate buffered saline (PBS) and injected i.p. in CIOA mice at a dose of 2 IU/mouse/daily (0.2 ml), under two schemes, from day 0 to day 10 (FSH1) or 10 days before arthritis induction (FSH2) of CIOA (n = 10 in each group in two experiments). Thus, we aimed to determine the effect of elevated FSH level on the initiation of arthritis and whether FSH can change the course of CIOA. Estradiol (17β-estradiol, Sigma-Aldrich) was administered orally as described previously. 24 Briefly, 4 μg of substance was dissolved in 0.3 ml sesame oil (Sigma-Aldrich) and mixed with 60 mg Nutella (Ferrero Co, UK). Another group of CIOA mice was treated with ED for 30 days starting with collagenase injection at a dose of 4 µg from day 0 of CIOA (n = 10 per group in two experiments).
Histopathological observations
The joints were fixed for 4 days in 4% paraformaldehyde (pH 7.0), decalcified by incubation in 4% nitric acid for 10 days, and embedded in paraffin so that 5 µm thick sections were cut on microtome (Sakura, Japan) and mounted onto poly-L-lysine coated slides. For detection of glycosaminoglycans joint sections were stained with 1% toluidine blue. For detection of proteoglycans the sections were stained with iron hematoxylin, followed by 0.001% fast green and 1% safranin O. The areas of total and negatively stained cartilage were measured to show glycan distribution and loss. The areas of total and negatively stained cartilage were examined to measure glycosaminoglycan (GAG) distribution and loss. Part of the non-stained picture was removed, so only the image of the tissue section remained as the analyzable area on the white background. Then all the pixels were counted as the image threshold level was set to 15 units and by the use of Adobe Photoshop CS2 (Adobe Systems, San Jose, CA, USA) the amount of staining in the region of interest relative to the whole section was calculated. Two different observers scored the sections and an average of the two scores was used for statistical analysis.
For TRAP staining, the sections obtained as described above were incubated for one hour in 1 mg/ml naphthol AS-TR phosphate (N-(4- Chloro-2-methylphenyl)-3-(phosphonooxy)naphthalene 2-carboxamide), 60 nmol/l NN-dimethylformamide, 100 nmol/l sodium tartrate, and I mg/ml Fast red TR salt solution (Sigma-Aldrich, St. Louis, USA).
Isolation of synovial cells
Patellae with surrounding soft tissue (tendon and synovium) were excised in 200 μl of serum-free RPMI 1640 medium (Sigma-Aldrich, Germany) for 2 h at 37°C. The washouts were collected separately for each animal and after centrifugation at 1200× g for 10 min the synovial cells were counted and used for flow cytometry analyses.
Flow cytometry
Freshly isolated BM cell populations (1 × 105/sample) were washed with 2% FCS/PBS and incubated with antibodies against CD105 (clone MJ7/18, PE-labeled; BioLegend, UK), CD33D1 (clone 33D1; APC-labeled; Biolegend, UK), CD29 (clone HMβ-1; FITC-labeled; Biolegend), FITC-labeled anti-mouse F4/80 antibodies (clone Ba18, Biolegend, USA), FITC-labeled anti-mouse CD3e antibodies (clone 145-2611, Biolegend, UK), FITC-labeled anti-mouse CD11c (clone N418, Biolegend, UK), and IgG isotype controls for 15 min at 4°C. After washing with 2% FCS/PBS the samples were analyzed by flow cytometry. CD14 and CD19 populations were retained by MACS using CD14 or CD19 microbeads (Miltenyi Biotec, Germany). Cells were washed and incubated with antibody against RANK (CD265, clone 6D5; FITC-labeled; Biolegend, USA) for FACS analysis (FACSDiva v6.1.2 Software, Becton Dickinson GmbH, San Jose, CA, USA.
