Abstract
Primary ectopic meningiomas, although widely reported in multiple sites of the body, are particularly rare in the pleura. Here, we report a 35-year-old asymptomatic woman who was found to have a large mass in the right pleural area on physical examination chest radiography. Chest CT scan showed a large irregular mass from the right second anterior costal pleura to the right supradiaphragm, in which calcified plaques of varying sizes were widely and heterogeneously distributed. The mass was connected to the pleura (anterior rib pleura, mediastinal pleura, diaphragmatic pleura) in a wide base, with oblique “Z” changes in the coronal view. After the contrast agent injection, the mass exhibited mild enhancement on both arterial and venous phase scans. Furthermore, a linear enhancement that was indicative of “pleural tail sign” changes in the pleura adjacent to the mass was observed. The disease was preoperatively misdiagnosed as malignant pleural mesothelioma, and the postoperative pathological diagnosis was right pleural meningioma (gritty type). Therefore, we carefully analyzed its imaging features and differential diagnosis by consulting relevant literature.
Introduction
Meningiomas are one of the most common tumors of the central nervous system, while meningiomas occurring outside the central nervous system are called ectopic meningiomas (Ectopic Meningioma, EM); the latter is a tumor that arises in tissues and organs without meningeal coverage, but has a meningioma morphological structure. Ectopic meningiomas are classified as primary (primary ectopic meningioma, PEM) and secondary (secondary ectopic meningioma, SEM). The primary type is usually benign and rare, accounting for only 1%–2% of all central nervous system tumors. 1 The primary ectopic meningioma reported here occurs in the right pleura, which is extremely rare in the previous literature. Due to the lack of understanding, it was misdiagnosed as malignant pleural mesothelioma (MPM) on the right side. Thus, we thoroughly analyzed the patient’s clinical presentation, imaging characteristics, and potential differential diagnoses, and critically reviewed the pertinent literature to raise awareness of this condition.
Case presentation
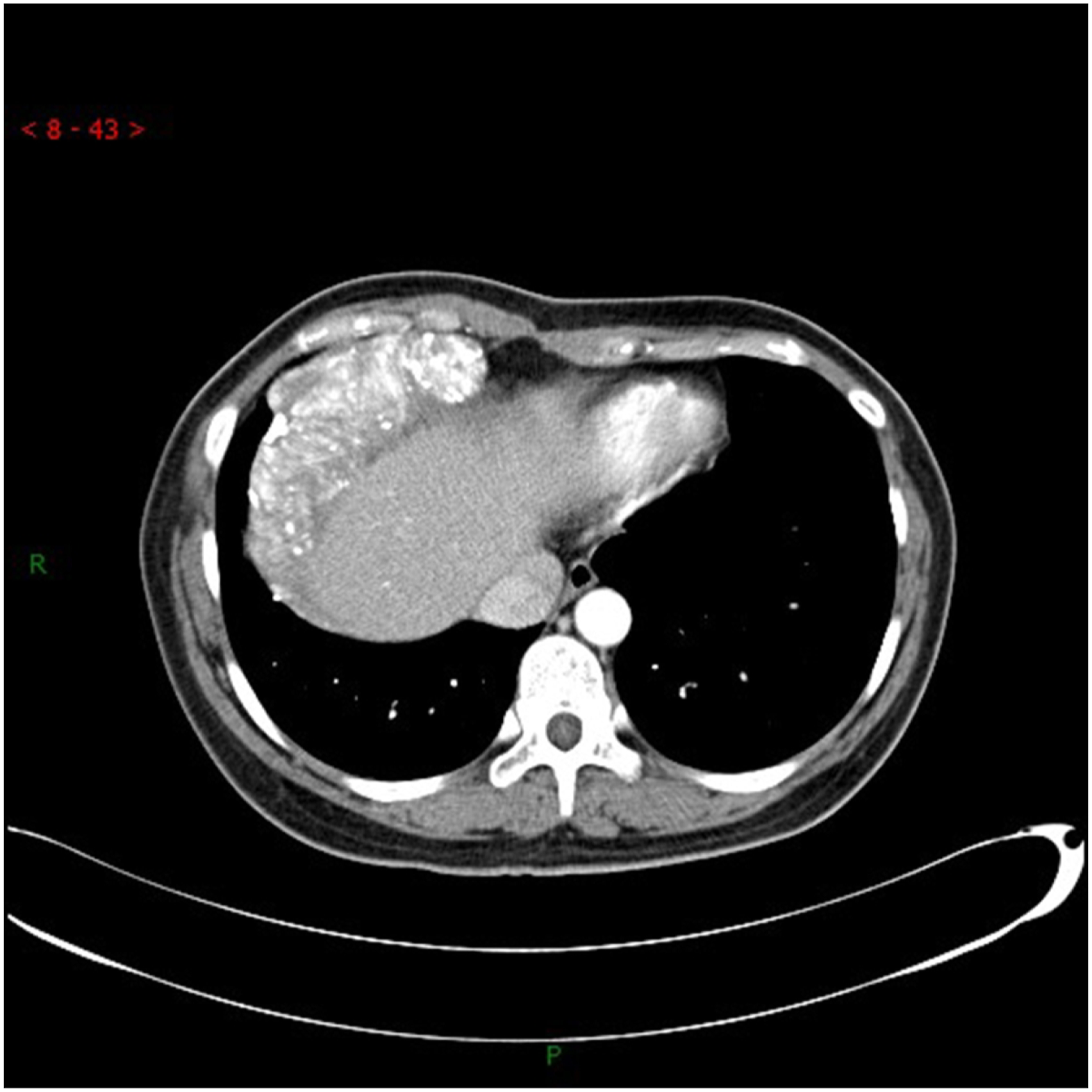
A 35-year-old female was found to have a right thoracic mass on chest X-ray examination. Thorax, laboratory tests, cranial CT scans and whole-body bone scintigraphy, and past history showed no abnormalities. Chest CT scan using US For the national GE Lightspeed 64-slice spiral CT scanner, the scanning parameters were 120 kV, current 250 mA, 0.6 s per week, pitch 0.984, and collimation 0.625 mm. At the end of the