Abstract
Background
Although intraoperative magnetic resonance imaging (iMRI) is well established in neurosurgery, its role in musculoskeletal oncology remains unclear.
Purpose
To assess the feasibility and safety of iMRI in bone and soft tissue tumor surgery, focusing on giant cell tumor of bone (GCTB) after denosumab.
Material and Methods
Fourteen patients (12 GCTB, 1 undifferentiated pleomorphic sarcoma [UPS], 1 chondroblastoma) underwent tumor resection with intraoperative 0.4 T MRI between 2017 and 2024. Outcomes included residual tumor detection, recurrence, and safety.
Results
iMRI identified residual tumor in 7 of 12 GCTB patients (58.3%), all histologically confirmed. At a median follow-up of 42 months, recurrence occurred in one GCTB case (8.3%). iMRI guided complete resection in a femoral head chondroblastoma, enabling full functional recovery. Soft tissue assessment was limited in the UPS case. No iMRI-related complications occurred.
Conclusion
iMRI is a feasible and safe adjunct in bone tumor surgery, enhancing detection of residual tumor and supporting joint-preserving procedures, particularly in GCTB after denosumab.
Keywords
Introduction
Intraoperative magnetic resonance imaging (iMRI) has become an indispensable tool in neurosurgery, particularly in the management of brain tumors and other complex intracranial pathologies. By allowing real-time imaging during surgery, iMRI enhances surgical precision, facilitates residual tumor tissue identification, preserves critical neurological structures, and ultimately improves clinical outcomes. Combining iMRI with other advanced imaging modalities and neuronavigation systems can further augment its utility, making it a cornerstone of modern neurosurgical practice.1,2
Notably, evidence has shown that iMRI significantly increases the rate of gross total resection (GTR) in glioma surgery. Indeed, previous studies have reported that the use of iMRI increased GTR from 18% to 73% compared to that with conventional surgery without intraoperative imaging. 3 Similarly, iMRI has demonstrated value for recurrent pituitary adenomas, with GTR rates increasing from 33.9% to 49.2%. 4
iMRI also plays a crucial role in preserving neurological function. In fact, studies have shown that when used in conjunction with diffusion tensor imaging (DTI) and tractography, iMRI allow for intraoperative visualization of eloquent areas of the brain, thereby reducing the risk of perioperative neurological deficits. 5
Beyond neurosurgery, research has explored the application of iMRI in musculoskeletal oncology. Recent interdisciplinary approaches involving neurosurgery and radiation oncology have demonstrated the utility of iMRI in identifying residual tumor tissues following preoperative denosumab treatment in giant cell tumors of bone (GCTBs). 6 Moreover, studies have reported that iMRI can aid in localizing recurrence in soft tissue sarcomas and assessing surgical margins during bone tumor resections, particularly when intraoperative pathological evaluation is not feasible.7–9 Nevertheless, reports detailing the use of iMRI in the surgical treatment of bone and soft tissue tumors remain scarce.
The present study therefore aimed to investigate the clinical utility of iMRI in musculoskeletal tumor surgery. Specifically, we evaluated mid-term outcomes in patients with GCTB following denosumab therapy, explored its application in the resection of malignant soft tissue tumors, and assessed its use in the intraoperative evaluation of femoral head chondroblastoma. Our analysis seeks to clarify the role of iMRI in improving surgical outcomes within the field of orthopedic oncology.
Therefore, the present study aimed to evaluate the feasibility and safety of iMRI in musculoskeletal tumor surgery, with a particular focus on giant cell tumor of bone (GCTB) following denosumab treatment.
Given the small number of patients, this report should be considered a preliminary pilot study, intended to provide insights into the potential role of iMRI in this field.
Material and methods
Study population and design
This study builds upon our previous pilot report and was conducted as a prospective, single-institutional study of patients who underwent surgery for bone and soft tissue tumors between September 2017 and December 2024. Diagnostic evaluation was based on clinical assessment, radiography, and MRI. Institutional review board approval was obtained from the Epidemiological Ethics Review Committee of Hiroshima University (approval No. 4169) prior to study initiation, and all participants provided written informed consent in accordance with the Declaration of Helsinki.
A total of 14 patients were included 12 with giant cell tumor of bone (GCTB), 1 with undifferentiated pleomorphic sarcoma (UPS), and 1 with chondroblastoma. GCTB diagnoses were confirmed histopathologically through open biopsy. UPS was diagnosed through core needle biopsy. Chondroblastoma was diagnosed based on characteristic radiological and clinical findings, as its location within the femoral head precluded biopsy.