Isolation and culture of murine osteoclast precursors
For 24 h BM cells isolated as described above, were grown with 10 ng M-CSF. Next, non-adherent cells were collected and seeded in complete RPMI-1640 medium with added 30 ng M-CSF for 3 days and then for 3 days with 50 ng M-CSF+50 ng sRANKL (Biolegend, UK). Cell supernatants were collected for ELISA measurements. The cells were stained with a solution consisting 50 mM acetate buffer, 30 mM sodium tartrate, 0.1 mg/ml of naphtol AS-MX phosphate, 0.1% Triton X-100 w/v, and 0.3 mg/ml of fast red violet LB (Sigma-Aldrich, Germany). After 1 h TRAP+ cells were observed microscopically.
Osteoblast differentiation
Bone marrow-derived cells were isolated by flushing femoral bones with complete RPMI-1640 medium (Sigma-Aldrich). The suspension was gently aspirated to disrupt cell aggregates and then centrifuged for 5 min at 1000 rpm. Bone marrow precursors (1 × 106/ml) placed in 24 well plates were cultivated in RMI-1640 medium containing 10% FBS, 2.5 mM HEPES, 5 mM glutamine, penicillin G (100 U/ml), streptomycin (100 μg/ml) (Sigma-Aldrich) for 24 h. Adherent cells 1 × 106/ml were cultivated in six well plates in α-minimal essential medium (α-MEM), containing 10% FBS, penicillin G (100 μg/ml), gentamicin (50 μg/ml), L-ascorbic acid (50 μg/ml), and 5 mM β-glycerophosphate (all from Sigma-Aldrich) for 21 days (mineralization medium) which was half changed every 3 days.
Mouse calvarial cell cultures were isolated from 4-day-old mice. After surgical deletion of calvaria and removal of adherent mesenchymal tissues, the cells were liberated by five sequential incubations in a solution containing 0.05% trypsin-EDTA and 0.1% collagenase (Sigma-Aldrich) for 15 min at 37°C. Cells collected from the second to fourth digestions were plated at a concentration of 1 × 106 cells/ml. After 24 h, media was changed and the cells were further cultivated in mineralization α-MEM. In parallel, part of the cells were cultured in mineralization medium plus TNF-α (20 ng/ml) maintained during days 7–14 or during days 14–21 in the presence or absence of FSH (50 ng/ml) or in the presence or absence of ED (50 ng/ml).
At day 21 BM-derived and calvarial osteoblasts were fixed with 95% ethanol and the mineralized matrix was stained with alizarin red S (2% w/v, pH 4.2, Sigma-Aldrich) for 5 min. The calcification deposits in the matrix appeared dark red. Cells were washed and the stain was extracted with 10% (v/v) acetic acid, neutralized by 10% ammonia, and the absorbance was measured at 492 nm.
ELISA assays
Blood was collected by retro-orbital puncture and was allowed to clot for 1 h at room temperature. The concentration of IL-8 in sera and supernatants separated at day 7 of cultivation was quantified by mouse IL-8 ELISA MAX™ Standard kits (Biolegend, USA), and osteocalcin at day 18, and FSH at day 30 were quantified by ELISA kits (MyBioSource, USA).
Statistical analysis
Data were analyzed by one-way ANOVA, and unpaired t-test, using InStat3.0 and GraphPad Software (La Jolla, CA, USA). Differences were considered significant when P < 0.05.
Results
Effect of FSH treatment on cartilage degradation
At day 30 of arthritis mice were killed and cartilage integrity was observed in different groups shown in Figure 1a. Safranin O staining proved proteoglycan loss in cartilage of arthritic mice increased in mice treated with FSH from day 0 of CIOA. No difference was observed between CIOA mice and those pre-treated with FSH (Figure 1b and d). Progressive loss of glycosaminoglycans (GAGs) was assessed by toluidine blue staining in CIOA group and FSH-treated arthritic mice. A significant difference between CIOA and FSH1 (injected from day 0 to day 10) groups was observed, while FSH2 (injected from day -10 to 0) group showed a similar result with arthritic mice (Figure 1c and d).

Effect of FSH on cartilage degradation. (a) Flowchart of hormone administration. (b) Loss of proteoglycans indicated by black arrows (safranin O staining). (c) Loss of glucosaminoglycans indicated by red arrows (toluidine blue staining). (d) Figure data of (b) and (c). n = 5 joints/group, scale bar = 100 µm. Data are means ± SD from three determinations.
