Abstract
Background
Neoadjuvant endocrine therapy is an alternative to neoadjuvant chemotherapy in women with inoperable luminal-like breast cancers. Neoadjuvant cyclin-dependent kinase 4/6 inhibitor treatment combined with endocrine treatment (CDK4/6I + E) is interesting given the combination’s utility in the treatment of metastatic breast cancer. Currently, the literature on the radiological response evaluation of patients treated with neoadjuvant CDK4/6I + E in a real-life setting is scarce.
Purpose
To conduct a radiological response evaluation of patients treated with neoadjuvant CDK4/6I + E in a real-life setting.
Material and methods
We retrospectively reviewed clinical, pathological, and radiological findings of six patients with luminal-like breast cancers treated with neoadjuvant CDK4/6I + E treatment. The radiological neoadjuvant CDK4/6I + E response was evaluated with the RECIST 1.1 criteria and the pathological residual disease was assessed using the Residual Cancer Burden (RBC) criteria.
Results
None of the patients achieved a complete radiological magnetic resonance imaging (MRI)–determined response or a complete pathological response; three (50%) patients had a partial radiological response; in the three others, the disease remained stable radiologically. All of the tumors were rendered susceptible to surgical treatment. Two out of six (33.3%) patients had a moderate response (RBC-II); four (66.7%) had an extensive residual disease (RBC-III) in the final surgical sample.
Conclusion
Although none of the patients achieved a pathologically complete response, neoadjuvant CDK4/6I + E treatment rendered all tumors operable. MRI appears to be reliable in the assessment of the neoadjuvant CDK4/6I + E treatment response in a real-life setting. Larger studies are warranted to confirm these results.
Introduction
Major clinical guidelines advocate the use of neoadjuvant chemotherapy (NAC) treatments to downstage inoperable locally advanced breast cancers so that they are operable or to downsize operable tumors to enable less extensive surgical treatment.1,2 Although NAC treatments are utilized to varying degrees, the number of patients now receiving this kind of therapy has increased.3,4 Chemotherapy (ChT) is the preferred choice as neoadjuvant therapy. In addition, anti-HER2 treatments (trastuzumab ± pertuzumab) have been combined with ChT in HER2-positive cancers. 1 It has been reported that the absolute benefit of ChT is more pronounced in estrogen receptor (ER)-negative tumors. 5 Luminal A- and B-like breast cancers are hormone receptor positive1,2 and respond poorly to chemotherapy. 5 While endocrine therapy is an alternative to ChT in women with inoperable luminal A-like breast cancers, ChT is preferred for women with luminal B-like breast cancers.1,2
Cyclin-dependent kinases (CDKs) are regulatory enzymes involved in cell division. 6 A dysregulation of the CDK4/6 pathway occurs particularly in ER-positive breast cancer, resulting in an increased rate of cellular proliferation. 7 CDK4/6 inhibition causes cell cycle arrest and prevents disease progression through the blockade of the cell cycle; this mechanism of action differs from that of ChT.6,7 To date, three different CDK4/6 inhibitors (CDK4/6Is), palbociclib, ribociclib, and abemaciclib, have been approved by the United States Food and Drug Administration and the European Medicines Agency for treatment of locally advanced or metastatic breast cancer. These medications are intended to be combined with endocrine treatment (CDK4/6I + E), either with aromatase inhibitors or fulvestrant. CDK4/6 inhibitors have revolutionized the treatment of luminal advanced breast cancer and are now considered to be the standard of care in this population.1,2 Marketing approvals have been based on large phase three trials that demonstrated a clear benefit in progression-free survival with all three CDK4/6Is and in overall survival with ribociclib and abemaciclib.8–10 Encouraged by improved results in metastatic breast cancer, some studies have been conducted to evaluate the efficacy of CDK4/6I + E in a neoadjuvant treatment setting. However, while a clinical response was seen mainly at the cellular level, clinical response rates have been varying, and the use CDK4/6I + E in a neoadjuvant setting warrants further research.11–14
Magnetic resonance imaging (MRI) of the breast subsequent to NAC has been shown to be more accurate than ultrasonography or mammography in detecting a pathological complete response and also for evaluating the residual disease size.15–17 Both the American and European guidelines recommend MRI in the evaluation of NAC treatment response and possible residual disease.18–20 The Response Evaluation Criteria in Solid Tumors (RECIST) criteria are arguably the most widely utilized criteria in women with advanced breast cancer. 21 We retrospectively reviewed clinical, pathological, and radiological findings of six patients with luminal-like breast cancers treated with neoadjuvant CDK4/6I + E treatment in our institution.
