Abstract
Pulmonary arteriovenous malformation (PAVM) is a rare vascular anomaly, for which transcatheter embolization with metallic coils is the standard of care. Although detecting recanalization after embolotherapy is crucial, direct visualization of residual flow with computed tomography or magnetic resonance (MR) imaging is generally difficult due to metal artifacts. We present a case of recanalized PAVM after coil embolization detected by ultra-short echo time MR angiography using a modified signal targeting with an alternative radio frequency spin labeling technique.
Keywords
Introduction
Pulmonary arteriovenous malformations (PAVMs) are rare pulmonary vascular structures with direct communication between the pulmonary artery and pulmonary vein circulations through a thin-walled sac.1,2 Embolotherapy with metallic coils is considered as the standard treatment for PAVM.2,3 However, a major concern of embolotherapy is recanalization, in which some patients develop neurological events.4,5
Digital subtraction angiography (DSA) is the most reliable modality for detecting recanalization, although it is not routinely performed because of its invasiveness. In recent years, non-invasive techniques, such as computed tomography (CT) and magnetic resonance imaging (MRI), with or without contrast agents, have been used to detect the presence of PAVM recanalizations 6–12. However, direct visualization of recanalizations with CT and MRI is limited because of the prominent streak or susceptibility artifacts induced by metallic coils. Herein, we described a case of recanalized PAVM treated by coil embolization detected by ultra-short echo time magnetic resonance angiography using a modified signal targeting with alternative radio frequency spin labeling technique (UTE-ASTAR MRA), in which no susceptibility artifacts were detected.
Case Report
A 63-year-old woman with multiple PAVMs (eight lesions) in the bilateral lung concomitant with hereditary hemorrhagic telangiectasia was referred to our hospital for endovascular treatment. Transcatheter embolization of the feeding arteries and/or sacs with platinum and stainless-steel coils was performed in multiple sessions, resulting in the complete occlusion of each PAVM. A follow-up unenhanced CT scan 4 months later revealed that the shrinkage rates of the diameters of the draining veins of the two PAVMs in segment 4 of the right middle lobes treated with platinum coils were 48% and 45%, respectively, indicating the recanalization of these PAVMs.
11
However, it was difficult to visualize and evaluate the continuity between the feeding artery, sac, and draining vein because of severe streak artifacts induced by the metallic coils (Figure 1(a)). Evaluation of recanalization after the embolization of a pulmonary arteriovenous malformation (PAVM). (a) Unenhanced computed tomography with partial maximum intensity projection (MIP) for image evaluation around the placed coils is difficult to interpret due to the prominent streak artifact from the coils. The platinum coils were placed in the feeding artery of the anterior lesion and in the sac of posterior lesion. (b) Ultra-short echo time magnetic resonance image (UTE MRI) with partial MIP showing the continuity of the lateral feeding arteries (arrows), sac (arrowheads), and draining vein (dashed arrows) in both embolized PAVM. (c–e) UTE MR angiography using a modified signal targeting with alternative radio frequency spin labeling technique (UTE-ASTAR MRA). The images were presented with partial MIP. The delay time of images (c), (d), and (e) was 200, 506, and 812 ms, respectively. On the UTE-ASTAR MRA, the continuity of the feeding arteries (arrows), sacs (arrowheads), and draining veins (dashed arrows) were serially visualized from 200 to 812 ms in both PAVMs. Note that the blood signals were visualized in the placed coils. (f) Time-resolved contrast-enhanced MRA. Asterisks in (b)–(f) indicate the image distortion due to the magnetic susceptibility artifact from stainless-steel coils placed in the PAVM adjacent to target lesions.
Magnetic resonance imaging parameters.

aThe acquired slice sections of 2 mm were reconstructed with 1.0-mm voxel intervals by mid-slice reconstruction.
bThe acquired slice sections of 2.5 mm were reconstructed with 1.25-mm voxel intervals by mid-slice reconstruction.
cReconstruction matrix.
N/A, not applicable; UTE MRI, ultra-short echo time magnetic resonance imaging; UTE-ASTAR MRA, ultra-short echo time magnetic resonance angiography using a modified signal targeting with alternative radio frequency spin labeling technique; TR-CEMRA, time-resolved contrast-enhanced magnetic resonance angiography; TR, repetition time; TE, echo time.

Procedure for acquiring UTE-ASTAR MRA. Three-dimensional UTE images were serially acquired at 200, 506, and 812 ms of delay time after applying the ASTAR pulse. The ASTAR pulse (tag) was placed on the range that covered the right ventricle and main trunk of the pulmonary artery in the axial direction (dashed parallelogram). Note that the peripheral portion of the pulmonary arteries in the left lung field is gradually visualized from phase 1 to phase 3 on UTE-ASTR MRA.
