Abstract
Background
Subpleural pulmonary interstitial emphysema is defined as the air in the subpleural portion of the lung, and the clinical relevance is not well understood.
Purpose
to evaluate the frequency, temporal course, risk factors, and clinical significance of subpleural pulmonary interstitial emphysema (PIE) in patients with pneumomediastinum resulting from ruptured alveoli and other causes.
Material and Methods
This was a retrospective study of 130 patients with pneumomediastinum on CT between January 2009 and December 2019 at 2 hospitals. Patients were divided into 3 groups as follows: spontaneous pneumomediastinum (n = 101), pneumomediastinum due to blunt trauma (n = 16), and pneumomediastinum due to another known cause (n = 13). The frequencies of radiographic features (subpleural PIE, peribronchovascular PIE, pneumothorax, pulmonary fibrosis, and emphysematous changes) between the 3 groups were compared by the χ2 or Kruskal–Wallis test. Odds ratios were calculated to evaluate candidate risk factors for subpleural and peribronchovascular PIE.
Results
Subpleural PIE was observed in 0%, 15.8%, and 31.3% of patients with pneumomediastinum due to another cause, spontaneous mediastinum, and blunt trauma, respectively. In most patients, subpleural PIE resolved spontaneously (85.7% within 8 days). Two patients with pulmonary fibrosis showed recurrent subpleural PIE on follow-up. Young age showed increased risk for subpleural PIE (odds ratio [OR] 0.9, 95% confidence interval [CI] 0–0.99).
Conclusion
Subpleural PIE was only detected in patients with pneumomediastinum due to ruptured alveoli and resolved spontaneously and rapidly. Subpleural PIE may be one route the air from ruptured alveoli to the mediastinum.
Introduction
Pneumomediastinum is defined as the presence of air within the mediastinum. It is associated with various conditions, including blunt trauma, presence of gas-producing organisms, the Valsalva maneuver, and esophageal rupture. Depending on the cause, pneumomediastinum may require surgical intervention and can even be fatal. Identifying the cause of pneumomediastinum is thus important.
In 1939, Macklin et al. established the basic mechanism of the development of pulmonary interstitial emphysema (PIE) and pneumomediastinum by proving that air from ruptured alveoli spreads to the mediastinum along the bronchovascular bundles, which results in pneumomediastinum (Macklin effect). Rarely, air can also spread to the subpleural or septal regions, which results in pneumothorax. 1 Pulmonary interstitial emphysema can be identified on computed tomography (CT) scans, which show densities consistent with air along the bronchovascular bundles, visceral pleura, and/or interstitial septa.2–7 The CT findings of peribronchovascular PIE have been well described and are useful for differentiating pneumomediastinum due to ruptured alveoli from pneumomediastinum due to other causes.8–11 Subpleural PIE has been found on the CT scans of adult patients with spontaneous pneumomediastinum. 5 In addition, the finding of pathological PIE in patients with interstitial lung disease was identified as a risk factor for the development of air leaks.12,13 However, the incidence of subpleural PIE among patients with other causes of pneumomediastinum and the clinical importance of subpleural PIE in patients with pneumomediastinum have not yet been completely clarified.
The aim of this study was to evaluate the frequency, temporal course, risk factors, and clinical relevance of subpleural PIE in patients with pneumomediastinum resulting from ruptured alveoli and other causes.
Materials and methods
This retrospective observational study was approved by the institutional review boards of the participating institutions. Informed consent was waived because of the retrospective nature of the study.