Potential of cultivated BM cells to differentiate into fibroblasts, mesenchymal and dendritic cells
BM cells isolated from sham, CIOA, FSH1, and ED groups were cultivated for 12 days after which the percentage of different cell types was determined. Flow cytometry analyses showed that CD29+ fibroblast population was decreased only in ED-treated mice compared to sham but no difference was found between non-treated and treated arthritic mice as well as between FSH-treated and non-treated groups (Figure 2a and d). The pool of mesenchymal CD105+ cells in BM was strongly reduced in CIOA and in FSH-treated mice compared to sham. This reduction was not significant in regard to ED-treated group but compared to CIOA group mesenchymal cell reduction was lesser (Figure 2b and d). A decrease of dendritic 33D1 population was observed in CIOA mice and in FSH-treated compared to sham, but not in ED-treated once. Also, ED treatment resulted in a lower reduction of dendritic cells compared to FSH-treated (Figure 2c and d). Control group treated with ED showed similar percentages of cell populations like a sham group.
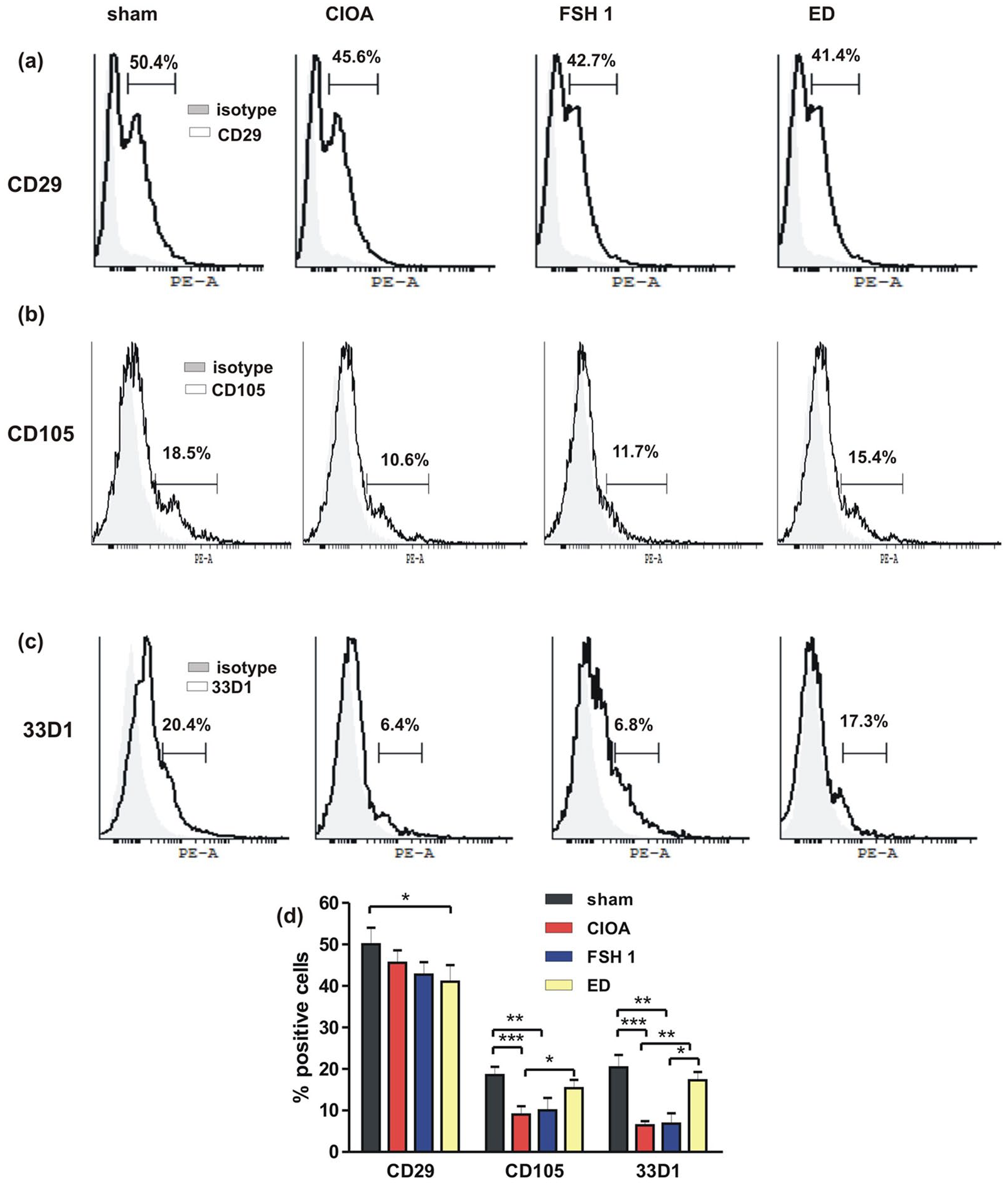
Flowcytometry data. BM cells were obtained at day 30 of CIOA: (a) Percentage of CD29+ fibroblast like cells. (b) Percentage of CD105+ mesenchymal cells. (c) Percentage of 33D1+ dendritic cells. (d) Figure data of (a), (b) and (c). Data are means ± SD from three determinations (n = 10 per group).
