Abstract
Background
Focal liver lesions (FLL) are abnormal growths that require timely identification. Contrast-enhanced ultrasound (CEUS) is a cost-effective imaging modality for characterising FLL with similar sensitivity to computed tomography (CT) and magnetic resonance imaging (MRI). Despite being recommended by NICE, its adoption within the national health service (NHS) is limited due to low clinical demand, limited referral, and lack of knowledge.
Purpose
To evaluate the impact of CEUS on patients with incidental FLL and assess the resource implications of introducing CEUS as a diagnostic service within the NHS.
Material and methods
A patient flow review and cost-minimisation analysis were conducted. This involved a targeted literature review, NHS Trust stakeholder consultations, and development of a Microsoft Excel cost-minimisation model to explore potential value of CEUS use versus CT and MRI by episode. A scenario analysis of the base-case explored increasing CEUS use to 50% and 90%. A sensitivity analysis was performed to assess how changes in assumptions impacted the model and the resulting cost estimates.
Results
The model, comparing a world with and without CEUS, showed that current use (base-case: 5%) resulted in cost savings of £224,790/year. The sensitivity analysis indicated that regardless of changes to the assumptions, CEUS still resulted in cost savings to the NHS. By increasing CEUS use to 50% and 90%, cost savings of up to £2,247,894/year and £4,046,208/year could be achieved, respectively.
Conclusion
By standardising CEUS use for characterising FLL, substantial cost savings could be realised, whilst reducing wait times and expanding diagnostic capacity, thus preserving limited CT and MRI capacity for high-priority cases.
Keywords
Introduction
Focal liver lesions (FLL) are discrete abnormal growths that include cystic and solid abnormalities. 1 These can often be found incidentally during imaging for other purposes, for example, work up for abdominal pain or during surveillance for liver metastases in patients with extrahepatic cancer (e.g. breast cancer). 2 FLLs can be benign or malignant, and require timely identification and characterisation to facilitate treatment. 3 However, with a lack of clear pathway for workup, multiple imaging modalities may be needed for characterisation. 2 Typical imaging methods include, but are not limited to, ultrasound (US) for detection with computed tomography (CT), and magnetic resonance imaging (MRI) for follow-up characterisation. 4 Alongside the absence of a clear pathway, the United Kingdom (UK) National Health Service (NHS) is facing resource pressures resulting in staffing issues, backlogs, and extended waitlists, including referrals from primary care.5,6
Contrast-enhanced ultrasound (CEUS) is another imaging modality for characterising FLL with similar sensitivity to CT and MRI, 7 and has shown a high diagnostic accuracy in differentiating between malignant and benign FLLs that were undetermined at the initial CT. 8 CEUS is therefore recommended by the National Institute of Health and Care Excellence (NICE) where an unenhanced US is inconclusive.13 Despite being cost-effective, with potential to increase imaging capacity and reduce radiation exposure, 9 there has been a lack of investment in service development and adoption within the NHS. This has led to low clinical demand, limited referral, and a lack of knowledge and training in performing CEUS. 10
The recent transition from Clinical Commissioning Groups (CCGs) to Integrated Care Systems (ICSs) in the NHS may provide an opportunity to further increase the value of a CEUS service. The structural changes have shifted budget allocation for providers: the national tariff system with reimbursement in a fee-for-service manner has evolved to block contract payments where the services provided can be managed at the hospital level and any surplus budget can be reinvested. 11
The objective of the research was to assess the impact of CEUS on the workflow of patients with incidental FLL and understand the resource impact of introducing CEUS at various levels as a diagnostic service within the NHS.
Materials and methods
A patient flow review and cost-minimisation analysis were conducted which followed three key stages, described below.
Targeted literature review
A targeted literature review was conducted to supplement the NICE guidelines and understand the patient flow for incidental FLL. The review aimed to establish the proportion of patients receiving each imaging modality, the management and, resources and costs associated with CEUS versus CT and MRI.
Search strategy.

Inclusion and exclusion criteria.

CEUS: Contrast-enhanced Ultrasound; CT: Computed tomography; Magnetic Resonance Imaging; PET: Positron emission tomography; RCT: Randomised controlled trial; SLR: Systematic literature review; US: Ultrasound.