Study population
The study participants were retrospectively selected from patients at 2 institutions between January 2009 and December 2019. The participants were initially selected from a picture archiving and communication systems database and then further refined by a review of their electronic medical records. The selection process used the keyword “pneumomediastinum” for all chest CT scans and their corresponding final reports provided within the study period. All of the patients enrolled in this study were again reviewed by 3 observers who reached consensus (A.N, N.T, and T.M.) on confirming the reports that were positive for pneumomediastinum. We excluded patients without thin-slice CT images (less than 1.25 mm). Patients who received positive-pressure ventilation within 4 weeks were also excluded because of possible alveolar damage due to positive-pressure ventilation. A total of 130 patients were ultimately enrolled in the study. All initial CT examinations were included in the analysis, and repeated CT examinations were only evaluated for follow-up assessments of subpleural PIE. Fig. 1 shows the flowchart for patient selection. Flowchart for patient selection.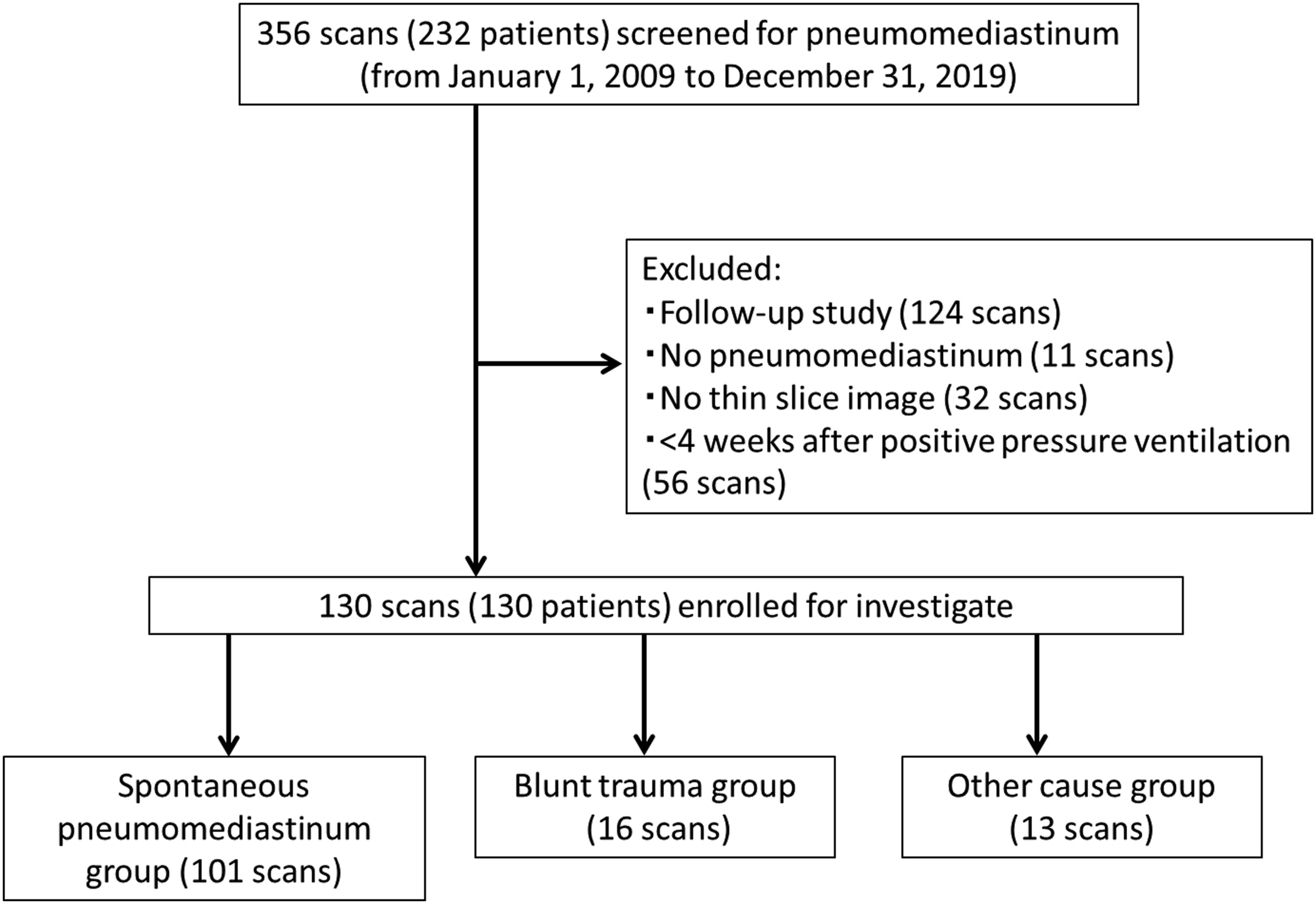
The patients were classified into 3 groups, based on the following conditions: (1) patients with spontaneous pneumomediastinum (pneumomediastinum resulting from ruptured alveoli not due to trauma); (2) patients with pneumomediastinum due to ruptured alveoli caused by blunt trauma; and (3) patients with pneumomediastinum due to a known cause other than damaged alveoli (e.g., air from the retroperitoneal space, peritoneal space, or the neck; perforated trachea, bronchus, or esophagus; and traumatic penetration of the thorax).