Because intraoperative MRI requires substantial logistical resources, operating room coordination, and intraoperative patient transfer, it was selectively applied only in cases where a clear clinical benefit was anticipated; therefore, this study was designed as a pilot study with a limited number of carefully selected patients.
Drug regimen
Patients with GCTB received neoadjuvant denosumab (120 mg subcutaneously) for a median duration of 6.8 months (range, 6–12 months). The administration schedule comprised weekly injections for the initial 4 weeks, followed by monthly maintenance dosing. The primary therapeutic goal was to promote subchondral bone consolidation, thereby facilitating joint preservation. Treatment duration and dose modifications were guided by clinical response and national reimbursement policies. 10 Surgical intervention was scheduled after adequate subchondral sclerosis was confirmed based on radiographic examination.
Surgical procedure
All surgeries were performed by a team of three experienced orthopedic oncologic surgeons. Surgical techniques were tailored to tumor pathology and anatomical site:
GCTB: Patients underwent extended curettage, followed by reconstruction using either bone graft or cement augmentation, in accordance with evolving institutional standards and JCOG1610 recommendations.
UPS: Wide resection was performed with vascular control and en bloc tumor removal.
Chondroblastoma: Curettage of the femoral head was performed using a joint-preserving approach with adjunctive reconstruction techniques.
(Further details of individual cases are provided in the Results section.)
Preoperative and intraoperative MRI protocol
Preoperative conventional MRI was performed within 2 weeks prior to surgery in all patients. In addition, after induction of general anesthesia and before tumor resection, an initial intraoperative MRI (iMRI) scan was acquired. This initial iMRI served both as a simulation of the intraoperative imaging environment and as a reference for tumor visualization. When both preoperative MRI and initial iMRI were available, the imaging set that provided clearer delineation of the tumor was selected as the reference for intraoperative assessment. During surgery, additional iMRI scans were obtained after tumor resection when residual tumor was suspected. Although the number of repeat scans was limited by operative time and staffing constraints, repeat iMRI was performed as flexibly as possible and, whenever feasible, continued until no residual tumor was detected.
iMRI
To evaluate residual tumor burden, all patients underwent iMRI on a 0.4-Tesla Lucent system (Hitachi, Japan). For this purpose, a custom-modified, MRI-compatible operating table was developed in collaboration with our neurosurgical and biomedical engineering teams (Figure 1). The imaging protocol included
1
T2-weighted fast spin-echo (slice thickness, 5 mm; FOV, 256 mm; TR, 1000 ms; TE, 140 ms) and T1-weighted gradient echo (slice thickness, 5 mm; FOV, 256 mm; TR, 27 ms; TE, 10 ms).
11
Intraoperative MRI equipment and operating table setup. Intraoperative magnetic resonance imaging (MRI) equipment and images obtained during imaging. A: Intraoperative MRI device (0.4T Lucent, Hitachi, Japan). B: Modified MRIcompatible operating table developed to allow safe patient transfer between surgical and imaging positions.
Preoperative MRI was acquired immediately before surgery under general anesthesia. iMRI was conducted following curettage (GCTB and chondroblastoma) or wide resection (UPS) to assess residual tumors.
A standardized iMRI workflow was established. In particular, baseline MRI was obtained days before or on the day of surgery. Surgery proceeded under general anesthesia with C-arm fluoroscopy to protect the joint surface. The limb was prepared using double-layered sterile drapes and transported to the MRI suite while strictly adhering to MRI safety protocols. iMRI findings were then compared to preoperative imaging findings to determine the presence of residual tumors. If present, additional curettage was performed. Repeat iMRI was conducted to verify complete tumor clearance. Final reconstruction involved cementation or cryoablation using liquid nitrogen, followed by internal fixation if required.
All imaging studies were reviewed by a multidisciplinary team comprising three orthopedic oncologists and two musculoskeletal radiologists to confirm tumor resection status and margin clearance.
Outcomes
The primary endpoints included the detection rate of residual tumor using iMRI, anatomical localization of residual tissue, histopathological confirmation, integrity of the articular surface, iMRI-related safety, imaging and operative time, and adverse events. Local recurrence was evaluated in patients with GCTB at a mean follow-up of 42 months.