injection, the concentration was 320 mg/mL non-ionic contrast medium iopamidol, the dosage was 1 mL/kg, and the flow rate was 2.5 mL/s. Chest CT mediastinal window showed a large irregular mass from the right second anterior costal pleura area to the right supradiaphragm. The maximum slice size was about 9.8 cm × 5.9 cm, with uneven density. There were extensive calcifications of different sizes, without exact signs of necrosis or cystic changes. The lesion’s anterior, left, and lower margins were connected to the anterior costal pleura, mediastinal pleura, and diaphragmatic pleura respectively, with a wide base. Additionally, in the coronal view, it displayed oblique “Z” changes that were distinct from surrounding structures, such as the liver and heart. The mass was slightly enhanced in the arterial and venous phases, and linear abnormal enhancement appeared to show “pleural tail sign” changes in the pleura adjacent to the mass, but no effusion was observed in the thoracic cavity. The lung window showed that the mass pushed adjacent to the lung parenchyma, and no other abnormalities were observed. Preoperative CT diagnosed malignant mesothelioma of the right pleura. Intraoperatively, a few zonal adhesions of the right pleura were observed. A tumor mass, measuring approximately 10.0 cm × 6.0 cm, was observed in the right pleural area. The mass exhibited both narrow and wide components and was closely associated with the chest wall and diaphragm. Intraoperative freezing revealed a hyperplastic shuttle cell with numerous gritty bodies. Postoperative immunohistochemistry: S-100 (−), SOX-10 (−), GFAP (−), EMA (+), PR (−), Claudin-1 (−), SSTR-2 (−), KI67 < 5% (+), CD34 scattered (+), ATAT-6 (−). Pathological diagnosis: Right pleural meningioma (gritty).
Discussion
Primary Ectopic Meningioma, PEM Cases have been reported in multiple sites such as the skin, 2 tongue, 3 and lung, 4 but reports of occurrence in the pleura are very rare. The pathogenesis of this condition remains unclear, and its origin is a subject of controversy. Clinical presentation is typically non-specific and largely depends on the tumor’s location, size, and relationship with surrounding tissue structures. 5 The diagnostic criteria for this disease include: typical pathological features of meningioma; tissues and organs without arachnoid endothelial cells and meningeal involvement at the anatomical site; extracranial growth or extracranial metastasis that is not intracranial meningioma.
This case was located in the right pleural area without arachnoid cell coverage, and preoperative imaging revealed no history of central nervous system tumor, ruling out the possibility of secondary.