Detection of different cell types positive for RANK in BM and synovium of FSH and ED-treated mice
On day 30 of CIOA BM cells were isolated and CD14, CD19, and CD3 were separated, and RANK positive cells were determined by flow cytometry. We found that at the chronic phase of CIOA approximately 53.4% of monocytoid CD14 cells were RANK+. FSH pre-treatment non-significantly increased this to 74.0%, and treatment starting from day 0 of CIOA to 63.3%. The group treated with ED showed a close percentage (10.0%) of RANK+ CD14 population to sham group (5.0%) and drastically lower compared to FSH-treated groups (Figure 3a and c). In regard to CD19 B cells, we observed that 15.0% of them were RANK+ in sham mice, rising to 45.2% in arthritic mice. FSH treatment from day 0 resulted in an increase of RANK+ CD19 cells up to 75.2% and non-significant increase (64.5%) was observed in FSH pretreated group compared to CIOA group. In ED-treated mice the increase of CD19 RANK positive cells (24.4%) was non-significant compared to sham group, while compared to CIOA and FSH-treated groups a strong decrease was observed (Figure 3b and d). The increased percentage of CD3e RANK+ cells in CIOA mice was diminished by FSH and ED treatment. But it should be noticed that the percentage of double positive cells was minor and the differences between groups, although statistically significant, varied by approximately 2% (Figure 3e).

Flow cytometry data. BM cells were obtained at day 30 of CIOA. (a) Percentage of CD14 RANK positive monocytoid cells. (b) Percentage of CD 19 RANK positive B cells. (c) Figure data of (a). (d) Figure data of (b). (e) Figure data of flowcytometry analysis of CD3 RANK positive cells. Data are means ± SD from three determinations (n = 10 per group).
At initial phase of CIOA in the synovial fluid of sham, CIOA mice and CIOA mice treated with ED FACS analysis showed the elevation of CD19+/RANK+ and CD3+/RANK+ cells in arthritic mice compared to sham. In contrast, these populations in ED treated mice were not significantly different from sham group (Supplemental Figure S1b and c).
Influence of FSH and ED on M-CSF+RANKL-induced osteoclast formation and on in vivo osteoclast generation
BM cells isolated from sham, CIOA, FSH 1, or ED groups were stimulated with M-CSF and RANKL. After 6 days TRAP positive multinuclear cells were counted. The number of these cells was higher in CIOA group compared to sham group and further, FSH increased TRAP positive cells, while ED decreased their number (Figure 4a and b). Also, the concentration of IL-8, one of the main cytokines involved in osteoclast differentiation, was assessed. Data showed that the development of CIOA was attended with generation of pre-osteoclasts in BM predisposed to osteoclast accumulation after appropriate stimulation. This process was augmented by FSH and in contrast, it was inhibited by ED (Figure 4c). Control group treated with ED showed similar percentages of cell populations like sham group.

Effect of FSH and ED treatment on osteoclastogenesis. (a) TRAP stained BM multinucleated osteoclasts after stimulation with M-FCS+RANKL at day 7 of cultivation. (b) Number of TRAP positive cells at day 7 of cultivation. (c) IL-8 level in the supernatants at day 7 of cultivation. Data are means ± SD from five determinations.