The titles and abstracts retrieved from the PubMed search were screened based on the predefined criteria. Full-text articles were obtained for studies that met the inclusion criteria or for those where exclusion could not be determined solely based on information provided in the abstract. Single screening was carried out to determine their eligibility based on the predefined inclusion/exclusion criteria. At each stage, a quality-control check was conducted by a second researcher who randomly selected and evaluated 20% articles.
Once the final list of included studies was determined, the data were extracted in predefined tables to address the research questions.
Engagement with NHS trust stakeholders
The aim of this engagement was to validate the flow of patients with incidentally detected FLL, identified from the literature review, with NHS hospital trusts. Any differences in inputs specific to the trust and sensitivities related to the flow were also explored.
Three consultant sonographers from three different University Trusts across the UK (North-East, North-West, and South-West) with varying usage of CEUS (two trusts had low and one trust had moderate CEUS use) were interviewed individually for 30 min each.
A semi-structured engagement guide was developed to aid discussion on current CEUS use, data availability, and trust requirements for increasing CEUS use.
A first draft of the patient flow and model structure was developed using data from the literature review and initial insights from the stakeholders. A 1-h web-assisted telephone interview took place with each of the three consultant sonographers to validate the model, including: patient flow and structure, prevalence of FLL, proportions of patients sent to each imaging modality, as well as sensitivities around the choice of imaging modality used for both first-line and second-line characterisation. Amendments to the model structure were made based on their input.
Development of cost-minimisation model
A Microsoft Excel based cost-minimisation model was developed to explore the use of imaging modalities by episode and demonstrate the potential value of CEUS use.
The model included multiple lines of patient management and followed patient episodes from initial detection of an FLL, with characterisation by different imaging modalities (CEUS, CT, and MRI) until completion of the imaging phase. To assess the impact of the different modalities, national tariffs (as of November 2022) (Appendix A) were used. 12 The model was constructed to capture episodes or events rather than the full patient journey to prevent the influence of confounding non-imaging factors such as patient surveillance and biopsy.
The base case was developed to align with approximate current use of CEUS. A sensitivity analysis of the key assumptions was performed to understand the sensitivities of the model and the associated impact on the differential costs.
A scenario analysis was conducted by increasing the use of CEUS to examine whether additional cost savings could be recognised. Two scenarios were analysed: 50% and 90% CEUS use.
Results
Literature review
A total of 101 publications were captured based on the search strategy. Following abstract and full-text screening, 26 publications were included (Figure 1). PRISMA diagram. I: Intervention; O: Outcome; P: Population; S: Study design. Of the 26 publications included: n = 18 reported on the proportion of patients receiving CEUS or other imaging modalities, n = 12 outlined the management of patients during or following characterisation, and n = 11 reported on costs associated with the imaging modalities.
Few publications describe the prevalence of FLL; one review by Boutros et al. (2010) reported that clinically apparent lesions can be detected in up to 33% of abdominal or liver adjacent procedures, 13 although the management of FLL has evolved since this time period. All other publications captured were institutional studies characterising liver lesions incidentally detected by ultrasound. Further to this, only one retrospective study by Alizadeh et al. (2021) reported the proportion of patients receiving different imaging modalities for FLL characterisation with 62% receiving CT, 35% another ultrasound, and 3% undergoing MRI; 14 however, as this was a small study in Iran (n = 66), the results may not be transferable to the UK without further verification. The remaining publications characterised the lesions based on one imaging method or did not segment results by modality.
Several publications described the proportion of lesions identified as benign or malignant and whether further examinations were required for classification. A prospective study by Smajerova et al. (2016) assessing FLL with CEUS found 59.1% of cases were benign, 29.8% were malignant, and 11.1% were inconclusive, requiring follow-up with CT or MRI. 15 Two other observational studies with smaller sample sizes (n = 117 and n = 142) reported higher rates of additional imaging following CEUS with CT or MRI, ranging from 29.8% up to 38.5%.16,17
Streb et al. (2019) reported significantly reduced time for completion of outpatient examinations with CEUS (5.2 days) versus CT and MRI (52.3 and 123.5 days, respectively; p < 0.01). CEUS also had a high percentage of same-day examination with the initial US (78.4%). 17 However, no publications described the total timing following an inconclusive result or the timing between diagnosis and characterisation, including time for report development, between imaging modalities.
