Abstract
Background
Due to their easy accessibility, CT scans have been increasingly used for investigation of gastrointestinal (GI) bleeding.
Purpose
To estimate the performance of a dual-phase, dual-energy (DE) GI bleed CT protocol in patients with overt GI bleeding in clinical practice and examine the added value of portal phase and DE images.
Materials and Methods
Consecutive patients with GI bleeding underwent a two-phase DE GI bleed CT protocol. Two gastroenterologists established the reference standard. Performance was estimated using clinical CT reports. Three GI radiologists rated confidence in GI bleeding in a subset of 62 examinations, evaluating first mixed kV arterial images, then after examining additional portal venous phase images, and finally after additional DE images (virtual non-contrast and virtual monoenergetic 50 keV images).
Results
52 of 176 patients (29.5%) had GI bleeding by the reference standard. The overall sensitivity, specificity, and positive and negative predictive values of the CT GI bleed protocol for detecting GI bleeding were 65.4%, 89.5%, 72.3%, and 86.0%, respectively. In patients with GI bleeding, diagnostic confidence of readers increased after adding portal phase images to arterial phase images (p = 0.002), without additional benefit from dual energy images. In patients without GI bleeding, confidence in luminal extravasation appropriately decreased after adding portal phase, and subsequently DE images (p = 0.006, p = 0.018).
Conclusion
A two-phase DE GI bleed CT protocol had high specificity and negative predictive value in clinical practice. Portal venous phase images improved diagnostic confidence in comparison to arterial phase images alone. Dual-energy images further improved radiologist confidence in the absence of bleeding.
Keywords
Introduction
Overt gastrointestinal (GI) bleeding is an emergent medical condition with a mortality rate of about 3% in the United States. 1 Upper GI endoscopy and colonoscopy are the main diagnostic modalities in the diagnosis and management of overt GI bleeding but do have limitations in the acute setting (e.g., limited reach into the small bowel, obscuration of the bleeding site by a clot, significant patient comorbidity such as shock or massive hemorrhage). 2 Additionally, in the emergent setting, the diagnostic utility of colonoscopy is limited without adequate bowel preparation.2,3
Because neither GI preparation nor oral contrast is required, CT can be acquired without delay upon patient presentation. Both arterial and venous sources of bleeding can potentially be detected with the use of a multiphasic protocol. 2 Historically, there has been wide variability in the CT protocols used to evaluate overt GI bleeding. Most sites perform unenhanced images to detect hyperdense ingested material (which can mimic bleeding) or hemorrhage, followed by late arterial and portal venous or delayed phase images. The portal venous or delayed phase images may increase radiologist confidence in the presence of luminal extravasation of contrast material, one of the main findings indicative of GI bleeding on CT, or it may improve detection of the source of bleeding. Recently, the Society of Abdominal Radiology GI Bleeding Disease-focused Panel has developed consensus recommendations for CT technique. 4 The utilization of dual-energy (DE) techniques in the CT protocol offers potential theoretical advantages by eliminating non-contrast image acquisition by using virtual non-contrast (VNC) images, and by increasing the conspicuity of extravasating iodine contrast by using dual energy virtual monoenergetic images. 2 The Society of Computed Body Tomography and Magnetic Resonance white paper on abdominal dual energy applications suggests that dual energy VNC images may have a role in improving specificity of CT for GI bleeding by avoiding overdiagnosis due to radiopaque intraluminal debris. 5
Some studies have investigated the role of DE CT protocol in identifying GI bleeding. However, the utility of VNC is not well established as these studies used a true non-enhanced phase instead of VNC images reconstructed from DE CT protocol.6,7 The current study aimed to estimate the performance of a dual-phase, DE GI bleed CT protocol in patients with overt GI bleeding in clinical practice and to estimate the added value of portal venous phase and DE images (virtual monochromatic 50 keV and VNC images, and iodine maps). We hypothesize that the addition of portal venous phase and DE images to the review of arterial phase images for evaluating possible GI bleeding could improve readers’ confidence in the presence or absence of GI bleeding.
Materials and methods
Study population and design
After approval by our institutional review board, we reviewed the clinical records of patients presenting with upper or lower GI bleeding between December 2018 and November 2019 who underwent a dedicated DE GI bleeding CT protocol. Inclusion criteria were patients aged 18 years or older consenting to the use of medical records for research purposes, documented history of overt GI bleeding with a subsequent GI bleeding DE CT protocol performed clinically. This dual-phase, DE GI bleed CT protocol is the default CT exam performed for overt GI bleeding at our medical center for patients with abdominal diameter <45 cm.