At day 30 of CIOA joint sections obtained as described above in Materials and methods were TRAP stained. A presence of TRAP positive cells was found in CIOA mice deep in the bone that was reduced in ED mice (Supplemental Figure S1a).
Influence of FSH and ED on osteoblastogenesis
BM cells obtained from sham, FSH 1 and ED-treated groups were cultivated for 21 days in a mineralization medium. Supernatants collected on day 14 were analyzed by ELISA in order to determine the release of IL-8 and osteocalcin. In the FSH group, the concentration of both molecules was increased, while in ED-treated group their concentration was decreased (Figure 5a and b). FSH did not significantly changed TNF-α production, while in ED-treated group TNF-α release was strongly inhibited (Figure 5c). Linear analysis at day 21 of cultivation showed that cytokine level correlated with the degree of calcification, according to alizarin red staining in ED group (Figure 5d) but such correlation was not found for FSH group (data not shown). After day 14 alizarin red stained cells appeared, these until day 21 formed large osteoblast nodules to a similar degree as in the cultures from CIOA mice and CIOA mice treated with FSH 1, but their number was increased in the cultures from ED-treated mice (Figure 5e and f).

Effect of FSH and ED on oteoblastogenesis. (a) IL-8 concentration in the supernatants of BM cells at day 14 of cultivation in mineralization medium; scale bar = 250 µm. (b) Osteocalcin level in the supernatants of BM cells at day 14 of cultivation in mineralization medium. (c) TNF-α concentration in the supernatants of BM cells at day 14 of cultivation in mineralization medium. (d) Linear analysis showing the correlation between TNF-α and the degree of calcification at day 21 of cultivation. (e) Alizarin red staining at day 14 and at day 21 of cultivation. Black arrows showed osteoblast nodules releasing matrix. (f) Number of osteoblast nodules at day 21 of cultivation. Data are means ± SD from three determinations (n = 10 per group). *P < 0.05; **P < 0.01, ***P < 0.001, one-way ANOVA. (g) Number of osteoblast nodules at day 21 of calvarial cells’ cultivation in the presence of TNF-α from day 7 to day 14 or from day 14 to day 21 simultaneously with FSH or ED.
Direct action of TNF-α on osteoblastogenesis was studied in primary calvarial cultures. The cells were cultivated in the presence of TNF-α at a concentration of 20 ng/ml from day 7 to day 14 or from day 14 to day 21. After this seven days’ period of exposure to cytokine cells remained attached and viable as shown by trypan blue staining. Data showed that TNF-α inhibited osteoblast generation and calcification if added at the initial phase of OB differentiation and more weakly if added at the point of phenotype selection. FSH did not influence the effect of TNF-α, while ED abrogated it to great extent TNF-α-induced inhibition of osteoblast generation caused at the initial phase but did not change osteoblast formation if TNF-α was added after day 14 (Figure 5g).
Influence of ED on different cell populations in the synovial fluid at the initial phase of CIOA
On day 7 of CIOA synovial fluid collected from sham mice, mice with CIOA and mice with COA treated with ED (4 µg/mouse/daily) was centrifuged, washed and the cells were counted and used for FACS analyses. Estrogen prevented the generation of double positive CD105+/F4/80+ compared to untreated arthritic mice (Figure 6a and b). No differences between groups were noticed in regard to CD29 positive cells (approximately 20% of total synovial cells) (Figure 6c and d). Elevation of the percentage CD11c positive cells was observed in mice with CIOA significantly inhibited in ED treated group (Figure 6e and f).

Flow cytometry data. Synovial cells were obtained at day 7 of CIOA. (a) Percentage of CD105+/CDF4/80+ cells and figure data (b). Percentage of CD29+ cells (c) and figure data (d). (e) Percentage of CD11c+ cells and figure data (f). Data are means ± SD from three determinations (n = 10 per group).