Results
The study cohort consisted of 14 patients (7 males, 7 females) with a median age of 32.3 years (range, 14–78 years). The GCTB subgroup included 12 patients (7 males, 5 females; median age 32.3 years, range 14–58 years). Tumor sites were the femur (n = 7), tibia (n = 3), and distal radius (n = 2). Two patients were presented with pathological fractures, and one had recurrent disease. The UPS patient was a 78-year-old female with a high-grade soft tissue tumor in the right gluteal region. The chondroblastoma patient was a 16-year-old male with disease involving the right femoral head.
GCTB treated with preoperative denosumab (n = 12)
All 12 patients underwent curettage following neoadjuvant denosumab therapy.
Reconstruction methods: One patient underwent reconstruction with autologous iliac crest bone graft augmented with synthetic bone, secured by locking plate fixation. The remaining 11 patients received cement augmentation after curettage, in line with JCOG1610 recommendations.
Residual tumor detection: Despite apparent gross total resection, iMRI identified suspicious residual lesions in 7 patients (58.3%). Additional curettage was performed, and all resected tissue contained viable tumor histologically. Residual foci were consistently located within areas of dense subchondral sclerosis induced by denosumab, which limited visual and tactile detection during surgery (Figure 2(a) and (b)). Intraoperative MRI equipment and operating table setup. (i) Comparison of preoperative and intraoperative magnetic resonance imaging (MRI) after the first curettage of a giant cell tumor of the right femur in a 32-year-old female. (a) Preoperative T2-weighted image. (b) Intraoperative T2-weighted image after the first curettage. Blue arrows indicate residual tumors identified on intraoperative MRI. II. A 17-year-old male who developed recurrence following surgery for a giant cell tumor of the right tibia, as identified using