DE GI Bleed CT Technique
The CT scans were performed on dual-source CT systems (Siemens Definition FLASH, Siemens FORCE; Siemens Healthineers, Germany). Tube voltage was determined by radiology technologist based on patient lateral width category (<30 cm, 30–35 cm, 36–45 cm, and 46–50 cm) with tube A having a voltage of 70, 80, 90, or 100 kV, respectively, and tube B having a voltage of Sn150 kV. Iohexol 350 (Omnipaque® 350, GE Healthcare, USA) was used as intravenous (IV) contrast material. The volume of injected contrast material was based on patient weight and ranged from 60 to 135 mL, followed by a 50 mL saline flush. The injection rate was 4 mL/second. The timing of the individual phases included a bolus-triggered arterial phase followed by a portal venous phase 70 s after the beginning of contrast injection. Triggering was performed by placing a region of interest on the aorta at mid-spleen level, and using a 150 HU threshold at 120 kV, and a transition delay of 6 s. For each phase of enhancement, axial 0.7 linear blend mixed kV, and dual energy images (VNC and virtual monoenergetic 50 keV images, and iodine maps) were generated using a slice thickness and reconstruction increment of 2 mm and 1 mm, respectively. Mixed kV coronal images using a slice thickness and reconstruction increment of 2 and 1 mm, respectively, were also created. The 0.7 linear blend mixed kV images were used as a surrogate for single energy images; however, they have been shown to have iodine signal superior to single energy 120 kV images. 8
Performance estimate in clinical practice
The standard practice in our institution is to report the presence/absence of active GI bleeding (contrast extravasation) as well as the location and source of bleeding, if possible. Clinical radiology reports for the CT exams were categorized as positive (i.e., the radiology report mentions evidence of definitive or possible GI bleed or potential source of active GI bleed) or negative. Additionally, the location of the suspected GI bleed and any potential source of GI bleed (e.g., tumor, vascular lesion, and inflammation, even in the absence of active bleeding) were recorded.
Reference standard
Two gastroenterologists investigated all patient records for information regarding the clinical course of GI bleeding, medical history, clinical outcome, endoscopic evaluations, the results of all testing at that hospitalization, as well as subsequent imaging available in the medical record. These gastroenterologists then created the reference standard for the presence, location, and cause of GI bleeding after consensus review of all clinical data.
Image interpretation to assess the benefit of portal venous phase and DE images
To assess the benefit of portal venous phase and DE imaging, we performed a multi-reader case–control study, which included all CT examinations in which clinical reports indicated intraluminal contrast extravasation, as the direct sign of GI bleeding (n = 31), regardless of the reference standard or presence of indirect signs of GI bleeding such as hematoma and tumor, and an equal number of CT exams without intraluminal contrast extravasation, randomly selected from the study population. Using a 100-point Likert scale (0 representing complete confidence in the absence of GI bleeding and 100 representing certainty that GI bleeding was present on a CT exam), as used by previous studies,
9
three pairs of GI radiologists with 10 and 32 years of experience in the first pair, 11 and 13 years in the second pair, and 14, and 34 years in the third pair (each radiologist in a pair reviewing half of the total number of cases and controls) independently reviewed imaging studies blinded to all clinical information (i.e., three radiologists interpreted each CT exam). Radiologists first evaluated arterial phase images and recorded their confidence in the following three items: (1) the presence of imaging findings indicating recent GI bleeding (e.g., intraluminal blood or contrast extravasation or hematoma); (2) presence of luminal extravasation; and (3) identifying etiology of GI bleeding (e.g., tumor, vascular lesions, and inflammation). Subsequently, at the same reading session, these radiologists evaluated additional portal venous phase images and recorded their confidence in the same items. Finally, radiologists viewed additional DE images (virtual monochromatic 50 keV and VNC images, and iodine maps) and recorded their confidence for the same three items, adding free-text comments if so desired. Fig. 1 provides an example of the combination of images the readers reviewed. Reviewed image sets. An example set of reviewed images of a 77-year-old man with rectal bleeding. The readers first reviewed mixed kV arterial (a) images, followed by mixed kV portal venous phase (b) images, and finally monoenergetic 50 keV arterial (c) images, as well as virtual non contrast (
Statistical analysis
A true positive examination required matching CT findings with the reference standard for both the presence and location of the GI bleeding. All data were analyzed using SPSS 25 (IBM, United States). Wilcoxon signed-rank test was used to evaluate the statistical significance of differences in continuous variables between pairs. Categorical variables were presented as a number (percentage). A p value of <0.05 was considered a statistically significant difference.