Influence of FSH and ED on serum osteocalcin and FSH levels
At day 18 of CIOA blood from sham mice and arthritic mice treated or non-treated with FSH or ED was collected. Data showed an increase of serum OC of CIOA group additionally enhanced as a result of FSH injection from day 0. When FSH was administered for 10 days before arthritis initiation OC levels were similar to those of CIOA group. ED group did not show significant difference compared to CIOA group and a decrease of serum OC was observed, compared to FSH-treated mice. Mice treated with FSH or ED were included showing no significant difference with sham group (Figure 7). FSH treated mice showed no significant difference in their serum level of FSH at the end of the experiment (day 30, Table 1).
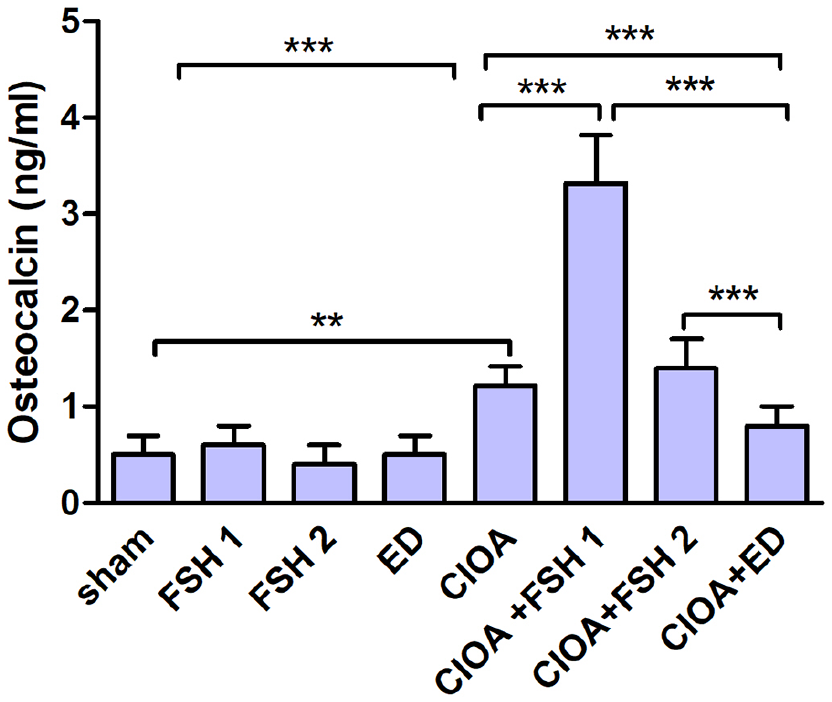
Serum osteocalcin level at day 18 of CIOA. Data are means ± SD from three determinations (n = 10 per group). Mice treated with FSH from day 0 to day 10 (FSH1); mice treated with FSH from day −10 to day 0; mice treated with ED (ED); CIOA mice treated with FSH under both schemes (CIOA + FSH1 and CIOA + FSH2); CIOA mice treated with ED (CIOA + ED). Data are means ± SD from three determinations (n = 10 per group).
Serum FSH levels (ng/ml).

Data are from 1 or 2 experiments, n = 10 per group.