intraoperative MRI. (c) Intraoperative T2-weighted image after the second curettage. The red arrow indicates a residual tumor that was very small and difficult to detect. (d) T2-weighted image at the time of recurrence. A tumor formed from the area indicated by the red arrow, which was suspected to be a residual tumor, extending posteriorly along the tibia.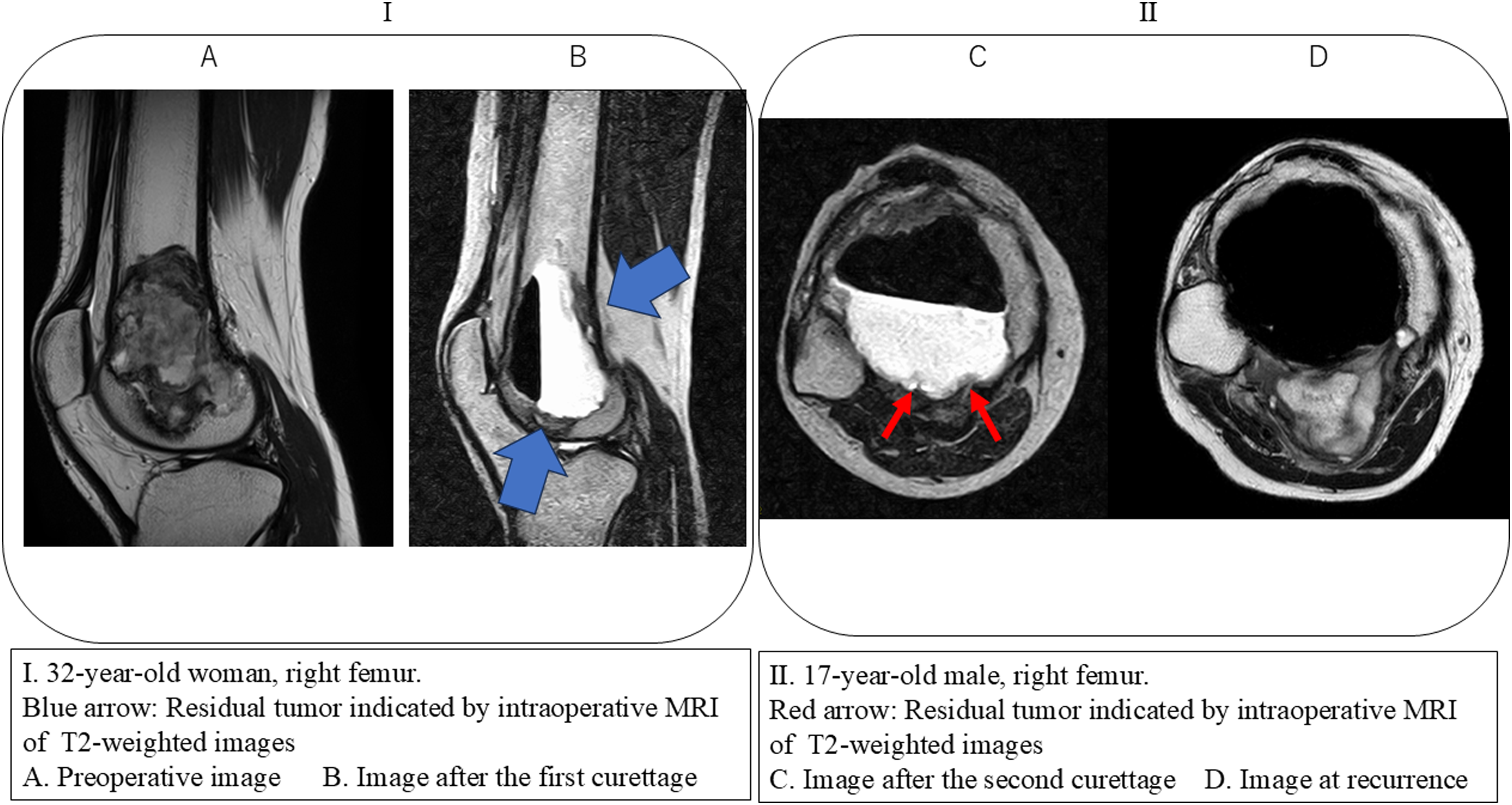
Safety and outcomes: iMRI procedures were completed without technical or anesthetic complications. The mean number of imaging sessions per surgery was 2.3 (range, 2–3), with an average cumulative imaging duration of 64 min. The mean total operative time was 163 min. At a median follow-up of 42 months (range, 29–60 months), one patient (8.3%) developed local recurrence. Retrospective analysis confirmed that the recurrence originated at a subtle abnormality noted on intraoperative imaging but not initially interpreted as definitive (Figure 2(c) and (d)). This lesion was later excised en bloc after re-initiation of denosumab, followed by reconstruction with a modular tumor endoprosthesis.
UPS of the right femur (n = 1)
The single UPS case involved a 78-year-old female with a high-grade sarcoma in the posterior thigh. A staged resection was performed: vascular control was first achieved through an anteromedial approach, followed by repositioning for en bloc tumor removal. Intraoperatively, iMRI was used to assess resection margins and exclude residual disease.
Findings: Cortical and medullary bone structures were clearly delineated, but soft tissue evaluation was limited by the low field strength (0.4 T). No residual tumor was identified (Figure 3). Intraoperative MRI evaluation in undifferentiated pleomorphic sarcoma. A 78-year-old woman with undifferentiated pleomorphic sarcoma of the right thigh. Ⅰ. Preoperative magnetic resonance imaging (MRI): (a) T1-weighted image, (b) T2-weighted image, with red arrows indicating the tumor location. II. Intraoperative MRI after tumor resection: (c) T1-weighted image, (d) T2-weighted image. No obvious residual tumor was present.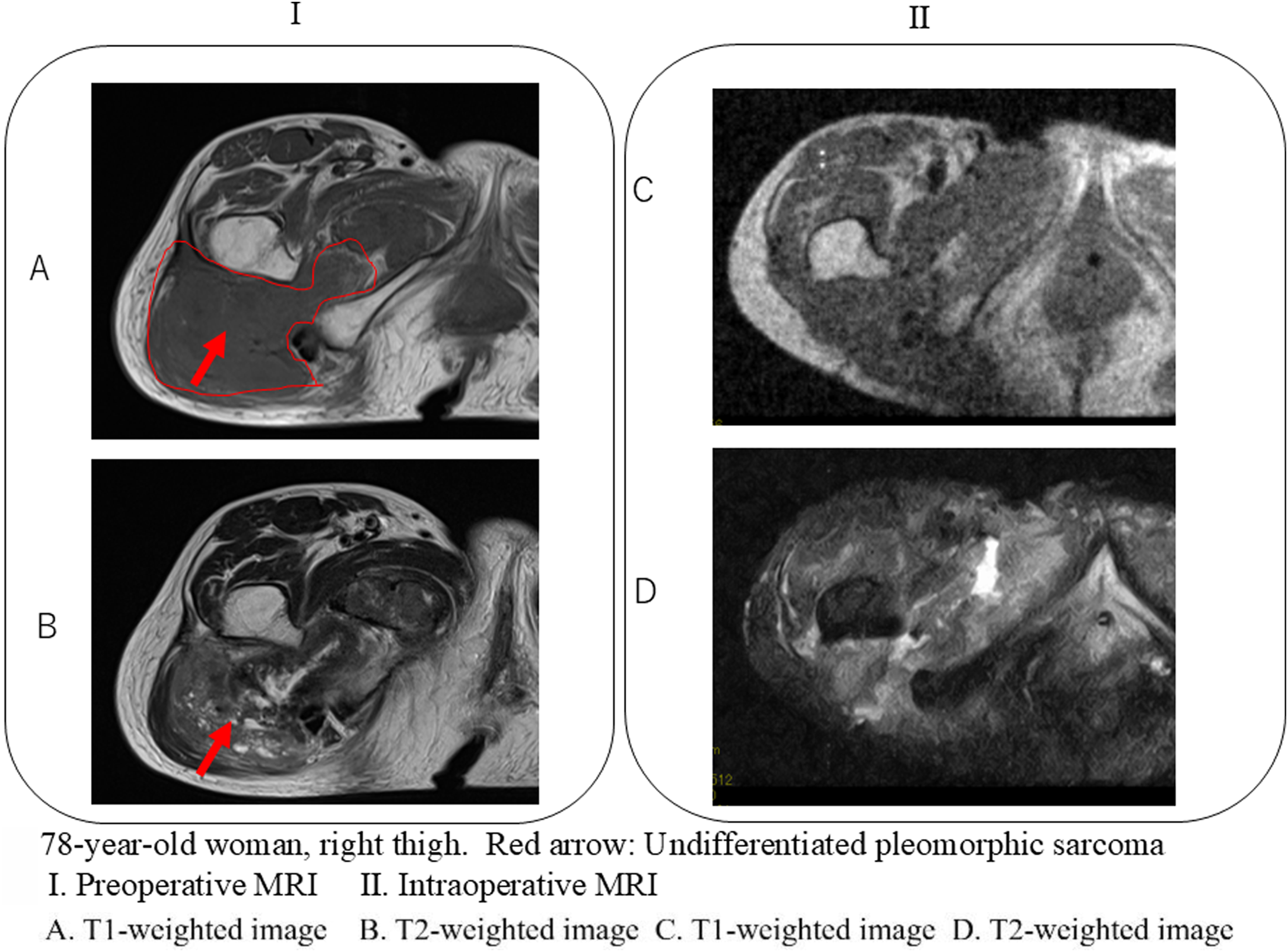
