Abstract
Background
Technological developments have led to an increased usage of external-body radiotherapy (RT) for the treatment of hepatocellular carcinoma (HCC). Transcatheter arterial chemoembolization (TACE) may be required later in patients treated with RT because of the high recurrence rate and multinodular presentation of HCC. However, despite the risk of liver function impairment, the cumulative liver damage correlated with TACE following a hepatic RT has not been adequately assessed.
Purpose
To evaluate the feasibility of TACE following RT for HCC.
Materials and methods
Sixty-seven patients with HCC who underwent TACE after RT were retrospectively evaluated between 2012 and 2018. We assessed increases in Child–Turcotte–Pugh (CTP) by ≥2 points at 1 month, the incidence of major complications, survival duration, and short-term mortality within 6 months after TACE. Furthermore, we evaluated the predictive factors for liver function impairment and short-term mortality.
Results
Eight patients experienced a CTP increase ≥2 points at 1 month. There were no cases of liver abscesses or bilomas. Nine patients died within 6 months following TACE. The mean liver dose (MLD) was a significant predictor of liver function impairment at 1 month (p = 0.042). Low liver functional reserve, distant metastasis (p = 0.037), MLD (p = 0.046), TACE type (p = 0.025), and TACE within 3 months following RT (p = 0.007) were significant predictors of short-term mortality.
Conclusions
Despite the feasibility of TACE following RT, clinicians should pay attention to impaired pretreatment liver function, following high dose RT, and the short duration between RT and TACE.
Introduction
Hepatocellular carcinoma (HCC) is a leading cause of global cancer-related death. According to the Barcelona Clinic Liver Cancer (BCLC) algorithms, there are several treatment options depending on the tumor stage.1,2 Transcatheter arterial chemoembolization (TACE) and systemic chemotherapy are recommended after the intermediate stage. However, treatment results are still unfavorable and various non-recommended treatments or a combination of treatments are used in clinical practice.1,3–5
Historically, external body radiation therapy (RT) mediated the development of radiation-induced liver disease (RILD) and was not used for the treatment of HCC. Recent technological developments facilitate the delivery of large doses to the tumor, while avoiding the healthy liver parenchyma around it have led to an increased usage of RT for the treatment of HCC.4–7 RT is often applied for the treatment of macrovascular invasion (MVI) associated with HCC. Furthermore, it is used as a palliative therapy for patients with poor prognostic features or with physical or technical unavailability to other treatments.1,4,6,8
TACE may be required later in patients treated with RT because of the high recurrence rate and multinodular presentation of HCC.1,9 Despite the substantial risk of liver function impairment associated with RT and TACE,10,11 researchers have not adequately assessed the cumulative liver damage correlated with TACE following a hepatic RT. The aim of this study was to assess the feasibility of TACE following RT. Additionally, we investigated the factors associated with impairment of liver function and short-term mortality after TACE following RT.
Materials and methods
Ethics
This multi-center, retrospective study was approved by the institutional review board at our institution (No. B190130). The study protocol complied with the ethical guidelines of the Declaration of Helsinki and those of the Ministry of Health, Labor, and Welfare for medical and health research involving human subjects in Japan. Informed consent was obtained from all participants.
Subjects
In this retrospective cohort study, we reviewed the records of 79 patients between January 2012 and December 2018 who met the following criteria: (a) HCC on cross-sectional images; (b) RT as treatment for HCC; and (c) TACE following RT for the treatment of the same or other nodules regardless of the duration between RT and TACE. We excluded patients who had incomplete data (n = 2), received transcatheter arterial embolization for the treatment of ruptured HCC (n = 1) or transcatheter arterial infusion chemotherapy (n = 7), or received RT more than once (n = 2). Consequently, 67 patients were enrolled.
Study outcome and definition
The primary outcomes comprised impairment in liver function, incidence of major complications, survival duration, and short-term mortality. Liver function impairment was defined as an increase in CTP score by ≥2 points at 1 month. This was clinically more relevant than a transient decompensation after TACE.12,13 TACE outcome was evaluated using objective response (OR) with modified Response Evaluation Criteria in Solid Tumors (mRECIST), 14 and major complications, such as the formation of biloma or liver abscess in the treated area, were assessed a month after TACE. 15 The purpose of TACE ranges from curative to palliative depending on the patients’ status and tumor stage. Therefore, an assessment of the objective response for each lesion was unsuitable for this study. We assessed the survival duration and short-term mortality, defined as death within 6 months following TACE 16 to determine the feasibility of the technique.