Results
Patients
Clinical characteristics of GI bleeding in the study population. CT findings include cases with evidence for active or recent GI bleeding on CT that were also confirmed on the reference standard, which was established by the review of all clinical records including endoscopy reports.

GI: gastrointestinal; AVM: arteriovenous malformation.
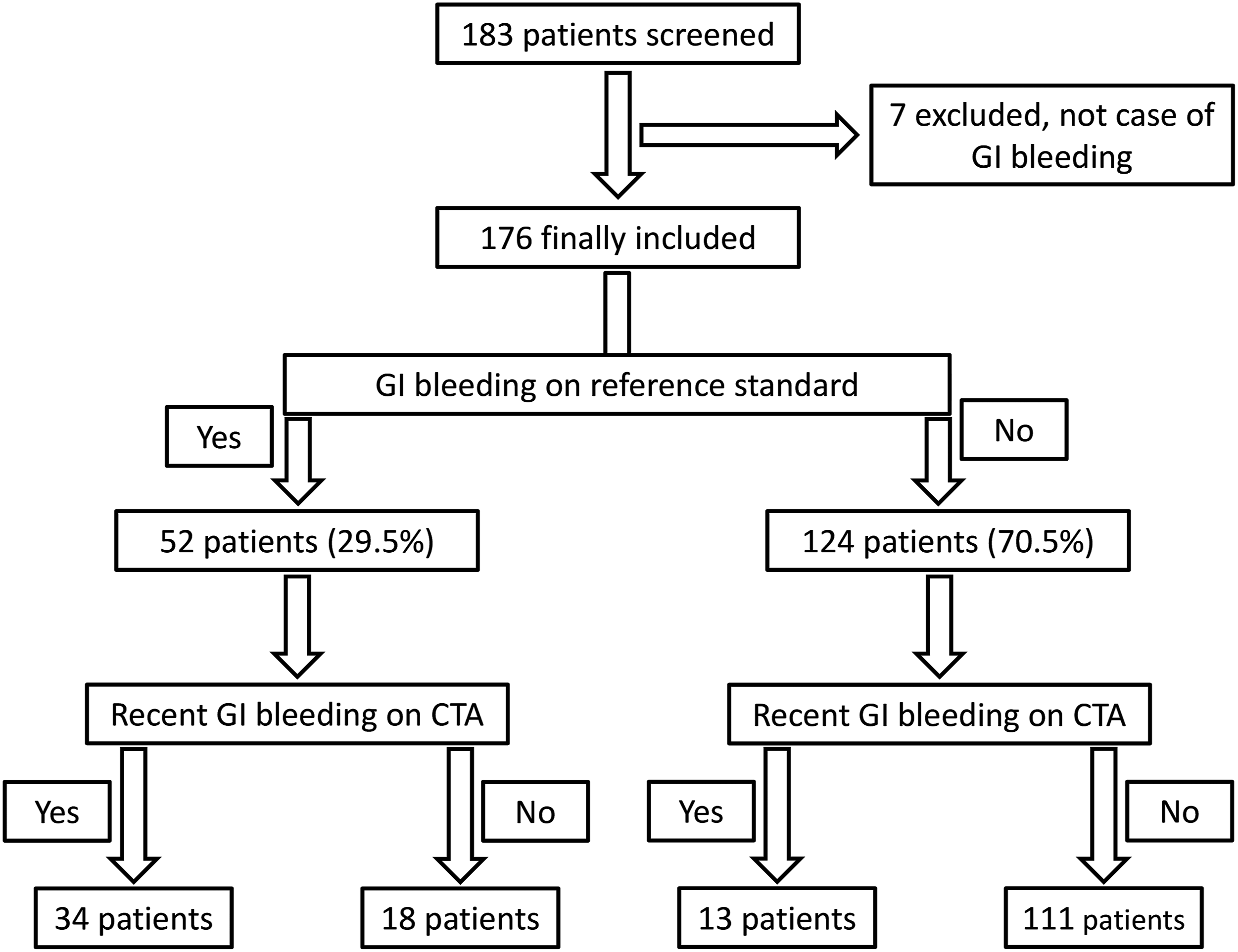
Study chart showing the utility of a dual phase, dual energy GI bleed CT protocol in identifying GI bleeding. The reference standard for the presence, location, and cause of GI bleeding was created by consensus between to gastroenterologists after the review of all clinical data including endoscopy, colonoscopy, and surgical reports.