Material and methods
Study sample
This retrospective study was conducted in the Tampere University Hospital Cancer Center. All patients with breast cancer in the Tampere University Hospital district are assessed in a multidisciplinary team (MDT) meeting attended by breast surgeons, pathologists, oncologists, and radiologists to schedule individualized treatment plans. The local indications for neoadjuvant treatment, including inoperable breast cancers (clinical tumor (T) stage 4 or nodal (N) stage 3), inflammatory breast cancer, and HER2-positive and triple negative disease with nodal involvement, adhere to the major treatment guidelines.1,2 The MDT meeting considers the neoadjuvant CDK4/6I + E treatment as an alternative to NAC in hormone receptor positive, HER2-negative, primarily inoperable luminal-like breast cancers which have no distant metastasis. As a part of disease staging, patients with locally advanced breast cancer in our institution are imaged with bone scintigraphy and full-body computer tomography to rule out the presence of distant metastasis. Patients with de novo distant metastasis were excluded as the intention of their treatment is life-prolongation.
We analyzed the patients (N = 6) who had received neoadjuvant CDK4/6I + E between the years 2018 and 2020. In our institution, the treatment-of-choice in the neoadjuvant setting has been palbociclib combined with either letrozole or fulvestrant. The response to neoadjuvant CDK4/6I + E therapy is determined with regular breast MRI follow-up every two to three months (or three cycles). The decision to proceed to surgery is made jointly in the MDT meeting by evaluating the operability and the achieved response based on MRI. If the tumor becomes operable and the discontinuation of neoadjuvant CDK4/6I + E therapy is indicated (e.g., adverse effects), the patient may undergo surgery even before the estimated best response has been achieved. The patient records were reviewed to collect clinical, pathological, and radiological data.
The study was approved by the local chair of the Tampere University Hospital district (study number: R19627S). The need for informed consent from the patients for the use of register data including the patient records and radiologic images was waived in compliance with local and national regulations and laws.