Radiation therapy technique
The patients underwent a 3D conformal radiation therapy (3DCRT) with an EXL-15DP (Mitsubishi Heavy Industries, Tokyo, Japan) with free breathing. The gross tumor volume (GTV) was defined as the primary hepatic tumor or macrovascular invasion, referring to the computed tomography (CT), magnetic resonance imaging (MRI), and angiography findings. The internal target volume (ITV) was defined as GTVs obtained from both the exhalation and inhalation CT. The planning target volume (PTV) included the ITV, plus a 5-mm margin in all directions. The treatment goal was 45–50 Gy in 15–25 fractions to the isocenter of the PTV.
Stereotactic radiation therapy (SRT) was performed using CyberKnifeTM (Accuracy Inc., Sunnydale, CA, USA) (CK) or True BeamTM/STx (TB-STx) (Varian Medical System, Palo Alto, CA, USA). Delineation was performed in each phase on the 4-dimensional CT, referring to CT, MRI, and angiography. The combination of multiple GTVs was used to define the ITV. The PTV included ITV with 2–5-mm margins, considering daily set-up variations. Gold fiducial markers (GFMs) were implanted near the target for tumor tracking by respiratory synchrony in all patients treated with CK. GFM was implanted in most patients to perform daily image-guided RT for TB-STx. Furthermore, gating methods were performed to account for their respiratory motion. The goal of SRT was to deliver 45–55 Gy in 4–8 fractions. The dose that covered 95% of the PTV (PTV D95) was the prescribed dose.
TACE technique
The choice of conventional TACE (cTACE) or drug-eluting beads (DEB-TACE) was determined by consensus between interventional radiologists and hepatologists and was based on the number, size, and distribution of lesions and the global liver function of the patients. HCC lesions within four tumors of the 7 cm criterion 17 were treated with cTACE. In contrast, lesions over the four tumors of the 7 cm criterion were treated with DEB-TACE. Regardless of the number and size of the tumors, DEB-TACE was selected for patients whose treatment range exceeded one lobe, older patients, and those with poor performance status because DEB-TACE is thought to have less post-embolization syndrome. Furthermore, it might provide a better safety profile because of the less systemic absorption of chemotherapy.1,9,18,19
Board-certified interventional radiologists with 14–18 years of experience performed all procedures under local anesthesia with 1.0% procaine. A 4-F catheter was advanced into the coeliac trunk or common hepatic artery over the 0.035-inch guide wire through an introducer sheath, via the common femoral artery. An iodinated contrast agent was used to advance the coaxial microcatheter into the feeding arterial branches after the angiograms.
cTACE was performed with a maximum dose of 50 mg and 8 mL epirubicin and Lipiodol (Guerbet, Pairs, France), respectively. Gelatin sponge particles (Gelpart: Nippon-Kayaku, Tokyo, Japan) were used for the embolization of the feeding artery following a chemotherapy injection. Super selective embolization was utilized in all possible cases. Additionally, the parasitic blood vessels were embolized if necessary. DEB-TACE was performed with 100–300 μm DC beads (BTG, London, UK) loaded with 50 mg of epirubicin or 50–100 μm Hepasphere (Nippon-Kayaku, Tokyo, Japan) loaded with 50 mg of fine powder cisplatin (IA-call; Nippon-Kayaku). 20 Embolization was performed until stasis in the tumor feeding vessels, preserving flow in the segmental and lobar arteries, based on the angiogram.
Data collection
The demographic, clinical, laboratory, and radiological datasets were recorded before and after the treatments. The tumors were staged according to BCLC algorithm. We assessed the liver function by the CTP, MELD, MELD-NA, and ALBI scores.21,22 RT and TACE criteria included the treatment type, range, and location. The RT type was divided into 3DCRT or SRT. The MLD and number of fractions (≤10 or >10) were also assessed. TACE type was divided based on the embolic agents used in cTACE or DEB-TACE. The range of RT was defined as v30 boundary and classified into segmental, lobar, and hilar categories. The range of TACE was classified into segmental, lobar, and bilobar, while referring to the available embolized vessels and non-contrast CT scan immediately after TACE. Furthermore, we assessed the location of TACE, inside or outside the radiated field, defined as v30. Information on the history of systemic therapy administration was collected.
Statistical analysis
We used summary statistics (means, medians, ranges, and standard deviations for quantitative variables and frequency and percentage for categorical variables) to describe the characteristics of the population. A paired t-test or Wilcoxon signed-rank test was used to compare the continuous variables, before and after the treatment. We conducted a univariate logistic regression analysis to evaluate the risk factors for both liver function impairment and short-term mortality. Multivariate analysis was not performed because the small sample size and low number of events per variable could potentially lead to inconclusive results. 23 We performed the statistical analysis using SPSS version 25 (IBM, NY, USA) and MedCalc version 19 (MedCalc Software Ltd, Ostend, Belgium). p-values were evaluated as two-tailed and the significance was set at p < 0.05.