In 52 cases with a diagnosis of GI bleeding by the reference standard, colonoscopy was performed in 19 cases with 16 cases positive for GI bleeding, with upper endoscopy being performed in 30 cases (27 being positive). Deep enteroscopy was performed in 10 cases (5 being positive), and capsule endoscopy was performed in 5 (4 positive). One positive case was confirmed with tagged RBC scan. In 11 cases, surgery reports confirmed GI bleeding in all of these cases. It should be noted that in some cases, more than one modality was positive for GI bleeding. In 4 cases, the diagnosis of GI bleeding of the reference standard was based on the review of clinical records and none of the above methods.
In 124 cases with no GI bleeding established on the reference standard, colonoscopy was performed in 38, endoscopy in 35, deep enteroscopy in 5, capsule endoscopy in 6, tagged RBC scan in 1, and surgery in 2. It should be noted that in some cases, more than one modality was used to evaluate the possibility of GI bleeding. In 45 cases, the clinical reports and follow-up were used to evaluate for GI bleeding.
Performance estimate of CT for overt GI bleeding
CT findings of GI bleeding (e.g., intraluminal blood/hematoma and tumor) were observed in 47/176 of the total patients (26.7%) (Figs. 3 and 4). Colonic diverticular hemorrhage. A case of active colonic diverticular hemorrhage in a 68-year-old male patient. Mixed kV coronal contrast-enhanced CT during the arterial phase reveals the extravasation of iodine contrast media from the diverticulum in the hepatic flexure (a: arrow). Mixed kV coronal contrast-enhanced CT during the portal venous phase shows extravasation of iodine contrast media diffusing in the colonic lumen (b: arrow). Esophageal varices and ischemic colitis. Findings other than extravasation of contrast material indicative of GI bleeding. A case of esophageal varices in a 62-year-old woman, as demonstrated by hypoenhancement of serpiginous luminal varices in the mixed kV arterial image (a: white circle) and hyperenhancement in mixed kV portal venous image (b: white circle). A case of ischemic colitis in a 62-year-old man, as demonstrated by fat stranding and intramural edema and wall thickening on the mixed kV arterial image (c: white circle) and mixed kV portal venous image (d: white circle). No evidence of vessel thrombosis was observed.

Compared to the clinical reference standard, the dual-phase dual energy CT protocol had a sensitivity of 65.4% (34/52; 95% CI: 95% CI: 50.8%–77.7%), a specificity of 89.5% (111/124; 95% CI: 82.4%–94.1%), a positive predictive value of 72.3% (34/47; 95% CI: 57.1%–83.9%), and a negative predictive value of 86.0% (111/129; 95% CI: 78.6%–91.3%). For patients with exclusively upper or lower GI bleeding, the sensitivity for colorectal bleeding was greater than the sensitivity for upper GI bleeding (79% vs. 50%, p=0.038). The sensitivity for patients bleeding only in the jejunum or ileum was 58%.
False-positive exams
Summary of clinical workup in patients with GI bleeding detected on dual phase, dual energy GI bleed CT protocol, but not on the reference standard.