Discussion
Taking into account that unbalanced osteoclast-mediated bone destruction is inherent for many joint disorders it is of major importance to reveal the exact pathways of osteoclastogenesis. Osteoclasts play together with bone restoring osteoblasts during skeletal remodelling. However, it is considered that postmenopausal osteoporosis is directly dependent on the declining levels of estrogen and can be reversed by estrogen treatment. 25 The present results indicated that FSH and ED expressed opposite effects on the osteoclastogenesis in vivo and in vitro. Based on the fact that the cell surface expression of RANK is a feature of the committed pre-OC we aimed to assay the percentage of different RANK positive cell populations in the bone marrow. The results clearly showed that in the chronic phase of CIOA CD14+ monocyte populations and CD19+ B cell populations represent a great part of RANK+ pool that might be involved in bone remodeling. FSH can accelerate cartilage and bone resorption through a strong elevation of CD14+/RANK+ and CD19+/RANK+ cells when the treatment started with arthritis initiation. The Ten-day application was enough to achieve this effect, while no effect was observed when mice were prophylactically treated before CIOA initiation. In regard to ED, previously we have established that estrogen improved the course of CIOA including decreased cartilage destruction and osteophyte formation. 24 Current results delineate some of the mechanisms of ED action using the same treatment scheme. Hormone prevented the accumulation of osteogenic RANK positive monocytoid and B cell populations which can reside at the sites of joint inflammation, after their release from BM. The same effect of ED was found in regard to CD19+/RANK+ cells in the synovium at the initial phase of CIOA, which confirmed such possibility. B cell depletion therapy with rituximab inhibits bone resorption and increases bone formation in RA patients pointing to the pathogenic role of B cells in bone homeostasis. 26 Also, activated RA B cells participate in osteoclastogenesis in vitro in a RANKL-dependent manner. 27 RANK and RANKL are important regulators of interactions between T cells and dendritic cells. 28 Concerning T cells, a minor population of CD3e+/RANK+ cells were detected, suggesting its negligible role in osteoclast formation at the chronic phase of CIOA. Probably, such CD3e+/RANK+ cells might be generated out of BM under specific conditions, like locally increased pro-inflammatory mediators. In fact, in the synovial fluid at an early stage of arthritis, a low percentage of CD3e+/RANK+ cells was observed, but not in ED treated mice. Histopathological data showed that the high percentage of RANK positive cells in BM of FSH-treated mice correlated well with cartilage degradation as a result of intensive glucosaminoglycan and proteoglycan loss. Fibroblasts are the major cells in the stroma of tissues that produce extracellular matrix molecules like collagen. 29 Mesenchymal stem cells express multilineage differentiation potential and morphologically, they can be hardly distinguished from fibroblasts. 30 While MSCs possess immunostimulatory properties fibroblasts are considered to be mediators of inflammation. 31 In the bone marrow, MSCs constitute an important component of the hematopoietic stem cell niche and have been shown to support hematopoiesis both in vitro and in vivo. 32 To date, no single MSC-specific marker exists. In addition, freshly isolated and culture-expanded CD105 cells show different expression of CD29, CD44, CD90, and CD106. 33 Conditionally, we considered CD29+ cells as like fibroblasts and CD105+ cells as mesenchymal once. In this context, the development of CIOA did not change the percentage of fibroblasts in BM but lowered the percentage of MSCs. In the synovial fluid there was similar percentages of CD29 positive cells in sham, CIOA, and ED treated mice. FSH did not influence CD105 expression while ED prevented the decrease of MSCs pool. It can be speculated that estrogen might have positive effect on arthritis because of the elevated influx of MSCs thus supporting reparative processes in the joint. The role of hematopoietic CD105 cells was proven by the presence of double positive CD105+/F4/80+ population in the synovial fluid at the acute phase of CIOA. Such cells were nearly absent in sham and ED treated mice which supposed potential anti-inflammatory effect of ED expressed in the early arthritis. DCs are professional antigen-presenting cells that activate T cells and unlock various immune responses. They share common precursors with osteoclasts depending on the environment in the BM, participate in bone resorption. 34 CIOA decreased the percentage of DC expressing the most common marker 33D1 in BM which was not influenced by FSH. This pointеd on the accelerated release of DC that if find an appropriate osteoclastogenic environment in arthritic individuals can participate in bone resorption. The CD11c is one of more restricted markers for DC, although it is not expressed on all DC, and it may also be expressed on other cell types. 35 In the synovial fluid we observed an increase of CD11c in the acute phase strongly reduced in ED treated group. ED attenuated the decrease of DC populations thus in regard to arthritis it means lower possibility for osteoclast number of mature osteoblasts and also depends on their bone-forming capacity. The surface markers and function of bone marrow cell populations may differ from that in the synovium and articular cartilage, and can influence in different manner cartilage erosion and osteophyte formation. When resorptive process is developed, osteoblasts are recruited to this site, where they start to synthesize and mineralize new matrix. 36 Osteoblastogenesis includes three phases defined as proliferation, matrix maturation and mineralization, each one characterized by the expression of different surface markers like ALP and osteocalcin, as well as by production of different cytokines including TNF-α, IL-1, IL-6 and IL-8.37,38 We compared the ability of BM cells obtained from untreated or treated with FSH and ED mice cultivated in mineralization medium to form osteoblast nodules and mineralization matrix. The results imply that the estrogen status of animals prior to culture can alter the response to estrogens. It was observed that in FSH-treated mice the release of IL-8 and osteocalcin were elevated around day 14 when started osteoblast formation, while ED treatment caused an inhibition of IL-8 and osteocalcin production, proving that they played an important role for osteoblast generation. The same tendency was noted in concern to TNF-α production: an increase in FSH1 group and a decrease in ED-treated group but in the late phase of mineralization.