Several publications have demonstrated that using CEUS is a cost-effective approach that results in cost savings compared to CT and MRI.7,15,18–20 Smajerova et al. (2016) found that using CEUS generated cost savings of $3013 versus CT and $308,352 versus MRI from a hospital perspective. 15 There is a lack of literature examining the healthcare utilisation, including hospitalisation, staff time, and other resources for CEUS and other imaging modalities.
The absence of literature on patient flow, particularly recent publications reflecting current management, resulted in examination of supplementary grey literature, including NICE guidelines and NHS data on hospital outpatient activity. According to hospital episode statistics (HES) data (2018–19; note this period was used to represent full NHS diagnostic capacity due to reduced services during the COVID-19 pandemic), the number of radiological examinations showing the liver were: 579,363 with CT, 606,066 with MRI, and 1,110,972 with US 21 and NICE outline that between 70 and 75% of FLLs assessed are benign. 3 Note the HES data only provide data on episodes rather than the number of patients. The full list of procedures included can be found in Appendix B.
Validation with stakeholders
The literature review enabled draft development of a patient flow incorporating four key stages for the characterisation of incidental FLLs, following episodes of diagnostic imaging (Figure 2), which was validated with NHS Trust stakeholders. The final patient flow. aBenign lesions allowing for diagnosis; bInconclusive or suspected malignancy warranting further investigation; cConclusive and inconclusive characterization with CT and MRI could represent benign or malignant lesions; dImaging complete represents completion of the imaging phase of diagnosis and may be followed by surveillance, biopsy, or treatment, although these are not captured by the model. 2 L: Second-line; CEUS: Contrast-enhanced Ultrasound; CT: Computed tomography; FLL: Focal liver lesion; Magnetic Resonance Imaging.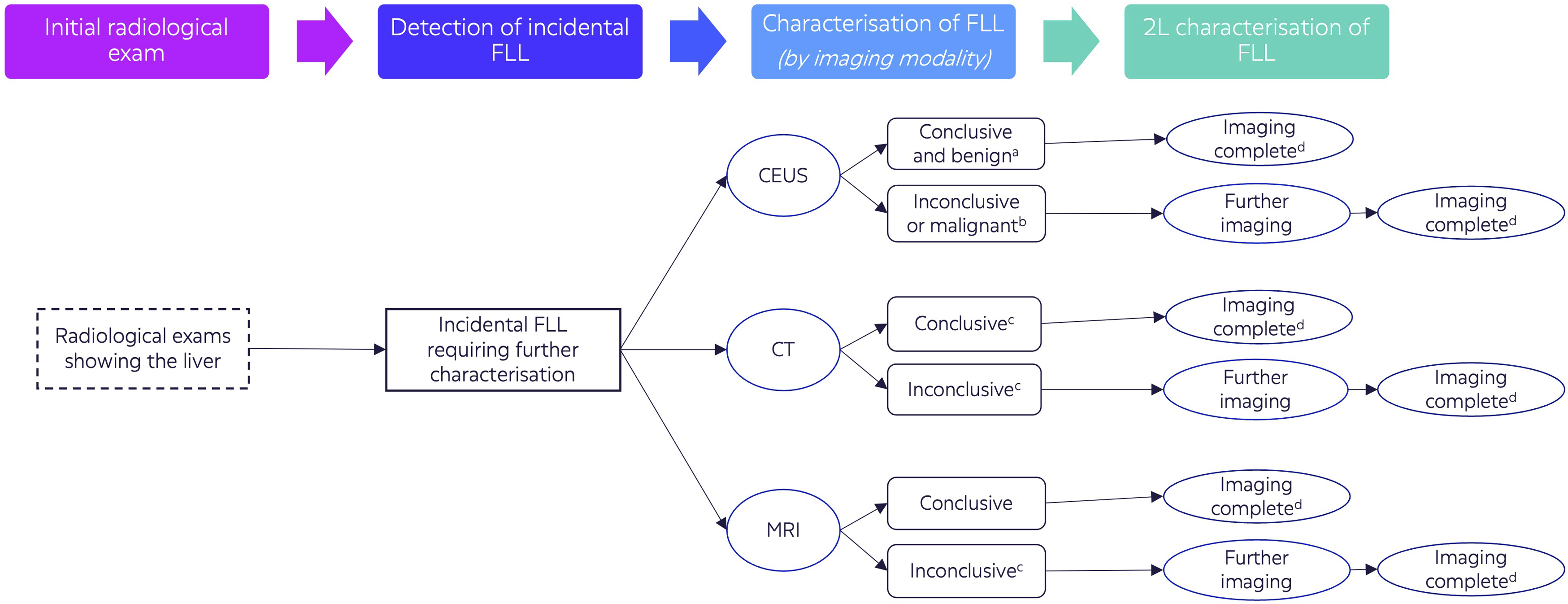
The first step in the patient flow varied by NHS Trust due to differing patient numbers; therefore, total NHS data based on the HES statistics was used in the base case. 21
For the ‘detection of incidental FLL’ step, the stakeholders advised that although FLL may be detected in 33% of radiological exams showing the liver, only 5% would require further examination. This is due to the majority of FLL identified being simple cysts that can be characterised immediately by the detection modality.