Results
Study population and tumor/treatment characteristics
Characteristics of the patients.

Note: Data are summarized as counts (percentage) and median (interquartile range) for the categorical and continuous variables, respectively.
RT: radiation therapy; HBV: hepatitis B virus; HCV: hepatitis C virus; CTP: Child–Turcotte–Pugh; BCLC: Barcelona Clinic Liver Cancer; MVI: macrovascular invasion; TACE: transcatheter arterial chemoembolization; TAI: transarterial infusion; MLD, mean liver dose; 3DCRT: 3D conformal radiation therapy; SRT: stereotactic radiation therapy; cTACE: conventional TACE; DEB-TACE, drug-eluting beads TACE.
a Some patients had more than one etiology.
Objective response, liver function impairment, and major complications at 1 month
Laboratory data and tumor status before and 1 month following TACE and major complications within a month.

Note: Continuous laboratory data are presented as mean ± standard deviation, continuous tumor data are presented as median (range), and categorical data are presented as counts (percentage).
TACE: transcatheter arterial chemoembolization; T-Bil: total serum bilirubin; PT-INR: prothrombin time internal normalized ratio; CTP: Child–Turcotte–Pugh; ALBI: albumin bilirubin score; MELD: model for end-stage liver disease; MELD-NA: MELD-sodium.
Neither abscess nor biloma was detected within a month (Table 2).
Survival duration and short-term (<6 months) mortality
The median and mean survival durations were 17.5 and 25.6 months, respectively. The 12-, 24-, and 36-month survival rates for the entire study population were 69.4%, 44.3%, and 32.4%, respectively (Fig. 1). Nine patients (13.4%) died within 6 months. However, there was no death within 1 month following TACE. Table 3 summarizes the demographics of these nine patients. Overall survival for all patients. The results are represented using the Kaplan–Meier method. The numbers of patients at risk are denoted on the x-axis. Patients who died within 6 months following TACE. CTP: Child–Turcotte–Pugh; ALBI: albumin bilirubin score; MELD: model for end-stage liver disease; MELD-NA: MELD-sodium; BCLC: Barcelona Clinic Liver Cancer; MVI: macrovascular invasion; MLD: mean liver dose; NBNC: non-B non-C; RT: radiation therapy; HBV: hepatitis B virus; HCV: hepatitis C virus; GI bleeding: gastrointestinal bleeding; TACE: transcatheter arterial chemoembolization; MLD: mean liver dose; 3DCRT: 3D conformal radiation therapy; SRT: stereotactic radiation therapy; cTACE: conventional TACE; DEB-TACE: drug-eluting beads TACE.

Risk factors for liver function impairment and short-term mortality
Predictors of liver function impairment at 1 month and for short-term mortality within 6 months.