For the UTE MRI, the continuity of the feeding artery, sac, and draining vein were visualized as high signal intensities in both treated PAVMs (Figure 1(b)). However, it was difficult to distinguish whether these signals were actual blood flow or thrombi. Thus, UTE-ASTAR MRA provided serial hemodynamic images and clearly visualized the blood flow signals inside the coils and the continuity of the feeding artery, sac, and draining vein (Figures 1(c) to (e)). In contrast, in the untreated lung field, only the pulmonary arteries were visualized, even at 816 ms. On TR-CEMRA, the feeding artery and draining vein of the treated PAVMs were simultaneously visualized in the pulmonary arterial phase (Figure 1(f)). Based on these findings, recanalization through the coils in the PAVMs in the right middle lobe was suggested. DSA performed in the secondary embolization revealed recanalization through the coils placed in the feeding arteries of both treated PAVMs (Figures 3(a) and (b)). These findings were consistent with those of UTE-ASTER MRA (Figures 3(c) and (d)). An additional platinum coil embolization of the recanalized lumen was performed. Digital subtraction angiography (DSA) during the second embolotherapy for the recanalized PAVM. (a) Anterior lesion. (b) Posterior lesion. The recanalization through the previously placed coils was confirmed in both lesions. Arrows, arrowhead, and dashed arrows indicate the feeding arteries, sacs, and draining veins, respectively. (c), (d) The partial MIP images of UTE-ASTAR MRA at the views corresponding to DSA images.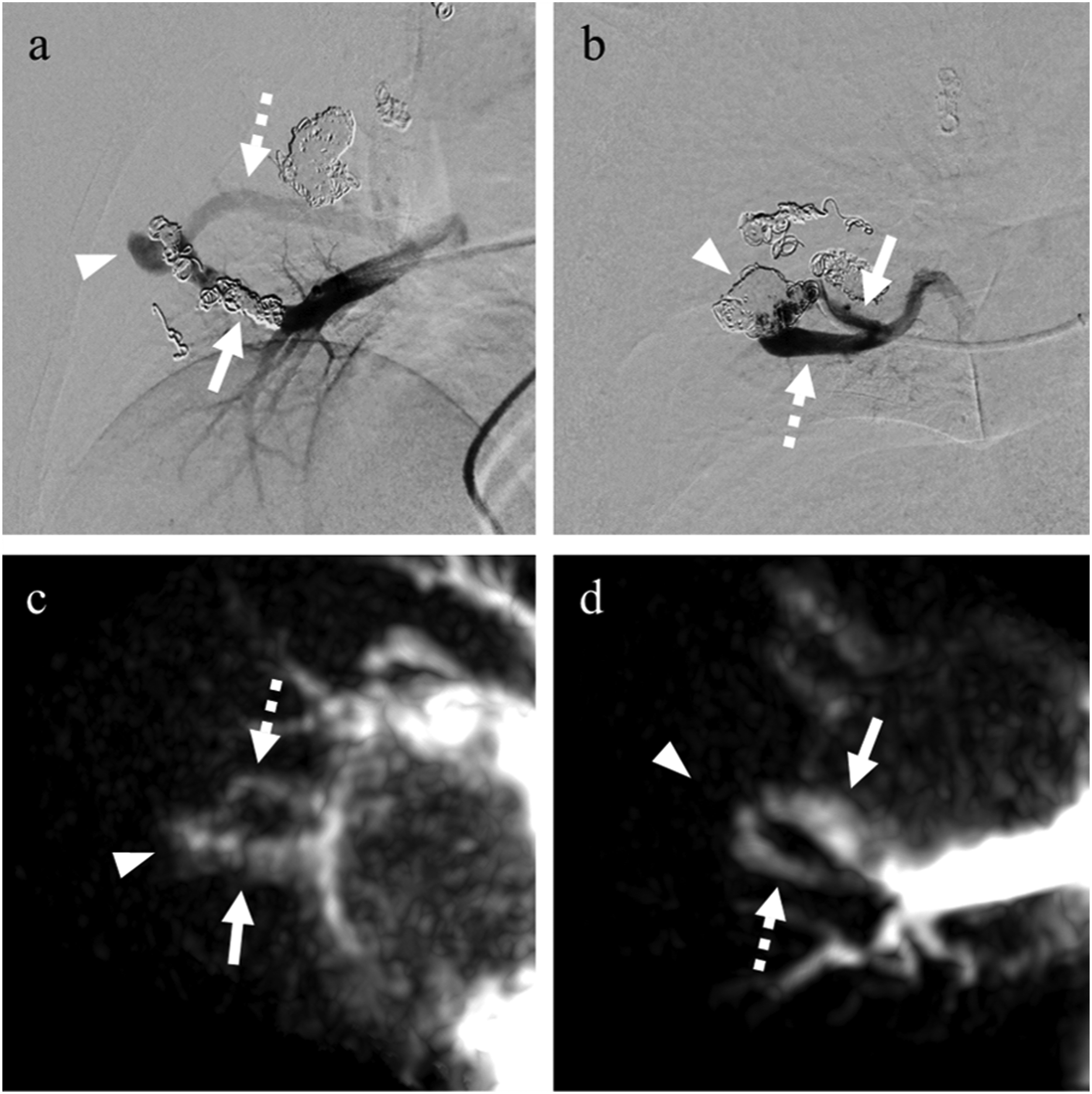
Discussion
In the present case, UTE-ASTAR MRA provided hemodynamic information using the ASL technique and enabled clear visualization of the continuity through the embolized coils from the feeding artery to the draining vein. These findings are similar to those of DSA.