Perioperative course: The imaging session lasted 31 min, and total operative time was 182 min. No intraoperative or postoperative complications occurred.
Chondroblastoma of the femoral head ( n = 1)
A 16-year-old male presented with a radiographically well-demarcated lesion in the femoral head. Curettage was performed after hip dislocation and trochanteric osteotomy. iMRI following the first curettage revealed a deep residual lesion in the epiphyseal region.
Additional treatment: Further curettage was performed, and histology confirmed viable chondroblastoma tissue. Ethanol was applied as a local adjuvant, and costal cartilage harvested from the ribs was implanted for joint preservation.
Outcomes: The iMRI session lasted 31 min, and total operation time was 201 min. No perioperative complications occurred. At the 24-month follow-up, the patient achieved full functional recovery without evidence of recurrence or femoral head osteonecrosis (Figure 4(a)–(g)). The patient successfully returned to competitive volleyball. Use of intraoperative MRI in femoral head chondroblastoma with cartilage grafting. A 16-year-old male with chondroblastoma of the right femoral head. (a) Preoperative radiography of the right femoral head. (b) Preoperative T2-weighted magnetic resonance (MR) image. (c) Intraoperative T2-weighted MR image after the first curettage. (d) Intraoperative T2-weighted MR image after the final curettage. Red arrows indicate chondroblastoma, whereas blue arrows indicate residual tumors identified on intraoperative MRI, emphasizing low-signal lesions. (e) Costal cartilage harvested from the sixth and seventh ribs as a donor. (f) Intraoperative image after costal cartilage transplantation. (g) T2-weighted MR image 3 months after surgery.