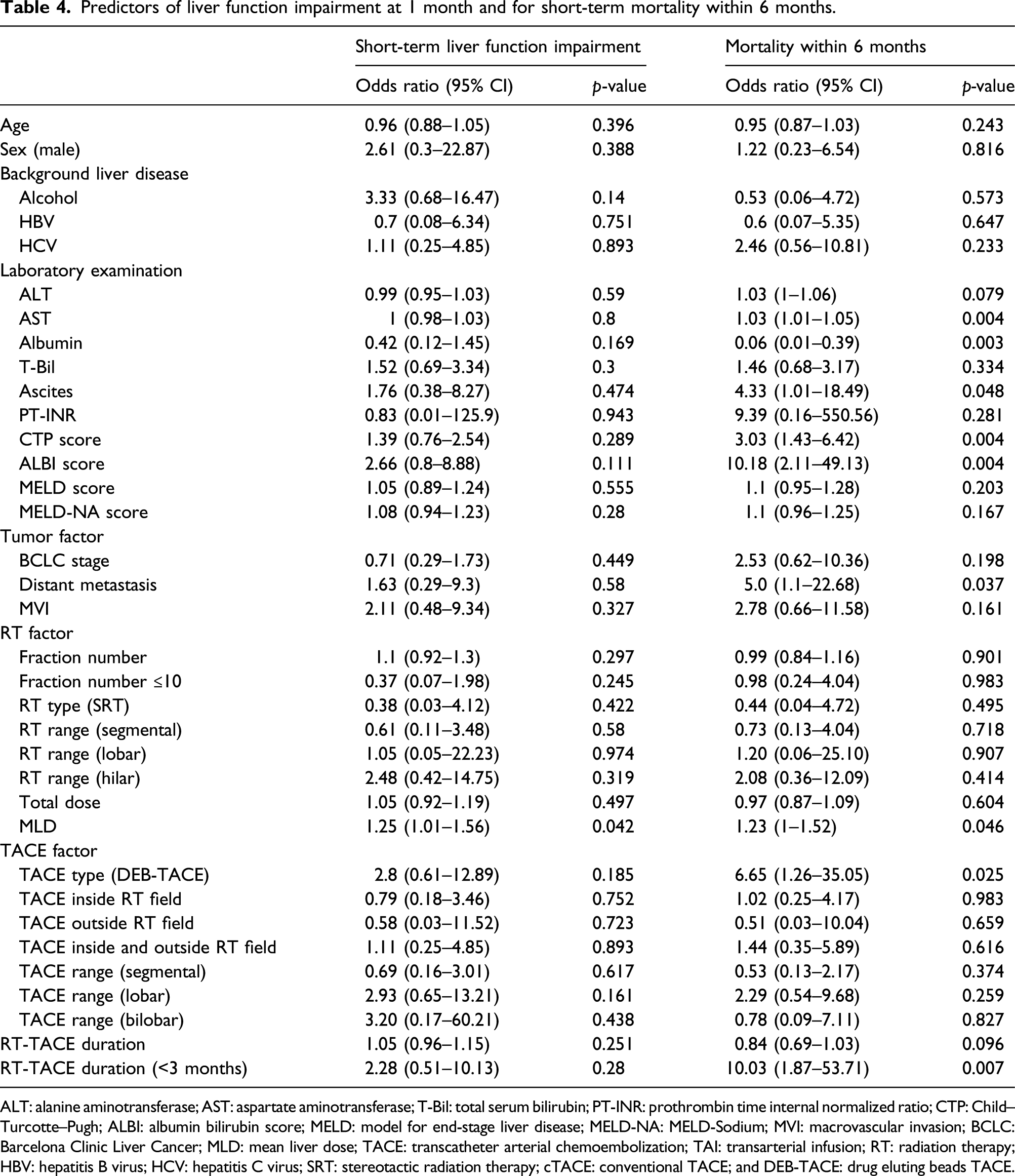
ALT: alanine aminotransferase; AST: aspartate aminotransferase; T-Bil: total serum bilirubin; PT-INR: prothrombin time internal normalized ratio; CTP: Child–Turcotte–Pugh; ALBI: albumin bilirubin score; MELD: model for end-stage liver disease; MELD-NA: MELD-Sodium; MVI: macrovascular invasion; BCLC: Barcelona Clinic Liver Cancer; MLD: mean liver dose; TACE: transcatheter arterial chemoembolization; TAI: transarterial infusion; RT: radiation therapy; HBV: hepatitis B virus; HCV: hepatitis C virus; SRT: stereotactic radiation therapy; cTACE: conventional TACE; and DEB-TACE: drug eluting beads TACE.
Discussion
TACE poses a substantial risk of liver function impairment and is not beneficial for patients with poor hepatic reserve.1,9,24 Previous studies reported a deterioration in the liver function in 11–24.5% of patients with an increase in CTP score by ≥2 points following TACE.12,13 Our results showed that eight patients (11.9%) experienced an increase in CTP score by ≥2 points, comparable to the rates reported in previous studies on TACE without RT. Hence, TACE following RT does not exacerbate the effect on liver function. Furthermore, it may be safe in terms of liver function impairment at 1 month.
The objective response rate of TACE in our study was over 74%, which is comparable to previous reports.1,2,10 Despite it, the median and mean survival after sequential TACE in the present study were 17.5 and 25.6 months, respectively. This short survival can be attributed to the poor clinical status of the patients as 68.7% of the patients had tumors of stage BCLC C. Despite their poor clinical conditions, the median survival duration was 17.5 months. This was relatively longer than the 14 months reported in a previous study, which included unresectable patients with HCC, predominantly with stage B BCLC, following TACE without RT. 25 We defined a period of 6 months as early mortality 16 and evaluated it to assess the feasibility of sequential therapy. Dhanasekaran et al. and Lencioni et al. reported a survival rate of 71–81% at 6 months following TACE. Additionally, Cammà et al. reported 0–10% mortality within the first month.23,26,27 The limited survival would make it difficult to assess the true effect of the sequential treatment on the tumor and liver function. Nonetheless, the early mortality within 6 months is comparable to that reported in previous studies, in which TACE was performed without RT. Thus, TACE in patients who underwent RT might be feasible with respect to both survival and short-term mortality and could be a treatment option for advanced HCC.