Imaging
Detailed imaging protocol for each computed tomography device.

Image analysis
Multi-detector computed tomography images were evaluated independently by 2 of 3 participating radiologists (A.N., and N.T. or T.M), and discrepancies were resolved by consensus that included the opinion of a third radiologist (S.M.). The following lung window settings were used to view the CT images: width, 1500 HU; level, −700 HU; and multiplanar reformatted images were also evaluated when needed.
The presence or absence of the following 5 image features was recorded: (1) subpleural PIE, (2) peribronchovascular PIE, (3) pneumothorax, (4) pulmonary fibrosis, and (5) emphysematous change. Subpleural PIE was defined as the presence of an air collection in the subpleural portion of the lung excluding peribronchovascular PIE on the CT image (Fig. 2). Reconstructed coronal (a) and axial (b) images of an 81-year-old man with spontaneous pneumomediastinum due to cough. Subpleural pulmonary interstitial emphysema on the mediastinal side is seen in the right upper lobe (a-arrows). A pulmonary vessel (b-arrowhead) is noted within the subpleural pulmonary interstitial emphysema, suggesting that the air pocket can differentiate subpleural PIE from pneumomediastinum.
Subpleural PIE was distinguished from pneumothorax by confirming that the visceral pleura was intact and that air was inside the pleura. Visualization of pulmonary vessels or internal septa also was used to confirm that air was inside the pleura. Pulmonary laceration was distinguished by its shape from subpleural PIE in patients injured by trauma. Because of normal pulmonary elastic recoil, the lung tissues surrounding a laceration pull back from the laceration, which leads to a laceration that manifests on CT as a round or oval cavity. 14 Intrathoracic extrapleural air collection was distinguished from subpleural PIE by the presence of web-like linear septa which does not continue to lung vessels. 15
