Abstract
Primary aortoenteric fistula is a rare entity with high mortality. Depending on the location of the fistula, primary aortoenteric fistulas may lead to upper or lower gastrointestinal hemorrhages. The majority of primary aortoenteric fistulas are associated with atherosclerosis or aneurysms of the aorta, without any previous aortic procedure. Local inflammation, compression, or erosion may lead to rupture of the aneurysm. Owing to difficulties in immediate and correct diagnosis, effective treatment of primary aortoenteric fistulas is often delayed, leading to almost 100% mortality in untreated patients. Here, we report a case of a repetitive mass of lower gastrointestinal hemorrhage due to a primary internal iliac arterial-sigmoid colon fistula. Finally, the patient was managed successfully through endovascular coil embolization.
Introduction
Aortoenteric fistula (AEF) is a rare entity that originates from spontaneous rupture of the aorta into the gastrointestinal (GI) tract. In terms of anatomic location, AEF includes aorto-esophageal fistula, aorto-duodenal fistula, and aortoenteric fistula. 1 The most common site of AEF is the duodenum due to its fixed retroperitoneal position and proximity to the aorta. 1 Primary AEF (PAEF), which often involves atherosclerosis or aneurysm of the aorta, has been reported in less than 0.07% of cases. 2 Moreover, secondary AEF (SAEF) due to aortic interventions or local infection is relatively common, with an estimated incidence rate of 0.3%–2%. 2 Spontaneous rupture of the aorta or aneurysm through the fistula may lead to massive GI hemorrhage with high mortality. Here, we report a case of recurrent lower GI hemorrhage due to a primary internal iliac arterial-sigmoid colon fistula. In addition, the patient was diagnosed with tubercular peritonitis, complicating the diagnosis and treatment. Finally, the patient was managed successfully through endovascular coil embolization and standard antituberculous treatment.
Case report
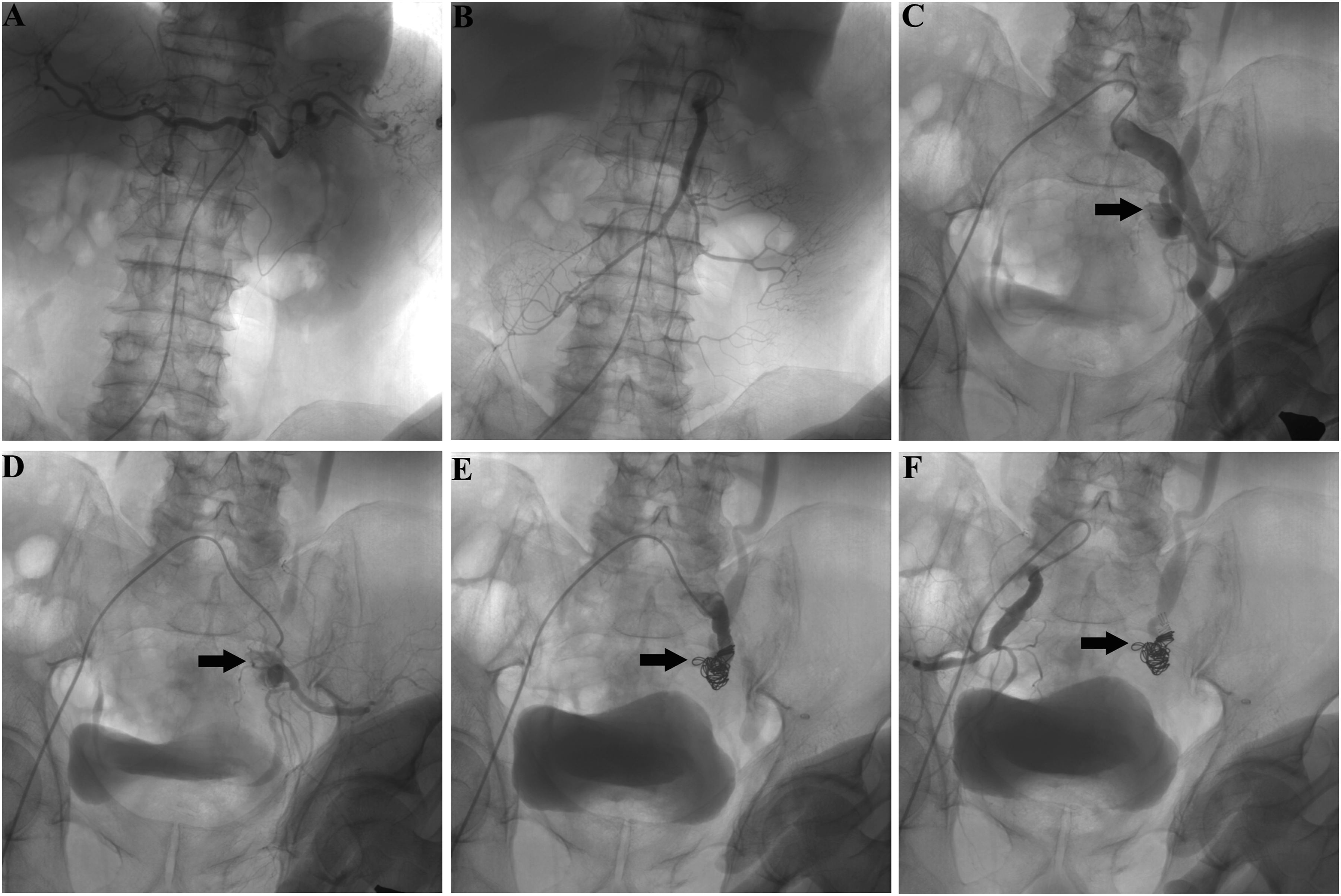
A 65-year-old Tibetan male presented with a 1-month history of recurrent massive lower gastrointestinal hemorrhage at our hospital. Two months prior, the patient was diagnosed with tubercular peritonitis and a sigmoid colonic mass with ulcers (Figure 1(a)). The patient subsequently received standard anti-TB therapy (isoniazide, rifampicin, ethambutol, and pyrazinamide) and was compliant with the treatment. However, the patient has experienced recurrent massive hematochezia and hemorrhagic shock twice since then. After admission, a CT scan of the abdomen revealed a left internal iliac aneurysm with a mural thrombus (Figure 1(b)). During hospitalization, the patient experienced massive hematochezia and a sharp decrease in Hb (from 70 g/L to 31 g/L). Owing to hemodynamic instability, the patient immediately received 4 units of red blood cells and was subsequently referred for angiographic evaluation. Initially, celiac trunk and mesenteric angiography did not reveal the source of the bleeding (Figure 2(a) and (b)). Selective arteriography of the left common and internal iliac arteries demonstrated gross extravasation of contrast agent through a fistula between the internal iliac artery aneurysm and the sigmoid colon (Figure 2(c) and (d)). A catheter was then selectively placed in the aneurysm and embolized with seven 12 mm Nester coils (Cook, Bloomington, IN, USA). Postembolization angiography revealed no active bleeding from either the right or the left internal iliac artery (Figure 2(e) and (f)). The patient received fluid resuscitation after the operation. One week later, the patient was discharged from the hospital without gastrointestinal bleeding and was maintained on anti-TB treatment for 1 year. He had remained well at the 3-year follow-up. Colonoscopy and contrast-enhanced computed tomography (CT) examination. (a) Colonoscopy revealed a sigmoid colonic mass with an ulcer (arrow). (b) CT scan showing a left internal iliac aneurysm with a mural thrombus (arrow). Emergency aortography and coil embolization. (a and b) Transarterial radiography of the celiac trunk and superior mesenteric artery did not reveal the source of the bleeding. (c and d) Transarterial radiography of the left common and internal iliac arteries demonstrated gross extravasation of the contrast agent (arrows). (e) Transcatheter arterial embolotherapy of the fistula between the internal iliac arterial aneurysm and sigmoid colon via coils (arrows). (f) Transarterial radiography of the right internal iliac artery demonstrated no bleeding.