Magnetic resonance imaging
The image acquisition parameters of the two scanners used during the study period. The sequences are listed in their acquisition order.
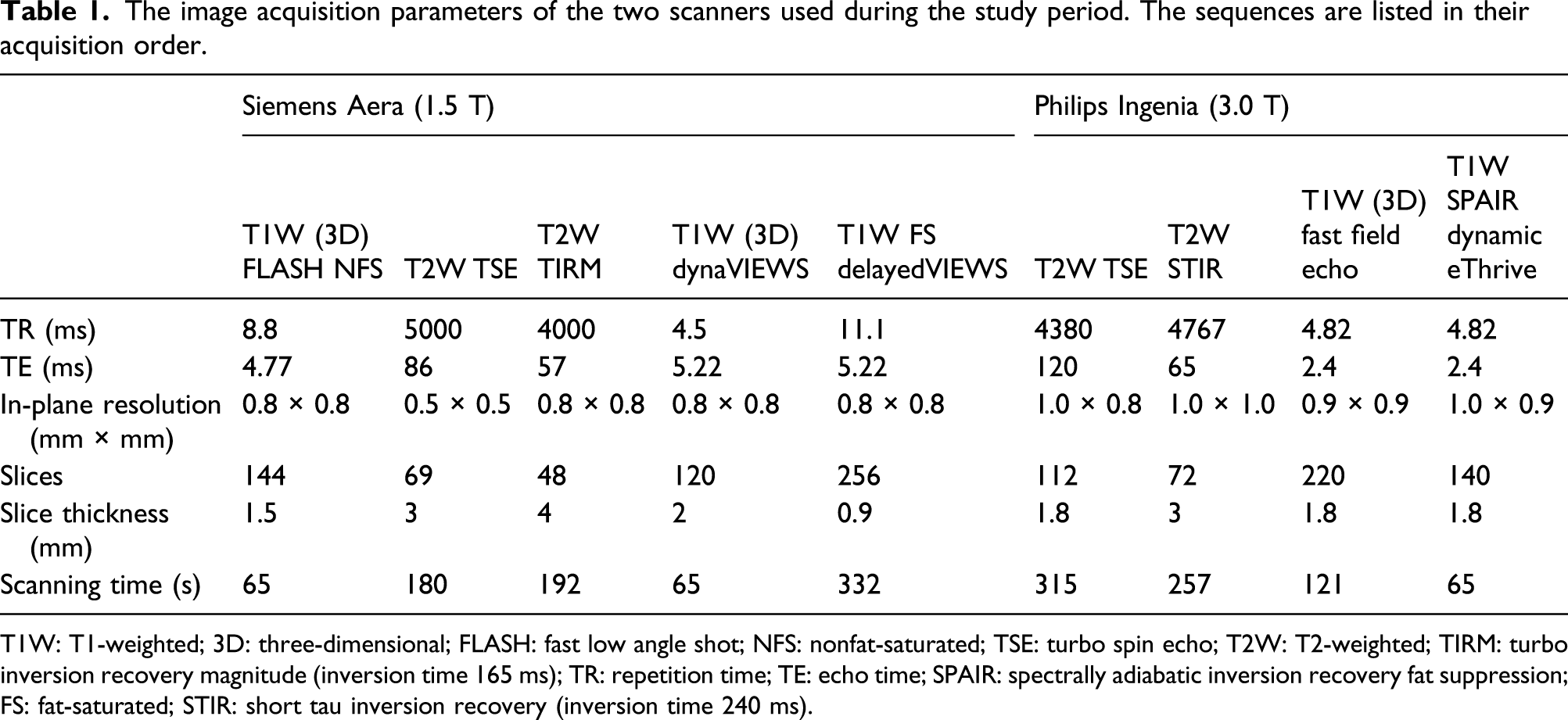
T1W: T1-weighted; 3D: three-dimensional; FLASH: fast low angle shot; NFS: nonfat-saturated; TSE: turbo spin echo; T2W: T2-weighted; TIRM: turbo inversion recovery magnitude (inversion time 165 ms); TR: repetition time; TE: echo time; SPAIR: spectrally adiabatic inversion recovery fat suppression; FS: fat-saturated; STIR: short tau inversion recovery (inversion time 240 ms).
Radiological tumor response evaluation
One reader (A.S.) with 8 years of experience in breast imaging, blinded to the histopathological reference standard, evaluated the radiological tumor response on DCE-MRI sequence in the post-contrast image that allowed the best visualization of the tumor. Tumor burden according to the RECIST version 1.1 classification was recorded first prior to CDK4/6I + E (baseline) and at the follow-up time points with follow-up examinations being compared to the baseline (Fig. 1). The response to the treatment was categorized as complete response (CR), partial response (PR), stable disease (SD), or progressive disease (PD) according to the measured changes in the tumor burden.
21
The absence of post-contrast enhancement was defined as a complete radiological response. Images (a) and (b) illustrate the case of a women in her mid-60s. The sum of the tumor diameters prior to treatment was 57 mm. A partial response according to the RECIST 1.1. criteria was detected after seven cycles of palbociclib and letrozole; the sum of diameters was 13 mm. At the final pathological evaluation, the tumor’s longest diameter was 150 mm and dermal invasion was detected (patient number 2 in Fig. 2). Images (c) and (d) illustrate the case of a women in her late 60s. The sum of the tumor diameters prior to treatment was 84 mm. The disease was stable according to the RECIST 1.1 criteria after four cycles of palbociclib and letrozole; the sum of diameters was 71 mm. At the final pathological evaluation, the tumor’s longest diameter was 47 mm.
