Abstract
Uterine fibroids (UFs) are one of the most common pathologies of the female genital tract. The incidence of UFs has been estimated at 25–80%. Tumor necrosis factor (TNF)-α is a cell-signaling protein involved in systemic inflammation and is one of the cytokines responsible for the acute phase reaction. The aim of the study was to evaluate the impact of clinically symptomatic UFs on TNF-α serum levels. A total of 149 Caucasian women were included: 85 patients admitted for surgery due to clinically symptomatic UFs (n = 85; study group) and 64 age-matched UF-free controls (n = 64). TNF-α serum concentrations between the groups were compared. Receiver operating characteristic (ROC) curves were also used as a statistical model to evaluate TNF-α as a marker for UFs. Mean TNF-α serum concentration in the study group was 0.34 ± 0.14 pg/mL; (in half of the subjects, the level did not exceed 0.39 pg/mL. Mean TNF-α serum concentration in the control group was 0.17 ± 0.09 pg/mL; in half of the subjects, the level did not exceed 0.14 pg/mL. The difference was statistically significant. Using the area under the ROC curve, we found that TNF-α serum concentration of 0.34 pg/mL can be used as a predictor for UFs in selected populations. In our study, we confirmed higher TNF-α serum concentrations in women with clinically symptomatic UFs.
Introduction
Uterine fibroids (UFs) are one of the most common pathologies of the female genital tract. Regardless of their benign nature, these tumors may also be the source of various health complaints. 1 The risk factors for UFs have been extensively described and include: age, low parity, Black race, obesity, positive family history, and genetic predisposition. 1 The ultimate goal is to identify both groups of patients who are at an increased risk for UFs. 2
According to various authors, fibroid metabolism is affected by steroid hormones, growth factors, and cytokines.3,4 Paracrine signaling plays an important role in cellular transformation of the myometrium. Initiators of fibroid formation remain to be fully elucidated, but estrogen and progesterone are believed to be the major promoters of fibroid growth. 4 Their effect on the development of UFs is inseparably related to the aforementioned factors like cytokines and chemokines.3,4 Various cytokines play a key role in the regulation of inflammation, neoangiogenesis, and tissue remodeling. Cytokines may be responsible for symptoms associated with UFs, such as pain, infertility, and obstetric pathologies. 5
Although many interleukins are involved in UF biology, tumor necrosis factor (TNF)-α appears to be one of the most important myometrium-associated cytokines. It is one of the cytokines responsible for the acute phase reaction. It is produced mainly by activated macrophages, but it can also be produced by other cell types. 6 Multiple biological functions of TNF-α throughout the body have been identified, including: fever, promotion of adhesion molecule expression, phagocytosis stimulation, appetite suppression, and modulation of insulin resistance. 6 Increased TNF-α expression has been found in fibroid tumors in comparison to the adjacent normal myometrium. 7 TNF-α secreted by adipocytes enhances UF proliferation. 8 Dysregulation of TNF-α production and distribution has been demonstrated in various human diseases, including cancers. 5 Therefore, it is of interest to determine the role of TNF-α in the biology and therapy of UFs.
Objectives
The main objectives of the study were as follows:
Evaluation of the impact of clinically symptomatic UFs on TNF-α serum concentrations;
Creation of a statistical model which, taking into account TNF-α serum concentration, could be used to determine whether a patient is UF positive or negative.
Material and methods
The study group included women with the diagnosis of clinically symptomatic UFs, who were referred to the First Department of Obstetrics and Gynecology, The Medical Centre of Postgraduate Education in Warsaw, Poland. The control group comprised women without ultrasonographically identified UFs, who were recruited from a gynecological outpatient clinic. Local Ethics Committee approved of the study. Informed consent was obtained from all participants. A total of 149 Caucasian women were recruited for the study. Women were deemed eligible for inclusion in the “study group” if transvaginal ultrasound revealed at least one UF (mean diameter of ≥10 mm). Selected women provided serum samples which were tested for TNF-α serum concentration. Difficulty with visualization of the lesion or suspected adenomyosis excluded the subjects from the study group. All women who presented any signs of active inflammation (signs of infection like fever, cough, sneeze) were excluded. All surgical patients were additionally qualified by the anesthesiologist. Exclusion criteria for both groups were as follows: active inflammation or allergy, fever, rheumatoid arthritis, systemic lupus erythematosus, inflammatory bowel diseases, postmenopausal status, malignancy, or pregnancy.