Discussion
PAEF, first described in 1829, is a rare condition and is associated mainly with atherosclerotic aneurysms of the aorta or with inflammatory aortic diseases. 2 As less common than other abdominal aortic aneurysms, PAEF due to iliac artery aneurysm is limited to only a few case reports.3–5
The classical clinical symptoms of PAEF include GI hemorrhage, abdominal pain, and a pulsatile abdominal mass. 2 Previous reports have indicated that GI hemorrhage may be obscure and is the initial presentation in more than 90% of PAEF cases, regardless of etiology. 6 Massive hemorrhage is often lethal if diagnosis and treatment are delayed. Therefore, PAEF should be suspected in all patients with massive GI hemorrhage, especially patients with aortic aneurysms. In the present case, the presence of repeated hematochezia and aortic aneurysm led us to suspect this diagnosis. However, there is still controversy regarding the best diagnostic modality for PAEF. Some authors have recommended initial upper or lower endoscopy and CT scans. 7 Endoscopy may identify the bleeding location and exclude other causes of bleeding. Contrast-enhanced CT may reveal the abdominal mass or the aneurysm, as in this patient. However, if there is bleeding without an identifiable source, angiography should be performed to demonstrate extravasation of contrast agent from the fistula into the GI tract.
Despite the high burden of tuberculosis (TB) infections in China, TB-associated PAEF has rarely been reported. TB may affect both the gastrointestinal (GI) tract and the arterial wall directly or indirectly.8,9 On the one hand, TB infection can result in pseudoaneurysms or aneurysms of both the aorta and the visceral arteries, which may subsequently break through the adjacent GI tract. On the other hand, the most common pathway is direct extension from adjacent TB-infected organs, such as the para-aortic lymph nodes, or local abscess. In the present case, a left internal iliac aneurysm, tubercular peritonitis, and a colonic mass were diagnosed before GI bleeding. However, no direct evidence of intestinal tuberculosis or arterial tuberculosis was found. Based on the computed tomography (CT) and colonoscopy results, we propose that the fistula between the sigmoid colon and internal iliac artery aneurysm might be due to the direct extension of tubercular peritonitis.
The treatment of PAEF should include controlling bleeding and repairing the fistula. Surgical repair of the fistula is the standard treatment. 6 However, it might not be suitable for elderly or hemodynamically unstable patients. In recent years, the endovascular approach has become an important alternative treatment option for PAEF to open surgery, with fewer complications and shorter hospitalization periods. 10 Previous reports have indicated that transcatheter coil embolization has been used to control emergency hemorrhage and can be life-saving. 11 Since our patient was hemodynamically unstable, transarterial radiography was rapidly implemented. The fistula was confirmed, and occlusion of the internal iliac aneurysm with embolization coils was successfully performed.
In conclusion, PAEF is a rare but fatal entity. The diagnosis of PAEF should be considered in patients presenting with massive or repetitive GI bleeding, especially patients with an aortic aneurysm. Failure or delay in diagnosis is often associated with high mortality. Compared with surgical repair, transcatheter arterial embolotherapy may be the preferred treatment option for hemodynamically unstable patients.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Sichuan Province [grant number 2023NSFSC0742] and Foundation of The Central Hospital Western Theater Command [grant number 2019ZY01].