Peribronchovascular PIE was defined as the presence of air near the bronchovascular sheath on the CT image (Fig. 3), which suggests the Macklin effect.8,9 A 66-year-old man with pulmonary fibrosis developed spontaneous pneumomediastinum. A reconstructed multi-detector computed tomography coronal image demonstrates air collection along the peribronchovascular connective tissue in the perihilar area, suggesting migration of air from the rupture site to the mediastinum.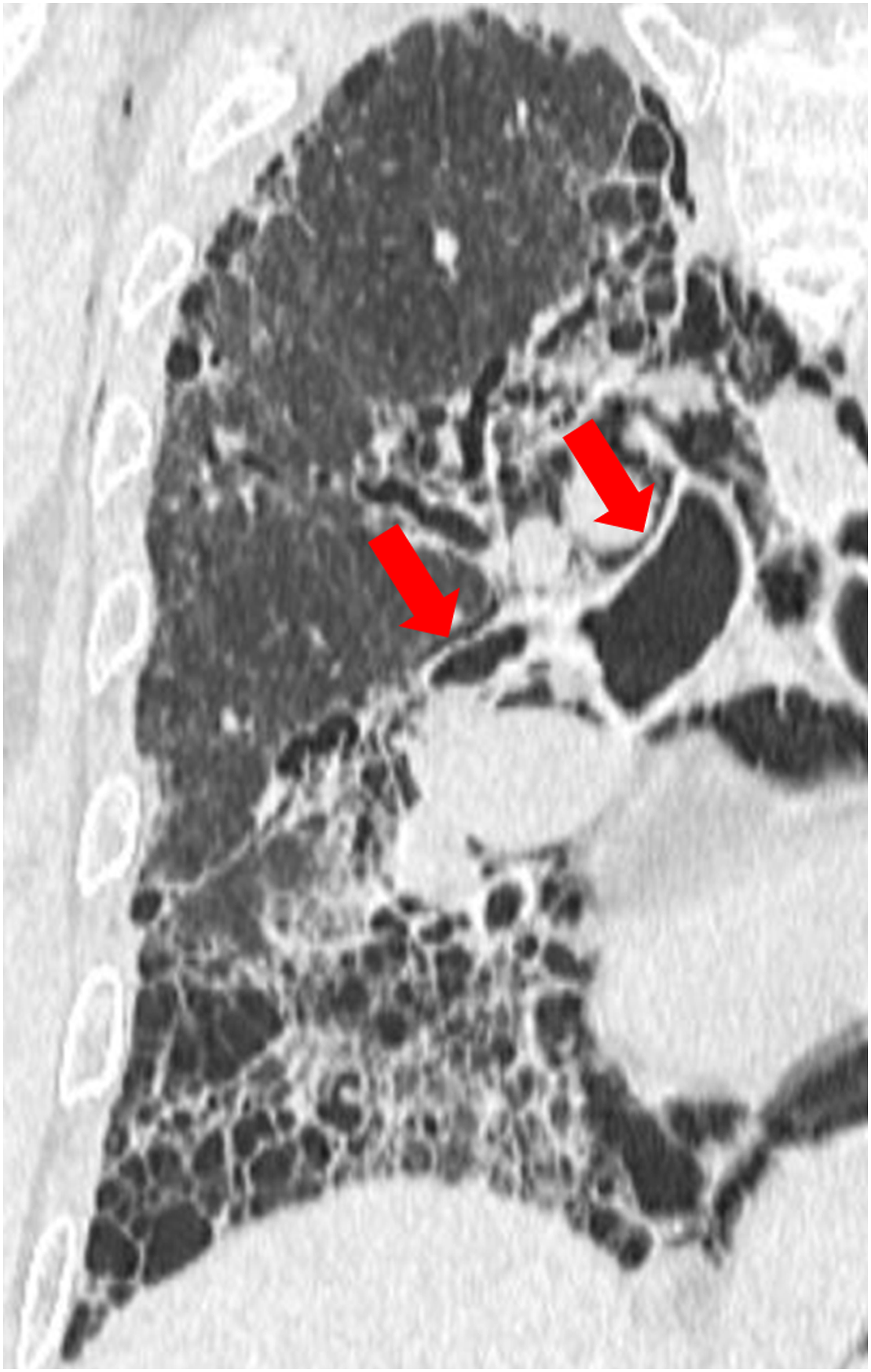
Area affected by honeycombing, traction bronchiectasis and bronchiolectasis, intralobular interstitial thickening, irregular interlobular septal thickening, irregular interfaces, and ground-glass opacity were judged positive for pulmonary fibrosis. 16 Definition of radiological terms was based on the standard glossary for thoracic imaging reported by the Fleischner Society. 17 For emphysematous change, areas or regions of low attenuation without visible walls were judged positive, and the lesion with scarring changes on the margin was excluded, to exclude scarring emphysema.
Statistical analysis
Categorical values from the imaging analysis and patient characteristics are presented as absolute numbers and percentages. Comparisons between 3 groups were performed by the Kruskal–Wallis test for continuous variables and the χ2 test for categorical variables. The Holm method was used for post hoc analysis. Logistic regression analysis with calculation of odds ratios was carried out to examine the association between subpleural PIE and patient background characteristics and imaging features. p values <0.05 were considered statistically significant, and adjusted p-values are shown presented for multiple comparisons. EZR software for Windows (Jichi Medical University Saitama Medical Center, Saitama, Japan) was used for statistical analysis.
Results
Patient characteristics and MDCT findings of the three groups of patients.

MDCT: multi-detector computed tomography, PIE: pulmonary interstitial emphysema.
ap-values were derived from the Kruskal–Wallis test or χ2 test.
bThe Holm method was used for comparisons between groups, and the reported p-values are the corrected values.
Asterisks denote statistically significant values.
More patients with spontaneous pneumomediastinum had peribronchovascular PIE (71/101, 70.3%) than patients with pneumomediastinum due to blunt trauma (6/16, 37.5%) or another cause (0/13) (p= 0.04, p < 0.01, respectively).