All women underwent ultrasound scanning (transvaginal or transabdominal, if necessary for better visualization). The ultrasound exam was conducted by a sonographer certified in genital tract sonography. UF was defined as a hypoechogenic and heterogenous area with distinct margins. Blood samples were collected from all patients for biochemical analysis. Venous blood samples were collected and the plasma was separated; aliquots were frozen at −80°C until needed for assays. Basal plasma levels of TNF-α were measured in duplicate in a single assay using a high-sensitivity enzyme-linked immunosorbent assay kit (Invitrogen, USA). The assay sensitivity was 0.09 pg/mL. The intraassay coefficient of variation (CV) was <5.5%.
Methods of statistical analysis
Parameters measured on the nominal scale were characterized by cardinality and percentages; parameters measured on the quotient scale were characterized using mean, median, lower and upper quartiles, standard deviation, and volatility. Group differences were analyzed by Mann–Whitney, Student’s t-tests, analysis of variance (ANOVA); individual group differences were analyzed by the Bonferroni test. Correlations between serum TNF-α levels (pg/mL) and total volume of UFs (cm3) were assessed by Spearman’s rank correlation test. The volume of all fibroid lesions was determined according to the prolate ellipse formula (a × b × c × 0.523), where a is height, b is width, and c is depth. Evaluation of the usefulness of the diagnostic test was performed taking into account the number of true-positive (TP), false-positive (FP), false-negative (FN), and true-negative (TN) results. In addition, the basic test accuracy measures were investigated: sensitivity, specificity, and related statistical calculations are also presented in the manuscript (Tables 1 and 2).
TNF-α as a marker for uterine fibroids.

TNF: tumor necrosis factor; TP: true positive; FP: false positive; FN: false negative; TN: true negative; UF: uterine fibroid.
TNF-α as a marker of uterine fibroids. Sensitivity, specificity, and related statistical calculations.

Predictive values for optimal discrimination between patients with UFs and controls, with normal and abnormal optimal threshold values, were calculated. Changes in sensitivity and specificity are presented in receiver operating characteristic (ROC) curves. Evaluation of the area under the curve (AUC) of ROC curves determines the diagnostic usefulness of a given parameter. Higher AUC of the ROC curve corresponds to better diagnostic efficiency of a given laboratory parameter.
The
Results
Age
A total of 149 Caucasian women were included in the study. The control group
comprised 64 subjects (42.95%) aged 22.0–53.0 years; half of these women were
<39.5 years (median (Me) = 39.5 years); mean age was 38.69 ± 7.76 years. The
study group comprised 85 subjects with UFs (57.05%) aged 25.0–52.0 years; half
of these women were <41.0 years (Me = 41.0 years), and mean age was
40.95 ± 6.48 years. There was no statistically significant difference in age
between the two groups (Student’s t-test; t = 1.94; df = 147;
Body mass index
Body mass index (BMI) among controls ranged from 18.29 to 38.01 kg/m2;
in half of them, BMI was <22.17 kg/m2
(Me = 22.17 kg/m2), and mean BMI was 23.45 ± 4.18 kg/m2.
In the study group, BMI values ranged from 17.72 to 39.06 kg/m2; in