Diagnostic pathological evaluation and pathological response evaluation
Diagnostic histopathological samples of tumors were obtained with core needle biopsy (CNB) under ultrasound guidance. Ultrasound-guided axillary lymph node needle biopsy samples were harvested to confirm the presence of axillary metastasis. All tissue specimens were placed into neutral buffered 10% formalin. After fixation, the samples were processed routinely, embedded in paraffin, and cut into 3–4 μm slices at four different levels and stained with hematoxylin and eosin (H&E). Diagnosis and treatment response to neoadjuvant CDK4/6I + E treatment were confirmed from the final surgical specimens. The CNB and surgical samples were evaluated twice by pathologists, first at the time of diagnosis and then at the multidisciplinary meeting.
The primary tumor bed area and the cellularity of the residual tumor were assessed with the aid of the MD Anderson Cancer Center RCB pathology protocol (at www.mdanderson.org/education-and-research/resources-for-professionals/clinical-tools-and-resources/clinical-calculators/calculators-rcb-pathology-protocol2.pdf?, accessed 28 January 2021). A Residual Cancer Burden class 0 equates to a pathologic complete response (pCR). The post-neoadjuvant pathological tumor stage (ypT) and lymph node stage (ypN) were assessed according to UICC TNM Classification of Malignant Tumours, 8th Edition. 22 Breast cancer subtypes and grades were determined according to the WHO 2019 guidelines. 23 Intrinsic breast cancer subtypes were determined according to the ESMO guidelines. 1 Residual Cancer Burden (RCB) was determined using the MD Anderson Cancer Center online calculator (http://www3.mdanderson.org/app/medcalc/index.cfm?pagename=jsconvert3). Prognostic immunohistochemical (IHC) staining panels were performed from both pretreatment needle biopsies and from fresh sampled tissue blocks from post-neoadjuvant surgical specimens.
Immunohistochemistry
Estrogen receptor (ER) ER15 (clone SP1), progesterone receptor (PgR) PR17 (clone 1E2), Ki-67 (clone 30-9), and HER2 (clone 4B5) immunostainings, and Ventana HER2 dual double-chromogen in situ hybridization were performed using a Ventana Benchmark Ultra immunostaining automated setup with ready-to-use primary antibodies from Roche Ventana. Detections for all mentioned primary antibodies were performed using the Ultraview DAB Detection kit (Roche Ventana). Immunostained slides were digitized using Hamamatsu Nanozoomer XR slide scanner at 40x magnification (0.23 µm/pixel). Immunohistochemical stainings were estimated visually from the scanned slides for ER, PgR, and HER2. Carcinoma cells were considered negative for ER and PgR if the immunohistochemically determined expression of any intensity was <1% and positive if expression was ≥1%. The Ki-67 proliferation index was analyzed using ImmunoRatio (version 2.5) image analysis software on three hotspots comprising at least 1000 cells. 24 Dual HER2 in situ hybridization signals were counted manually in either light microscope (until September 2019) or from 5-layer z-stack images starting in September 2019 (no offset, ± 3.0 µm). HER2 status was considered positive according to the ASCO CAP 2018 Guidelines. 25 HER2 was determined to be negative at an external institution prior to treatment in the case of one patient; the information regarding the methodology used was not available.
Statistical analyses
Nominal values are presented as absolute values and as percentual shares. Continuous values are expressed as means ± standard deviations unless otherwise stated. SPSS 27.0 for Windows (version 27.0, 1989-2020, SPSS Inc., Armonk, NY, USA) was used for statistical analyses.