Biloma and liver abscess are well-known post-TACE complications, with reported incidences of up to 3.3% and 2.5%, respectively.26,28–31 Additionally, dilatation of the bile duct and formation of biloma often occur following RT.32,33 Therefore, the rate of biloma and liver abscess would likely increase after the sequential therapy, compared to either RT or TACE alone. However, we could not detect the above-mentioned complications in the cohort even when TACE was combined with RT. This promising result highlights the safety of performing TACE with regard to local complications in patients who received liver RT earlier.
RT for the liver is reportedly associated with the risk of RILD. Moreover, MLD substantially predicts its development.34,35 According to Velec et al., MLD is a significant risk factor for a CTP score with >2 points increase, 3 months following SRT for HCC with range 9.6–20.7 Gy. 36 The MLD of RT was found to be a significant predictor of short-term liver function impairment at 1 month following TACE. MLD was higher in eight patients with exacerbated liver function (12.6 ± 4.3 Gy) than in those who did not experience the outcome (9.7 ± 3.4 Gy), comparable to previous reports. 36 Similarly, patients who died within 6 months following RT had received higher MLD (12.4 ± 3.5 Gy), compared to those who did not (9.7 ± 3.5 Gy). Thus, it is important to assess the MLD before TACE; the risk for a potential liver deterioration should be carefully evaluated when the MLD is high.
RILD occurs as an acute response during or within a few weeks of RT, or as a late response, months to years after the completion of RT, 34 resulting in liver function impairment with >2 points increase in CTP score at 3 months following SRT for HCC. 36 In our study, the short duration between RT and TACE (3 months) was a significant risk factor of liver deterioration and short-term mortality in this study. It is presumed that TACE caused further liver damage when RILD was still ongoing. It is possible that there might be latent liver deterioration that is not reflected in the blood test, or that the response to TACE following RT may be stronger than TACE without preceding RT. Further research is needed in this regard. Thus, the short interval between RT and the following TACE may be considered a risk factor, highlighting the need to evaluate the interval between TACE and preceding RT.
Patients with distant metastases were included in this study. It is generally known that patients with distant metastasis have an extremely short life expectancy of 6–8 months. 1 The reason for including these patients in this study was that we evaluated their condition before TACE and judged that liver lesions rather than metastasis were a prognostic factor. Only one of the 12 patients with metastasis died within 6 months due to worsening of lung metastasis, while patients with distant metastases have risen to be a risk factor for early death within 6 months. The reason for this discrepancy is presumably that patients with distant metastases have more advanced intrahepatic disease.
Our study had several limitations. First, the study design was retrospective, with a relatively small sample size. Second, coexisting medical conditions, such as diabetes and cardiovascular diseases, were not evaluated. Third, the dose and type of systemic therapy were not sufficiently assessed. Since systemic therapy was administered to only one patient who died within 6 months after TACE, its impact on the results of this study seems modest. Fourth, our population is diverse in terms of TACE or RT techniques and the duration between them. Therefore, univariate analysis was performed using these variables, and the study results could give us overall understanding about the several situations of TACE when performed following RT.
In conclusion, performing TACE following liver RT poses no more risks than TACE alone, with comparable rates of short-term liver impairment and severe complications. This sequential treatment may be feasible. However, more attention should be paid to situations when there is an impairment of the pre-treatment liver function or after a high-dose RT. Moreover, it is advisable to delay TACE until RILD subside.
Footnotes
Author contributions
Conceptualization: T.O. and Y.K.; design of the work: E.U., T.I.,T.O., and K.S.; acquisition: M.H., E.U., T.I., H.H., J.I., H.M., K.S., and T.G.; analysis: M.H., E.U., T.I., M.Y., R.S., K.S., and T.M.; writing—original draft preparation: M.H., E.U., and T.I.; and writing—review and editing: Y.K., T.O., H.H., J.I., H.M., K.S., T.G., K.S., M.Y., R.S., K.S., and T.M. All authors have read and agreed to the submitted version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Institutional review board statement
This was approved by the institutional review board at our institution (No. B190130). The study protocol complied with the ethical guidelines of the Declaration of Helsinki and those of the Ministry of Health, Labor, and Welfare for medical and health research involving human subjects in Japan. All procedures were in accordance with the ethical standards of the institutional and/or national research committee for research on human participants and the tenets of the Declaration of Helsinki 1964 and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants.