half of them, BMI was <25.10 kg/m2 (Me = 25.10 kg/m2),
while mean BMI was 26.13 ± 4.49 kg/m2. Significantly higher mean BMI
was observed in the study group as compared to controls (Student’s t-test;
t = 3.73; df = 147;
Serum TNF-α levels
In the control group, serum TNF-α concentrations ranged from 0.012 to 0.53 pg/mL;
in half of them, serum concentration was <0.14 pg/mL (Me = 0.15 pg/mL), mean:
0.17 ± 0.09 pg/mL. In the study group, TNF-α serum concentrations ranged from
0.05 to 0.59 pg/mL; in half of the group, serum concentration was <0.39 pg/mL
(Me = 0.39 pg/mL), mean: 0.34 ± 0.14 pg/mL. Significantly higher levels of serum
TNF-α were observed in the study group as compared to controls (Mann–Whitney
test; Z = 6.99;
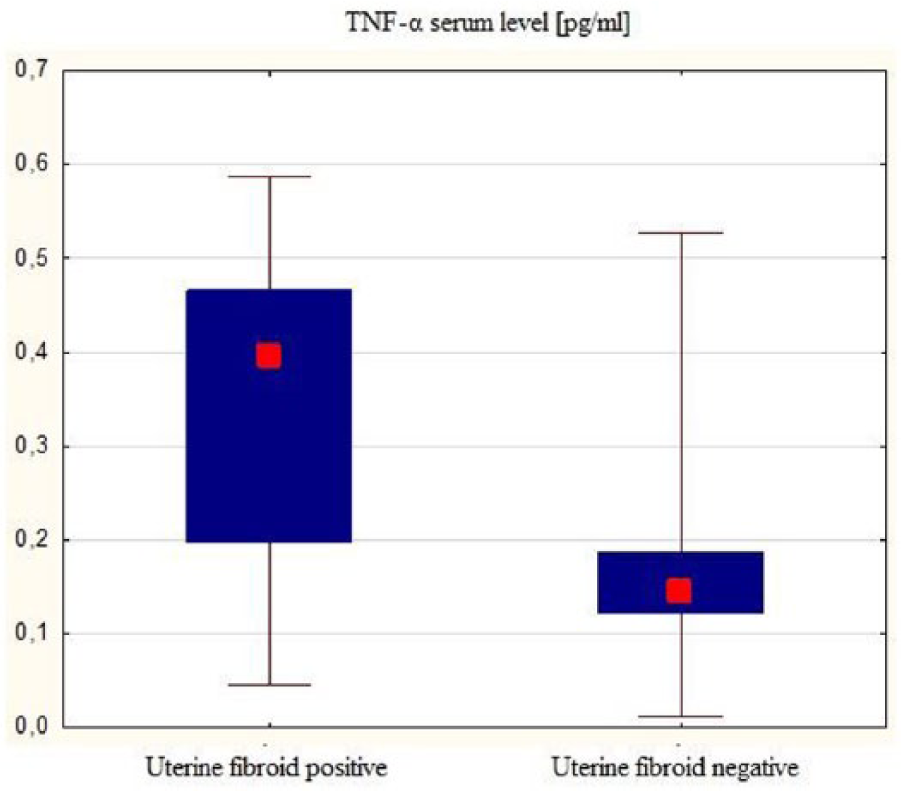
TNF-α serum levels in both groups—comparison.
TNF-α serum level as a marker for UFs
Next, we evaluated the usefulness of serum TNF-α concentration for diagnosing UFs
using ROC (AUC) surface area. The area under the ROC curve is characterized by
sensitivity and specificity. The AUC was 0.835, and the cut-off point for TNF-α
(0.34 pg/mL) was statistically significant (z = 9.9;
TNF-α serum concentration of ≥0.34 pg/mL—a woman will be categorized as UF positive;
TNF-α serum concentration of <0.34 pg/mL—a woman will be categorized as UF negative.
The results are presented in Figure 2. Sensitivity, specificity, and other related calculations are presented in Table 2.

ROC with TNF-α serum levels cut-off point for uterine fibroids.
TNF-α serum level and a total tumor burden
A statistically significant positive correlation (R = 0.296;
Correlation of serum TNF-α levels to uterine fibroid volume obtained by Spearman test.

UF: uterine fibroid; TNF: tumor necrosis factor.
Discussion
TNF-α is expressed in immune cells, but it is also found in smooth muscle cells as a response to tissue injury or upon immune response. TNF-α can induce a wide range of intracellular signal pathways (e.g. apoptosis, cell survival, and inflammation). 9 In many cases, elevated serum TNF-α concentration is one of the causes of clinical symptoms and patient complaints.
In our study, we found that patients with confirmed UFs had almost twice as high mean
serum TNF-α levels as compared to controls (statistically significant at
The abovementioned data suggest the presence of an “inflammation-like” state in women with UFs. Due to the complexity of the pathophysiological pathways in which TNF-α participates, we believe that it will not be a specific marker for UFs. Possibly, consideration of other biochemical parameters could increase sensitivity.2,12 Our results showed that TNF-α serum concentration is elevated in women with clinically symptomatic UFs. It is possible that TNF-α may prove useful as an additional clinical marker for the diagnosis and therapy of UFs after studies on bigger groups.
Footnotes
Acknowledgements
All of the STROBE guidelines were indicated in the preparation of this manuscript.
Declaration of conflicting interests
M.C. and G.J. have received personal fees from Gedeon Richter not related to this work.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Funding
This study was funded by the Medical Centre of Postgraduate Education, Warsaw, Poland (grant no.: 501-1-21-27-17).
Informed consent
Informed consent was obtained from all individual participants included in the study.