Results
We enrolled six postmenopausal women. The mean age of the patients at the time of diagnosis was 70 ± 5.3 years (range 65–79 years). All the patients were treated with palbociclib combined with endocrine treatment. The starting daily dosage of palbociclib was 125 mg, with the exception of one patient who received letrozole for ∼300 days before her treatment and, due to her underlying medical condition, she was prescribed palbociclib at a reduced daily dose of 75 mg. In two patients, dose reductions, or a delay in starting palpociclib treatment cycles, were carried out as clinically indicated and recommended by the manufacturer. Letrozole was the first line endocrine treatment for all patients. However, one patient, after being on palbociclib + letrozole for ∼100 days, switched to palbociclib + fulvestrant. The median number of CDK4/6I + E treatment cycles before surgery was 8 (range 4–10). The mean duration of the period between the initiation of the endocrine treatment and surgical procedure was 298 ± 137 days (range 203–566 days).
Prior to the neoadjuvant treatments, the patients had radiologically and clinically large tumors (T2 in two (33.3%) patients, T3 in one (16.7%) patient, and T4 in three (50%) patients). Nodal metastasis was suspected or histopathologically confirmed in five (83.3%) patients before the CDK4/6I + E treatment; one patient with a T4 breast cancer had no confirmed nodal metastasis. The sum of diameters of the target lesions was 59 ± 14 mm (range 45–84 mm) at the baseline and 37 ± 22 mm (range 13–70 mm) at the last follow-up MRI prior to surgery. None of the patients achieved a complete radiological response; 3 (50%) had a PR and 3 (50%) patients were in a SD state. However, all of the breast cancers were rendered operable. Three patients underwent mastectomy and axillary lymph node dissection (ALND). One patient was subjected to ablation and sentinel lymph node biopsy. Two patients underwent breast conservation therapy and ALND.
Patient characteristics. All the patients had HER2-negative diseases.
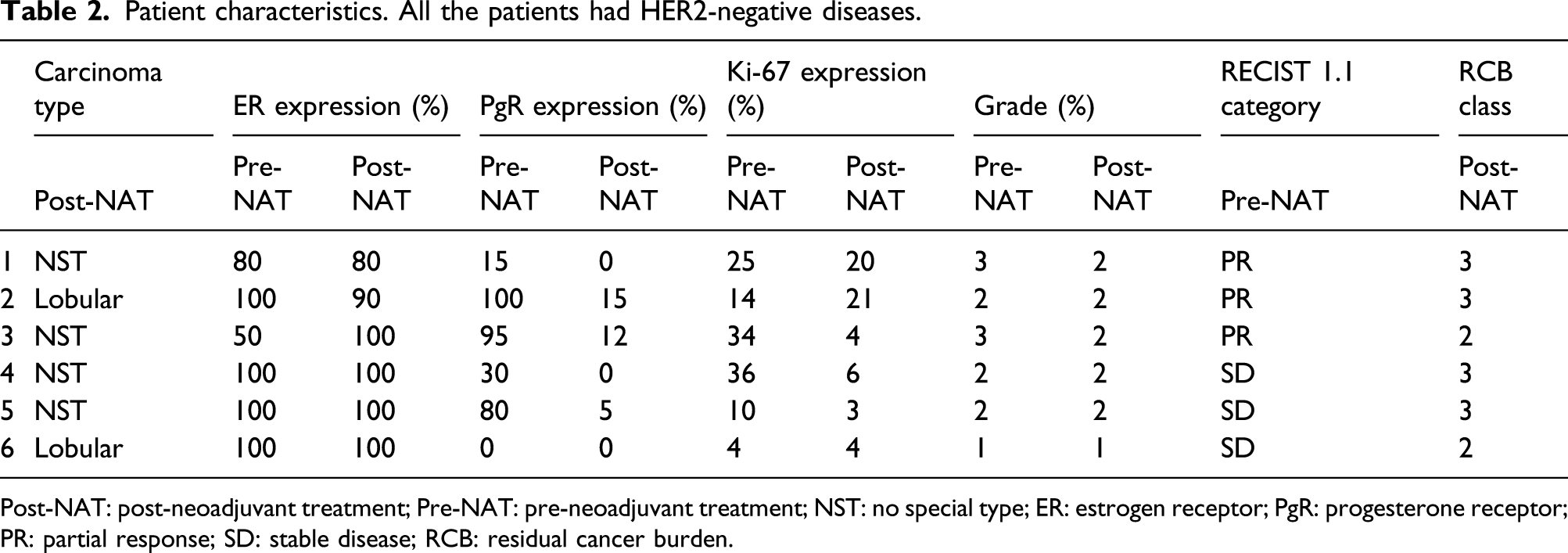
Post-NAT: post-neoadjuvant treatment; Pre-NAT: pre-neoadjuvant treatment; NST: no special type; ER: estrogen receptor; PgR: progesterone receptor; PR: partial response; SD: stable disease; RCB: residual cancer burden.