Subpleural PIE was observed in 16 of 101 (15.8%) patients with spontaneous pneumomediastinum, 5 of 16 (31.3%) with blunt trauma, and no patient with another cause. The differences between groups were not significant (p = 0.07). Subpleural PIE was detected in a total of 21 patients as follows: on the right (n = 11), on the left (n = 7), and bilaterally (n = 3). A single subpleural PIE lesion was seen in 16 patients and multiple lesions were seen in 5. A total of 29 lesions of subpleural PIE were found, as follows: 17 along the interlobar pleura (Fig. 4(c)), 11 on the mediastinal side (Fig 2(a), and (b)), and 1 on the peripheral side (Fig. 5(c)). Fourteen patients had ipsilateral peribronchovascular PIE, 2 did not have ipsilateral peribronchovascular PIE but had contralateral peribronchovascular PIE, and 5 did not have peribronchovascular PIE. Pneumothorax was noted in 4 patients, as follows: 2 with pneumothorax ipsilateral to subpleural PIE and 2 with contralateral pneumothorax. Peribronchovascular PIE was also found ipsilateral to the pneumothorax in the patients with contralateral pneumothorax in relation to subpleural PIE (Table 3). (a) A 15-year-old teenage boy with suspected acute fibrinous and organizing pneumonia shows subpleural PIE on the mediastinal side of the left upper lobe. (b) Subpleural PIE is not apparent in the right lung on the initial scan. (c) 15 days later, subpleural PIE appeared in the right middle lobe along the interlobar pleura. Abbreviation: PIE = pulmonary interstitial emphysema. (a) Axial image of a 72-year-old woman with subpleural PIE on the mediastinal side in the right upper lobe (arrows). Underlying lung shows pulmonary fibrosis. (b) and (c) One month after primary CT, the subpleural PIE in the right upper lobe was resolved, but new subpleural PIE was detected peripherally in the apex of the lung (arrows). Peribronchovascular PIE lesions were also found in the hilar region (a, b arrow heads). Abbreviation: PIE = pulmonary interstitial emphysema. Patient characteristics and MDCT findings of the patients with subpleural PIE. MDCT: multi-detector computed tomography PIE: pulmonary interstitial emphysema. +: present. -: absent.


The patients with spontaneous pneumomediastinum were more likely to have pulmonary fibrosis than the patients with pneumomediastinum due to blunt trauma or another cause (56.4% vs 6.3% vs 15.4%, p < 0.01). Pneumothorax was more common in the patients with blunt trauma than spontaneous pneumomediastinum (62.5% vs 23.8%, p = 0.01).
Follow-up CT was performed for 14 of 21 patients with subpleural PIE. Subpleural PIE had not resolved in 4 of the 14 patients (28.6%) within 3 days after their follow-up CT. Subpleural PIE resolved in 2 of the 4 patients within 8 days of follow-up, and the other 2 patients with unresolved subpleural PIE were lost to follow-up. One patient with spontaneous pneumomediastinum was found to have developed de novo subpleural PIE ipsilateral to the original lesion at the 1-month follow-up visit (Fig. 5, Table 3 [case 3]), which disappeared at the 2-month follow-up visit. Another patient with spontaneous pneumomediastinum developed a new contralateral subpleural PIE lesion at 15 days of follow-up (Fig. 4, Table 3 [case 5]), which disappeared at the 1-month follow-up visit.
Univariate logistic analysis of the relationship between PIE and patient background characteristics and imaging findings.

PIE: pulmonary interstitial emphysema OR: odds ratio, CI: confidence interval, SPM: spontaneous pneumomediastinum.
Asterisks denote statistical significance.
Discussion
In this study, we found that subpleural PIE can be seen in patients with spontaneous pneumomediastinum and patients with blunt trauma but not in patients with pneumomediastinum due to another cause. Subpleural PIE may suggest that air derived from the ruptured alveoli.
In patients with spontaneous pneumomediastinum, the frequency of subpleural PIE was similar to the frequency seen in a previous study by Kim (15.8% [16/101] vs 17.6% [6/34], respectively).
5
The frequency of subpleural PIE in patients with pneumomediastinum due to blunt trauma (31.3%, 5/16) was higher than the frequency in patients with spontaneous pneumomediastinum, but the difference was not statistically significant. This result might be accounted for by the fact that for the patients with pneumomediastinum due to blunt trauma, the time to diagnostic CT from the onset of symptoms was shorter than the time to diagnosis for the patients with spontaneous pneumomediastinum. Peripheral PIE reportedly disappears sooner than central PIE disappears.