Discussion
iMRI has been extensively adopted in neurosurgery given its demonstrated efficacy in maximizing tumor resection and minimizing recurrence in high-risk brain tumors.1–4 Building upon this precedent, a previous pilot study reported the novel application of iMRI in orthopedic oncology, specifically for GCTB, suggesting its potential in aiding the intraoperative detection of residual lesions. 6
The current study found that the detection rate of residual tumor tissues in GCTB using iMRI was 58.3% (7/12 cases), despite having achieved gross total resection under direct visualization. These residual lesions were in areas of subchondral sclerosis induced by preoperative denosumab, which rendered intraoperative identification particularly challenging. Over a median follow-up period of 42 months, 1 patient (8.3%) experienced local recurrence. Although this observed recurrence rate appears lower than previously reported rates for GCTB, which range from 20%–30% in general12–15 and 30%–50% in patients treated with denosumab prior to surgery,16,17 these comparisons should be interpreted with caution given the small sample size and lack of a comparator group.
Importantly, a retrospective review of the patients who developed recurrence revealed subtle intraoperative iMRI abnormalities at the site of later recurrence (Figure 2(c) and (d)), highlighting the diagnostic sensitivity of iMRI. This finding underscores the critical need for enhanced intraoperative vigilance, particularly in the surgical management of patients who had received denosumab, in whom tumor boundaries may be masked by reactive bone changes. 16 Although denosumab has become a mainstay in managing unresectable or metastatic GCTB, 17 its use should remain selective. When performing surgical curettage following denosumab, iMRI may serve as a valuable adjunct for achieving more complete resection by facilitating the detection of residual tumor tissue, which is a known contributor to local recurrence.
The role of iMRI in soft tissue sarcoma surgery remains less established. Despite having clearly visualized the osseous anatomy in our UPS patient, the lower field strength (0.4 T) of the MRI unit limited the delineation of the soft tissue margins. This inability to precisely define the resection margins likely contributed to reduced diagnostic utility, as also noted in a limited number of prior studies.7–9 Strategies that enhance visualization, such as placing radiopaque sheets between the surgical planes, may improve interpretability. Nonetheless, the use of higher field strength systems (1.5 or 3 T) would likely be a prerequisite for reliably detecting microscopic soft tissue remnants.
The third case involved chondroblastoma in a skeletally immature patient, in whom complete tumor clearance with maximal preservation of the articular cartilage was critical. Unlike GCTB, chondroblastoma does not present with sclerosis, and iMRI clearly delineated a deep residual lesion not evident during direct inspection. Additional curettage was performed, with a second iMRI confirming complete tumor removal. The surgical strategy, which included rib cartilage grafting, promoted joint preservation and return to full sports participation without evidence of osteonecrosis or recurrence.
The inclusion of non-GCT cases was intended to explore the broader applicability of iMRI for intraoperative margin assessment, not to compare oncological outcomes.
Taken together, these findings support the use of iMRI as an intraoperative adjunct to bone tumor surgery, particularly when performing function-sparing procedures in anatomically complex or sclerotic cases. Its utility is most apparent in curettage-based surgery following denosumab, wherein conventional tactile and visual assessments are insufficient.
The present study has several limitations that warrant discussion. First, iMRI requires considerable financial and technical resources and is currently available only at select centers. Moreover, the integration of iMRI into bone tumor surgery necessitates close collaboration with neurosurgical teams, radiologists, and MRI technologists to ensure safety, particularly during patient transfer and metallic object exclusion.
Second, although computed tomography offers superior visualization of cortical bone and shorter acquisition times, it exposes patients to ionizing radiation and lacks sensitivity for soft tissue contrast and marrow infiltration. In contrast, MRI offers superior soft tissue resolution and is preferable in the absence of significant sclerosis or when radiation exposure is a concern.
Finally, given the limited sample size, no formal confidence intervals were calculated for recurrence outcomes, and all findings related to recurrence should be interpreted as exploratory. The small sample size, particularly for non-GCTB tumors, substantially limits the generalizability of our findings. Therefore, the present work should be regarded as a pilot feasibility study rather than a definitive evaluation. Nonetheless, our results provide preliminary evidence that iMRI may enhance surgical accuracy and may have the potential to influence local recurrence in selected bone tumor cases. Future multi-institutional studies with larger cohorts and higher-resolution MRI systems will be essential to validate these findings and clarify the role of iMRI in musculoskeletal oncology.
In conclusion, this pilot study demonstrates that iMRI is a safe and feasible adjunct in bone tumor surgery. It allows for improved detection of residual tumor tissue, particularly in GCTB after denosumab therapy, and should be regarded as a hypothesis-generating approach with respect to its potential impact on local recurrence.
Footnotes
Acknowledgments
Ethical consideration
This study was approved by the Epidemiological Ethics Review Committee of Hiroshima University (approval No. 4169).
Consent to participate
Written informed consent was obtained from all participants (and from the parents/legal guardian of the minor patient).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