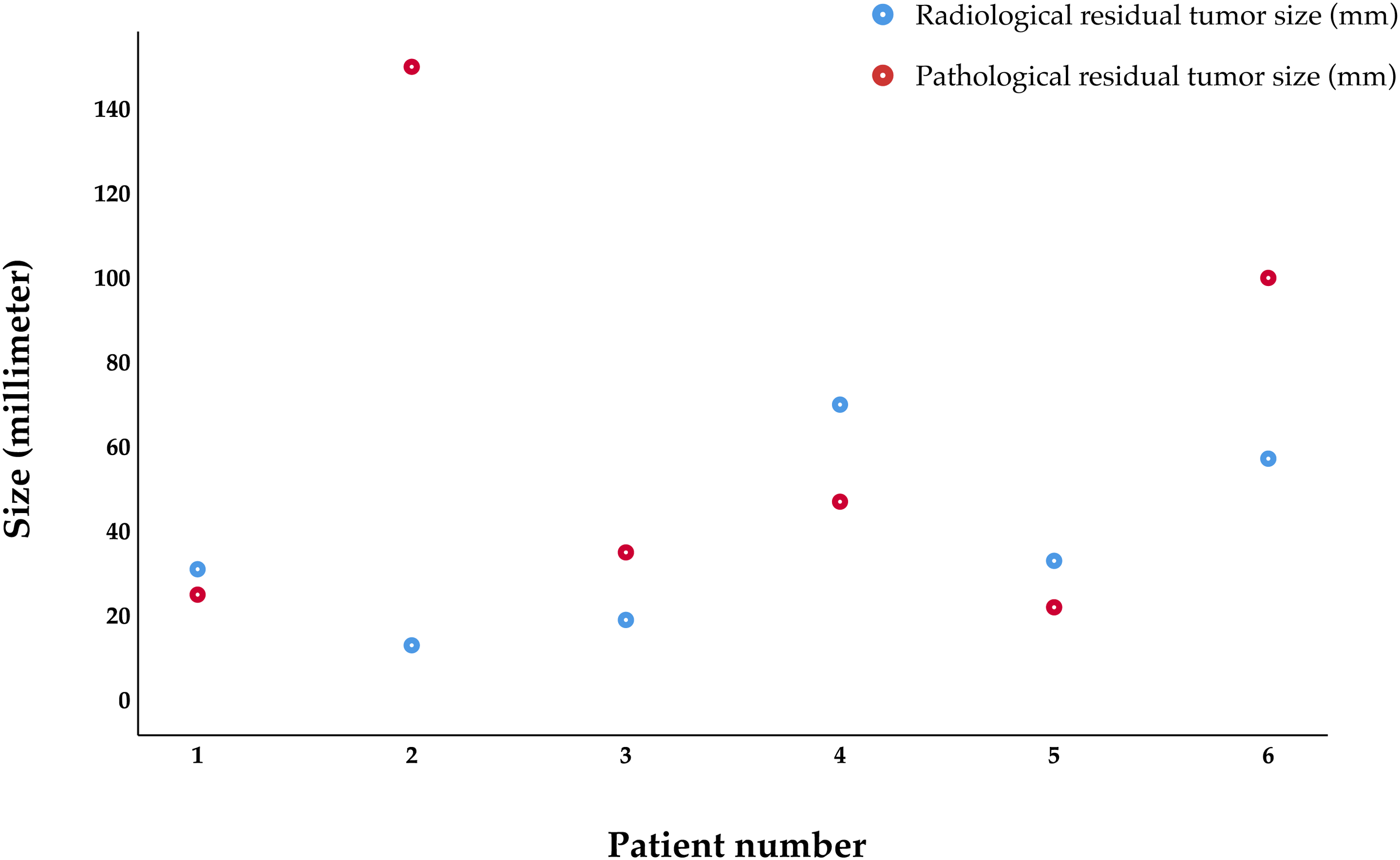
Dot histogram illustrating the discrepancy between the final sums of diameters on magnetic resonance imaging prior to surgery and the longest diameter of the largest residual tumor.