9
Since subpleural PIE disappears sooner than peribronchovascular PIE, subpleural PIE might have already disappeared on a CT performed a long time after the onset of pneumomediastinum. This also explains the reason that subpleural PIE is less frequently encountered than peribronchovascular PIE. Another reason that subpleural PIE is less frequently observed is that air leaking from ruptured alveoli is more likely to spread along the peribronchovascular bundle to the mediastinum than to spread to the subpleural region, which Macklin
1
also described. Remarkably, however, 23.8% (5/21) of our patients did not manifest peribronchovascular PIE despite the presence of subpleural PIE. Based on this finding, we think that subpleural PIE from leaking ruptured alveoli can spread to the hilar side through the subpleural space to cause pneumomediastinum (Fig. 6). Potential explanation from subpleuralPIE to pneumomediastinum. ①Air from ruptured alveoli goes to the subpleural space result in subpleural PIE. ②Subpleural PIE flows through the subpleural space toward the hilar side. ③Subpleural air flows into the mediastinum through the hilum and cause pneumomediastinum. Abbreviation: PIE = pulmonary interstitial emphysema.
The presence of multiple peribronchovascular and/or subpleural PIE lesions in a single patient suggests that ruptured alveoli occurred in many different locations. Most subpleural PIE lesions disappeared relatively rapidly, which indicates a favorable outcome. Residual subpleural PIE was seen on follow-up CT of 4 patients. Two patients with pulmonary fibrosis manifested new subpleural PIE lesions on follow-up CT, with simultaneous exacerbation of mediastinal and subcutaneous emphysema. Pulmonary fibrosis is likely to develop in patients with repeated episodes of ruptured alveoli at multiple sites, a conclusion based on previous reports of the relationship between pulmonary fibrosis and ruptured alveoli.18,19
Subpleural PIE was more common in younger patients in this study. Greenough et al. concluded from their study of PIE in premature babies that the increased amount of pulmonary connective tissue in the immature lung is likely to trap air and develop PIE. 20 Therefore, we suspect that the same mechanism is involved in the findings that younger patients have more subpleural PIE than older patients. In addition, in daily practice, postinflammatory changes are often seen in the peripheral lung fields of elderly patients, and postinflammatory adhesions may impede the migration of air to the peripheral lung.
Previous studies have shown that PIE is more common in patients with interstitial pneumonia than in patients without interstitial pneumonia, and these patients with interstitial pneumonia are at risk for pneumothorax.1,12 In this study, pulmonary fibrosis was found to be a risk factor for peribronchovascular PIE but not for subpleural PIE. The result might be accounted for by the low rate of subpleural PIE detected in patients with pulmonary fibrosis due to fibrotic lesions located in the subpleural areas. Tachibana et al. found that pathological PIE in patients with interstitial lung disease was a risk of pneumothorax (OR 4.2, 95% CI 1.42–11.76). However, we did not find an association between pneumothorax and either subpleural or peribronchovascular PIE. 18 This inconsistent result might be accounted for by differences between the study populations. The presence of PIE with or without mediastinal emphysema in patients with secondary pneumothorax due to interstitial pneumonia warrants investigation. In addition, if we assume that subpleural PIE from perforated pleura is the cause of pneumothorax, it is possible it would be undetectable, similar to a perforated and collapsed bulla.
This study has limitations. The major limitation is its retrospective nature. The number of participants differed among the 3 study groups, and the total number of participants was not sufficient. The second limitation is the absence of histopathological findings that could verify the actual location of the subpleural PIE observed in the patients. An additional larger study with histopathological confirmation of our findings is needed.
In conclusion, subpleural PIE was detected in patients with spontaneous pneumomediastinum and patients with pneumomediastinum due to blunt trauma, which indicates that ruptured alveoli lead to both subpleural and peribronchovascular PIE. The association between subpleural PIE and underlying pulmonary disease or pneumothorax was not significant. In most patients, subpleural PIE resolves spontaneously. However, in patients with refractory pneumomediastinum associated with chronic lung disease, recurrent subpleural PIE, which reflects repeated episodes of ruptured alveoli, might occur.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