The involvement of ED in the processes of osteoblastogenesis showed that it was correlated with the presence of TNF-α in the environment. In primary calvarial cultures, the addition of TNF-α inhibited the initiation of osteoblast accumulation and mineralization that was diminished in the presence of ED. At the same time, the addition of FSH did not have a significant effect on osteoblast generation. We observed correspondingly that the impact of both hormones is related not only to the direct effects on osteoblastic activity, but also to the indirect effects on osteoclasts through a change in IL-8 secretion. The increase of the number of TRAP+ cells in FSH1 group correlated with higher IL-8 secretion, and a decrease of TRAP+ cells in ED-treated group correlated with inhibition of IL-8 secretion. The anti-arthritic effect of ED was observed and in vivo proven by the reduced formation of osteoclasts in bone seen at the late stage of CIOA (day 30). One of the most frequently used markers of the osteoblast differentiation is osteocalcin. At late chronic phase of CIOA the negative bone-resorbing effect of FSH treatment was associated with increased serum OC levels. In case of ED treatment, as it can be expected, the decrease of serum OC was relevant to the improved CIOA development. We administered FSH before CIOA initiation and at the early stage of arthritis development. Data showed that the elevation of hormone level influenced the early processes of joint inflammation. It enhanced osteoclastogenesis even after its withdrawal as evident from the similar serum FSH level, 20 days post treatment. Further, the effect of FSH on the established CIOA (after day 18) should be investigated to define its arthritogenic role.
Conclusion
The imbalance of hormone levels leads to altered bone structure and dynamics during menopause and aging. 39 In fact, increased bone loss can be due to the high FSH serum levels during menopause while estrogen serum levels are still normal. 40 The effects of FSH and ED on bone remodeling should be considered with regard to the immune cells and inflammatory cytokines. 41 Detection of more specific mesenchymal, fibroblast and DC markers will help detailed description of FSH and ED effects on osteoarthritic processes. Present results demonstrate that FSH might trigger CIOA progression toward the osteoclast phenotype while ED might act as a potent inhibitor of bone resorption. It was associated with altered bone marrow cell differentiation and involvement of pro-inflammatory cytokines TNF-α and IL-8 as well as osteoblast-specific matrix protein osteocalcin. It should be noted that all the experiments were conducted in non-ovariectomized mice in active age. Thus, the administration of both hormones might increase their levels above physiological. The consequences of this on ovarian functions need to be further investigated.
Supplemental Material
sj-pdf-1-iji-10.1177_20587384211016198 – Supplemental material for Follicle stimulating hormone and estradiol alter immune response in osteoarthritic mice in an opposite manner
Supplemental material, sj-pdf-1-iji-10.1177_20587384211016198 for Follicle stimulating hormone and estradiol alter immune response in osteoarthritic mice in an opposite manner by Lyudmila Belenska-Todorova, Ralitsa Zhivkova, Maya Markova and Nina Ivanovska in International Journal of Immunopathology and Pharmacology
Footnotes
Author contributions
L. B-T and R.Z. performed the main experiments, M.M. designed the manuscript and compiled the data and N.I. supervised the study and revised the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Fund, Republic of Bulgaria (Grant DN 13/6/15.12.2017).
Ethics approval
Ethics approval for this study was obtained from Bulgarian Food Safety Agency (Protocol №352/06.01.2020) and approved by the local Animal Ethical Committee at the Institute of Microbiology, Bulgaria.
Animal welfare
All animal procedures followed international, national, and Institute of Microbiology guidelines for humane animal treatment and complied with relevant legislation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