Although it is important to evaluate the prevalence of recanalization because it can occur in up to 50% of embolotherapy patients and may cause neurologic events after embolization of PAVMs,4,5 the direct diagnosis of recanalization of PAVMs with CT or MRI remains a challenge. Recently, the utility of UTE MRI in the detection of PAVM recanalization has been reported. 14 This method visualizes the continuity through the embolized coils from the feeding artery to the draining vein, but physicians might misinterpret static tissues, including thrombi in coils and sacs as lesions, given their high signal intensity. Therefore, imaging modalities that provide hemodynamic information in addition to UTE MRI remain necessary to evaluate recanalization. UTE-ASTAR MRA could provide useful information for the non-invasive definitive diagnosis of recanalization. To the best of our knowledge, this is the first report demonstrating the utility of UTE with ASL in the detection of PAVM recanalization after metallic coil embolization.
Similar to the present report, several clinical studies have reported that UTE with ASL reduces the artifacts typically observed after coil or stent placement in intracranial lesions.15–17 However, applying this technique in evaluating vascular lesions in the body, such as PAVMs, remains challenging given that additional techniques, including respiratory and/or electrocardiogram synchronization, are required to acquire images of the body trunk. Recently, Mori et al. reported the utility of UTE MRA with time-spatial labeling inversion. Their method used the ASL technique with deep learning reconstruction for the assessment of a renal artery aneurysm treated with metallic coils. 18 This method enabled visualization of the blood flow signals adjacent to the placed metallic coils, which was similar to our case, but it still did not provide serial hemodynamic images similar to those of UTE-ASTAR MRA or DSA. As described above, the continuous visualization of the feeding artery, sac, and draining vein through the placed coils is crucial to achieve a definitive diagnosis of recanalization. Therefore, UTE-ASTAR MRA might be more suitable for assessing recanalization of embolized PAVMs.
It is notable that UTE-ASTAR MRA can provide high temporal resolution similar to those of hemodynamic images at 300 ms intervals without contrast medium. Generally, the temporal resolutions of DSA and TR-CEMRA are approximately 100 ms and 2000 ms, respectively. Our method, which has a suitable temporal resolution comparable to that of DSA, was superior to TR-CEMRA in detecting recanalized PAVMs. In addition, it was also useful for the initial diagnosis of PAVMs and therapeutic effect even in other body lesions treated with metallic devices, including coils and stents. Indeed, we confirmed that UTE-ASTAR MRA could provide hemodynamic images in untreated PAVMs and detect residual blood flow in visceral artery aneurysms treated with platinum coils (unpublished data).
UTE-ASTAR MRA has additional advantages over contrast-enhanced CT and TR-CEMRA. First, this method does not require exposure to ionizing radiation or iodine- or gadolinium-based contrast agents, which may result in nephrotoxicity, anaphylaxis, nephrogenic systemic fibrosis, and tissue deposition of gadolinium. This might be particularly useful for young and/or pregnant patients with PAVM who require regular follow-up over an extended period. Second, UTE-ASTAR MRA has a relatively briefer acquisition window in the late expiration phrase, which was triggered by respiratory gating results in reduction-in-motion artifacts, compared with the settings of TR-CEMRA.
Despite these advantages, UTE-ASTAR MRA has some limitations compared to TR-CEMRA and CT. First, the acquisition time of UTE-ASTAR MRA was approximately 10–12 min, which was much longer than that of TR-CEMRA and CT. Second, UTE-ASTAR MRA had limits on the number of acquisitions in a single session, and therefore it was difficult to cover the entire lung field with high spatial resolution during a single session. However, given that there was no time constraint with UTE-ASTAR MRA, the image of the entire lung field could be acquired by planning multiple sessions. Third, the spatial resolution of UTE-ASTAR MRA is relatively low compared with that of CT, owing to the susceptibility to artifacts and limitations of slice thickness imposed by the available equipment. This drawback may be avoided using a higher magnetic-field MR imaging scanners that can achieve higher signal-to-noise ratios or using a newly developed deep learning reconstruction. Finally, the capability of UTE-ASTAR MRA to detect minor signals from residual flow might depend on the packing density and material of the coils. In this case, severe image distortion could occur around the stainless-steel coils. Therefore, the application of UTE-ASTAR MRA for lesions treated with stainless-steel coils might be limited.
In conclusion, the present report suggests that UTE-ASTAR MRA is a useful method for detecting recanalization of PAVMs after coil embolization without contrast injection and radiation exposure.
Footnotes
Acknowledgements
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. We thank Yoshimasa Ikeda and Yusuke Ayabe for their technical advice regarding magnetic resonance imaging.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