Discussion
Our limited data imply that the MRI-based response evaluation of neoadjuvant CDK4/6I + E treatment seems to parallel the pathological outcome. Our real-life sample suggests that a size reduction evident on DCE-MRI is indicative of a tumor response. This proposal is important not only when monitoring the disease burden but also when planning the surgical procedure based on the extent of dynamic contrast enhancement seen on MRI.
The indications for neoadjuvant treatments in our institution adhere to the current treatment guidelines.1,2 The decision to proceed to surgery with the patients followed in this study was made in the MDT meeting according to an evaluation of the MRI-based response. While the response was assessed as significant in four cases, the other two women had less marked responses. Five patients were considered to have achieved the maximum response from the neoadjuvant treatment prior to surgery. One patient receiving CDK4/6I and letrozole experienced signs of liver toxicity associated with letrozole; CDK4/6I + E was therefore discontinued, and the patient was operated. At the time of the surgery, all tumors were regarded as operable.
The use of the RECIST 1.1 criteria enables an objective and reproducible assessment of the response to treatments. 21 In a recent meta-analysis by Cheng et al., 26 it was stated that the RECIST 1.1 criteria perform well in the NAC setting, predicting a pCR with pooled sensitivity and specificity values of 70% and 83%, respectively. None of the patients in our study sample achieved a radiological complete response. There are no reports on MRI’s applicability in tumor response evaluation in a real-life setting where the neoadjuvant CDK4/6I + E treatments are tailored to patients individually based on their responses to treatments, treatment tolerance, and preferences. Only the CORALLEEN trial, comparing ribociclib and letrozole (N = 52) against the standard NAC (N = 54) in patients with luminal B-like breast cancers, has reported the RECIST-based radiological responses in patients undergoing follow-up MRI after 24 weeks of treatment 27 which corresponds to six 4-week cycles. Our patients, based on their individual treatment plans, underwent four to 10 neoadjuvant CDK4/6I + E cycles. In their sample, the MRI-based CR rate in the CDK4/6I + E arm was 14.3%. 27 It is noteworthy that more patients achieved a radiological CR in the CORALLEEN trial than in the other neoadjuvant CDK4/6I trials; other groups have reported that the proportion of patients achieving a radiological CR (confirmed either with mammography, ultrasound, or magnetic resonance imaging) has ranged between 1.6 and 7%.11–13,27
Achieving a pCR is associated with a favorable prognosis in the NAC setting.24,25 None of the patients in our study sample achieved a pCR. This is in line with previous studies on neoadjuvant CDK4/6I + E; these have reported that the pCR rates vary between 0.0 and 4.4%.11–14,27 To the best of our knowledge, in addition to the present study, only the CORALLEEN trial reported RCB classes for patients treated with CDK4/6I + E in the neoadjuvant setting. Their results revealed a similar trend to our findings: the majority of the patients (93.9%) had RCB classes 2–3. 27 None of our patients achieved a complete pathological response. The CORALLEEN trial reported that only 6.1% of the patients had RCB classes 0–1 after the neoadjuvant CDK4/6I + E treatment. 27
Our review of these six real-life patients treated in our institution supports the findings reported in the CORALLEEN trial. Furthermore, they suggest that the RECIST 1.1 response evaluation criteria indeed may characterize neoadjuvant CDK4/6I + E treatment response adequately in real-life settings even when the number of CDK4/6I + E cycles varies. However, although the MRI-based response evaluation using the RECIST 1.1 criteria seems appropriate in monitoring the treatment response, these results must be interpreted with caution. Ko et al. 28 suggested that the capability of MRI to predict the residual