The proportion receiving each modality in the ‘characterisation of FLL’ was not considered to be representative due to low current usage of CEUS (≤5%–30%, with varying use by Trust). Stakeholders also described that CT and MRI would typically be performed with contrast, and if malignancy is detected, further imaging may be required to investigate the primary liver tumour in the case of liver metastases.
For the final step of ‘second-line characterisation’, stakeholders provided additional clinical context, where conclusive results are available in up to 90% of benign lesions with CEUS. Further imaging in 10% of cases occur with CT and MRI and the same situation can occur with CT and MRI as the first exam.
Model outputs
The model was developed to compare a world without CEUS use compared to a world with CEUS. The base case assumed CEUS use in 5% of cases to characterise FLL, aligned with current usage. This was equivalent to 5891 out of 117,830 patient episodes. This resulted in cost savings of £224,790 per year to the NHS (Figure 4).
Sensitivity analysis
Sensitivity analyses of the base case were carried out to examine the impact of key assumptions made for the model (Figure 3). The analysis showed that regardless of the changes made, CEUS still resulted in cost savings to the NHS if used in only 5% of cases (Table 3). Overview of scenarios explored in the sensitivity analysis. 2 L: Second-line; CEUS: Contrast-enhanced Ultrasound; CT: Computed tomography; FLL: Focal liver lesion; Magnetic Resonance Imaging; US: Ultrasound. Outputs of the sensitivity analysis. Base case assumptions: a5% prevalence was assumed in the model. bCT and MRI with contrast was assumed. cInconclusive results in 10% of cases was assumed. dCEUS for 2 L characterisation was assumed in 5% of cases. Pink represents a lower cost saving whereas green shows a high cost saving with CEUS. 2 L: Second-line; CEUS: Contrast-enhanced Ultrasound; CT: Computed tomography; FLL: Focal liver lesion; Magnetic Resonance Imaging.

The assumption of examining with CT and MRI without contrast had the greatest impact on cost savings, reducing them from £224,790 to £23,565; however, this scenario was deemed unlikely unless patients were considered at risk of adverse reaction to contrast media.
A positive impact on cost savings identified from the sensitivity analysis was the prevalence of FLL of which would require characterisation. If this number was similar to that identified from the literature, cost savings with CEUS in 5% of cases could be £1,483,610.
Scenario analysis
The scenario analysis explored the impact of increasing CEUS use (Figure 4). In scenario 1, first-line characterisation using CEUS was adjusted to 50% (58,915 out of 117,830 episodes), leading to a cost saving of £2,247,894 per year. Scenario analysis of CEUS use to characterise FLL.
In scenario 2, standardising CEUS use for characterising FLL, with use of 90% (106,047 out of 117,830 episodes) demonstrated that cost savings of £4,046,208 per year could be achieved. A 90% cap was used assuming 10% of cases may be high-risk thus requiring initial characterisation with CT or MRI rather than CEUS.