Conventional meningiomas usually occur in the intracranial and spinal canals, and CT mostly shows a slightly hyperdense mass with well-defined borders, which may be accompanied by necrosis, cystic degeneration, and calcification. There were variations in enhancement intensity observed between different subtypes, and most of the parenchymal components displayed significant homogenous enhancement. Some tumors are accompanied by a “tail sign” at the edge, and adjacent bone is often accompanied by hyperplasia and sclerosis. In this case, the tumor presented as a mass with mixed density and multiple calcified plaques. There were “pleural tail signs” at the edge of the enhanced scan, well-defined borders of the mass, and hyperplasia and sclerosis of the anterior ribs in the adjacent part. These findings are similar to the typical signs of meningioma. Among them, multiple calcified plaques were the characteristic signs in the present case, that is, it leads to increased and heterogeneous density of the tumor on CT. Gritty Meningiomas are often accompanied by numerous calcified plaques and even complete calcification of the entire tumor. 6 For the calcification of meningiomas, Nakasu S et al. 6 scholars reported that it is one of the markers of slow tumor growth rate. In this particular case, the large tumor mass had already compressed the surrounding structures, but the patient did not display any corresponding clinical symptoms. This led us to speculate that the slow growth rate of the tumor may have allowed the body to compensate for the mass more efficiently. It is worth noting that the main components of the tumor are soft tissue and calcified plaques. At the same time, there is only slight enhancement in the soft tissue area on contrast-enhanced scans, which is consistent with a case of primary posterior mediastinal gritty meningioma reported by Lu C et al., 1 but it is inconsistent with significant enhancement of typical gritty meningioma. 7 The possible reasons for the analysis were that the tumor calcified plaques were widely distributed, and the measurement of tumor CT enhancement values was affected by the partial volume effect of calcifications and the interference of adjacent rib bone opacities. Lee et al. 8 further reported that MRI enhancement does not depend on the presence of calcified tissue and that most of the tumor signal is enhanced even in fully calcified meningiomas. It is well known that MRI has high soft tissue resolution and can more accurately reflect the cystic degeneration, necrosis degree and blood supply of the tumor. Regrettably, no MRI scan was done on the patient, which could have provided additional information. Although the final diagnosis of this case relies on immunohistochemistry, we can identify the extent, contour, and growth pattern of the lesion, as well as its relationship with adjacent tissues and organs preoperatively by imaging examination, which is also very important to determine the best treatment for this case and reduce the risk of complications of surgery. 9 The patient in this case recovered well following the surgical procedure and did not report any significant discomfort during the 4-month follow-up period. Imaging reexamination revealed no evidence of tumor recurrence or distant metastasis.
This disease is preoperatively misdiagnosed as malignant pleural mesothelioma (MPM). First, the authors did not consider the malignant growth characteristics of MPM, such as its ability to invade the pleura, chest wall, ribs, and other surrounding structural tissues, as well as its potential for lymph node and distant metastasis. In contrast, gritty meningiomas are typically benign tumors classified as WHO grade I, and we did not find malignant signs from CT images. Second, typical signs of MPM are associated with pleural effusion in addition to extensive irregular pleural thickening,10,11 which is not consistent with this disease. The authors analyzed that the reason for the absence of pleural effusion may be that the tumor did not disrupt the normal filtration and recharging homeostasis of the pleura. On the other hand, a substantial body of literature suggests that MPM with calcified plaques is mostly associated with asbestos contact, with calcified plaques typically found distributed on the pleural surface.
11
In this case, the calcified plaques of the mass are widely and unevenly distributed, and many mineralized concentric structures and psammomatous bodies are found in the tumor and at the edge under electron microscope, so there are significant differences in the distribution and pathogenesis of calcified plaques between the two. In addition, the imaging features of metastatic pleural disease (MPD) and MPM are similar, and the former generally has a clear history of primary malignancy and related clinical symptoms, so the two are easily differentiated. Finally, we also need to differentiate it from pleural thickening and calcification due to non-tumors such as trauma, infection, and pneumoconiosis. This condition is not an independent disease, but rather a complication or residual change resulting from the acute onset of the aforementioned diseases. When the cumulative extent of such pleural lesions is significant, it is frequently accompanied by symptoms such as chest pain, ventilatory dysfunction, and thoracic collapse. This differs from the case we reported (Figures 1–5). Chest CT revealing a large irregular mass extending from the right second anterior pleural rib to the right diaphragm, with calcified plaques of varying sizes widely distributed within. Chest CT revealing a large irregular mass extending from the right second anterior pleural rib to the right diaphragm, with calcified plaques of varying sizes widely distributed within. Mass exhibiting a wide base connected to the pleura (anterior costal pleura, mediastinal pleura, and diaphragmatic pleura), with coronal plane displaying a slanted ‘Z' shape alteration. Hematoxylin and eosin (H&E) staining demonstrating granuloma-like structures inside the tumor. Immunohistochemistry indicating positive epithelial membrane antigen (EMA) presence. Hematoxylin and eosin (H&E) staining demonstrating granuloma-like structures inside the tumor. Immunohistochemistry indicating positive epithelial membrane antigen (EMA) presence.



In conclusion, PEM has a low incidence and lacks specific clinical manifestations, and imaging diagnosis is very challenging. Because its tissue origin and structure are consistent with central nervous system meningioma, there are common meningioma features on imaging, and mastering its imaging features and pathological changes is helpful for the diagnosis and differential diagnosis of the disease.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