tumor size after NAC was less accurate when evaluating ER-positive breast cancers versus triple negative or HER2-positive breast cancers. Fukuda et al. 29 also noted that the accuracy of an MRI evaluation with the RECIST criteria was less reliable in luminal subtypes and that the absence of enhancement on MRI did not necessarily indicate a pCR after NAC. However, the mechanism of action of CDK4/6I + E differs remarkably from chemotherapy; while CDK4/6I + E arrests cell proliferation, chemotherapy evokes the death of proliferating cells. We hypothesize that the reduced cell proliferation may eventually translate to changes in the cell proliferation–induced neovasculature, which in turn may be falsely interpreted as a treatment response.
A major limitation of our study is our small patient sample. Indeed, the utilization of CDK4/6I + E treatments in neoadjuvant setting has gained acceptance only recently which translates to the small patient sample. Furthermore, there were no patients with PD or a pCR. This also is expected because the somewhat scant literature on neoadjuvant CDK4/6I + E indicates that breast cancers seldom progress or respond completely if they are subjected to neoadjuvant CDK4/6I + E treatments.11–14,27 We believe that despite these limitations, the likely increasing utilization of neoadjuvant CDK4/6I + E treatments justifies this report as the notion that the RECIST 1.1 response evaluation criteria indeed may characterize neoadjuvant CDK4/6I + E treatment response adequately in real-life settings even when the number of CDK4/6I + E cycles varies is clinically relevant. Another drawback is that only the DCE sequence was available for the response evaluation. Multiparametric MRI could potentially be of more assistance in evaluating tumor response. Other sequences could also aid in recognizing the small proportion of patients who might achieve a pCR. At the moment, however, there is no evidence that a pCR to CDK4/6I + E treatments would translate into improved survival. Therefore, it is important to evaluate whether a pCR has true prognostic significance in these patients before conducting more elaborate imaging studies.
In conclusion, MRI-based response evaluation using the RECIST 1.1 criteria seems appropriate in monitoring the treatment response of CDK4/6I + E in real-life setting, these results must be interpreted with caution given the small sample size of this study. More systematic research will be needed to confirm these findings. We urge both radiologists and clinicians to carefully interpret the response seen in MRI.
Footnotes
Author contributions
M.M., A.S., T.T., and O.A. substantially contributed to the work by conceptualizing the work, analyzing, and interpreting the data, drafted the article, approved the final version, and agreed to be accountable for the content of this manuscript.
A.J., A.S., N.M, L.T., and I.R-K. substantially contributed to the work by designing the work and interpreting the data, drafted the article, approved the final version, and agreed to be accountable for the content of this manuscript.
M.T. substantially contributed to the work by collecting the data, drafted the article, approved the final version, and agreed to be accountable for the content of this manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Doctors Salminen, Sassi, Tervo, Rinta-Kiikka, Tolonen, and Arponen report no conflicts of interest. Doctor Moisander reports consultant and travel fees from Takeda and Roche. Doctor Mäenpää reports consultant fees from Lilly and travel fees from Pfizer. Doctor Tiainen reports consultant and travel fees from Novartis, Roche, and Pfizer.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Research ethics and patient consent
The study was approved by the local chair of the Tampere University Hospital district (study number: R19627S). The need for informed consent from the patients for the use of register data including the patient records and radiologic images was waived in compliance with local and national regulations and laws.