Discussion
The outputs of the cost-minimisation model demonstrate that there is an opportunity to support the NHS in improving diagnostic services as aligned to the Long-Term Plan. The first being through cost savings to the NHS that could be generated by standardising CEUS use (in 90% of cases) for the first-line characterisation of FLL. Alongside cost savings, by adopting CEUS, NHS capacity can be improved by freeing up to 57,802 CT or 34,881 MRI appointments. This equates to 7.6% of CT appointments and 3.6% of MRI appointments. Furthermore, due to low set up costs of CEUS and short test time, diagnostic capacity could be expanded, thereby ensuring appointments for CT and MRI are reserved for the most severe and life-threatening cases, as well as supporting follow-up care with guidance of biopsies. An opportunity for implementation would be the incorporation of CEUS into diagnostic centres which would not only increase hospital capacity, but also support transitioning from diagnostic services from secondary and tertiary care to community care, while still preserve tertiary services for high-risk patients. An important consideration for implementation in the community setting, with limited medical teams, is the increasing sensitivities to contrast requiring emergency treatment and the limitation of CEUS where it can be difficult to characterise small lesions in the upper part of the right liver lobe, that is, segment 8 in some patients. Therefore, patients should be identified and referred appropriately according to risk and location of lesion.
Current demand for diagnostic services is outstripping capacity and is a major contributor to NHS funding pressures. 22 This is reflected by high wait lists with 1,593,000 patients waiting to receive a diagnostic service and up to 30.5% waiting 6+ weeks. 23 CEUS provides an opportunity to reduce wait lists and time to testing, particularly as CEUS can be integrated as part of the initial ultrasound session when an FLL is first detected, saving time for referral and reporting. This aligns to author’s conclusions from Streb et al. (2019) where 78.4% of examinations with CEUS were performed the same day as the initial examination by ultrasound, thus avoiding delays incumbent with scheduling follow-up appointments. 17 Examinations with CT and MRI are associated with time to test of 5 and 22 days, and time to report of 1 and 3 days, respectively, 24 although it should be noted these are averages with wide margins contributing to the high waiting lists. By using CEUS which is associated with short lead times, reduced referrals and immediate results, patient anxiety could be reduced. This is also supported by author conclusions from Choi et al. (2016), a retrospective study in South Korea, where patient anxiety could be minimised with reduced unnecessary follow-up 25 .
The strength of this analysis was the design ensuring representation of the NHS across the UK. This involved incorporating NHS Trust stakeholders across different regions ensuring that regional differences in patient management were captured. The second element contributing to the representative design was incorporating both low and active users of CEUS to consider varying perspectives and allow for greater sensitivity of the model. There are however weaknesses due to the logistical challenges in accessing patient-level data, leading to reliance on published literature, NHS data, and expert opinions for validation. However, this is commonly recognised for economic evaluations, with NICE methods acknowledging that decision-making should proceed despite weaknesses in the evidence base, provided the highest level of evidence generation has been included. 26 Regardless, a prospective study to capture patient-level data is recommended to improve rigour, expand the results, and assess the potential for adopting a CEUS service in specific NHS Trusts using tailored data. Another limitation was that all stakeholders were from university hospitals, which could have biased the results towards more challenging cases that require initial examination with CT or MRI due to inherent risk factors, meaning results may differ for district general hospitals. Despite these limitations, similar findings are expected in terms of directional cost savings that can be achieved with increasing CEUS use. This is supported by the sensitivity analysis on the core assumptions that showed cost savings could still be realised. Other analyses such as assessing risk factors for malignancy and resource availability, could further understand the impact on resource savings. Although the model only examined the flow of patient episodes, integrating these aspects in the future could provide a better understanding of the full patient pathway.
In conclusion, this patient flow review and cost-minimisation analysis demonstrated substantial resource savings when increasing the use of CEUS for the first-line characterisation of FLL compared to traditional approaches of CT and MRI. These results further support current NICE guidance recommending CEUS as a cost-effective option, whereby CEUS could be included in the diagnostic pathway as an option to preserve the use of limited CT and MRI capacity for higher priority cases. Furthermore, by introducing a standardised pathway using CEUS to increase diagnostic imaging capacity, this can deliver cost savings and improve the critical economic situation of the NHS, whilst reducing wait times and patient anxiety without compromising diagnostic quality.
Supplemental Material
Supplemental Material Assessing the impact and resource implications of contrast-enhanced ultrasound on workflow of patients with incidental focal liver lesions on the UK national health service
Supplemental Material for Assessing the impact and resource implications of contrast-enhanced ultrasound on workflow of patients with incidental focal liver lesions on the UK national health service by Megan O’Brien, Louisa Oliver, Nick Proctor, Maro Siakantari, Peter Cantin, Colin Griffin, and Ben Stenberg in Acta Radiologica Open
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Access Infinity received budgetary compensation from Bracco who market SonoVue® for the conduct of the analysis and writing of this publication.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Bracco.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