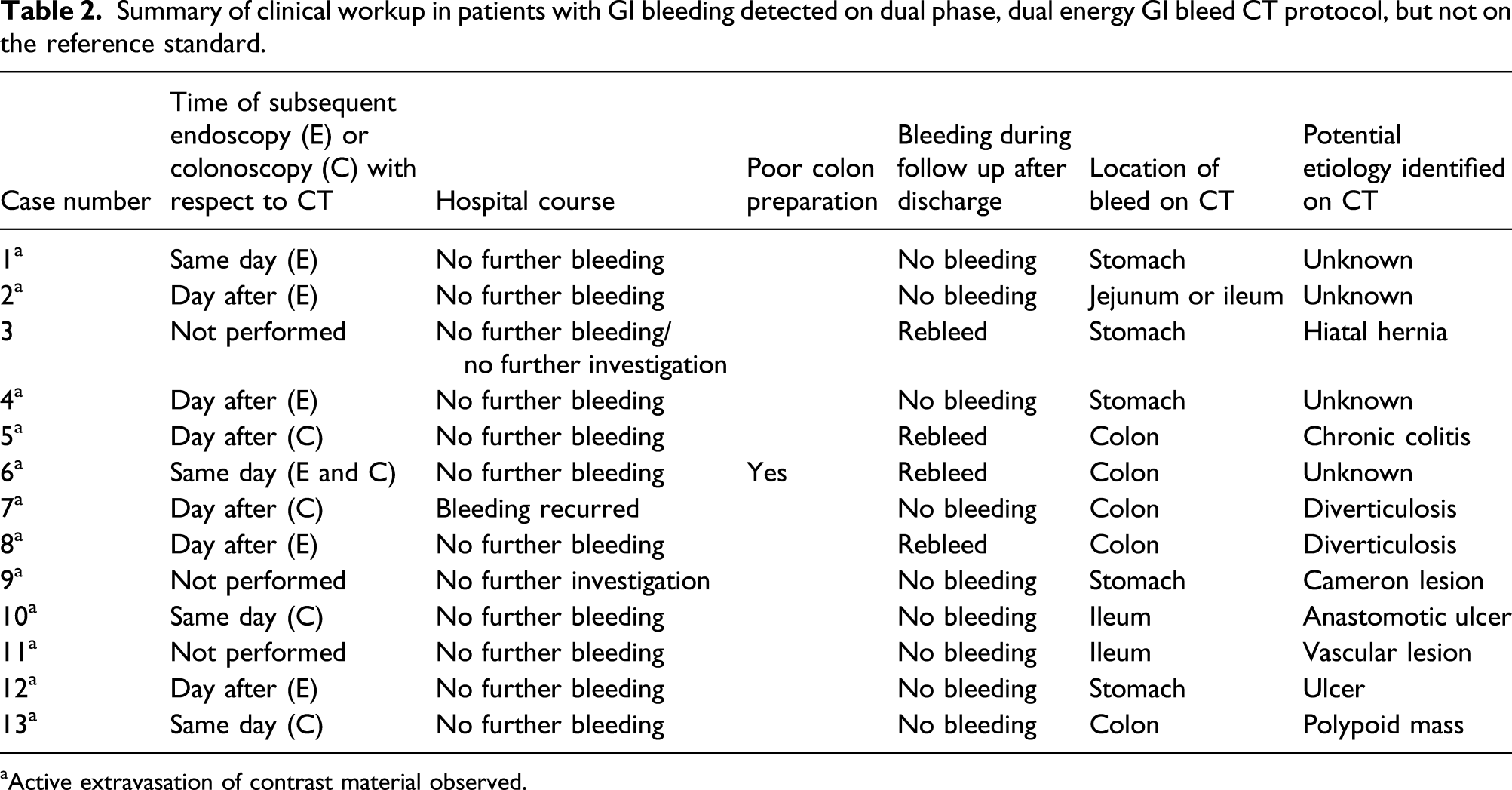
aActive extravasation of contrast material observed.

Contrast extravasation. Contrast extravasation from a colonic diverticulum detected on CT, which was not visualized on colonoscopy performed a day later. A: mixed kV arterial phase image; B: mixed kV portal phase image; C: virtual non-contrast image (note active extravasation of iodine is removed); D: monoenergetic DE 50 keV arterial phase image; E: monoenergetic 50 keV DE portal phase image. DE: dual-energy.
Added diagnostic value of portal phase and dual energy (DE) images
A case–control methodology was used to assess the value of or need for portal phase and dual energy images in 62 cases (25 women and 37 men). In this cohort, there were 22 CT examinations (6 women and 16 men) with GI bleeding determined by the reference standard and 40 cases without GI bleeding (19 women and 21 men) on the reference standard. There was no statistically significant difference between the two groups with regard to sex or age.
In patients with GI bleeding on the reference standard, when considering all CT findings indicative of active or recent GI bleed, the median (range) for average confidence of the radiologists for the presence of GI bleed after review of mixed kV arterial phase images was 69 (range 3–98), which increased to 89 (3–100) after adding mixed kV portal phase images (p = 0.002). Portal phase images increased diagnostic confidence of two readers by 10% or more in 32% of cases. Fig. 6 shows an example of an increase of more than 10% in readers’ confidence in the presence of GI bleeding from the arterial to the portal phase. For patients with confirmed GI bleeding, there was not a significant improvement in confidence after adding additional dual energy images (63 [3–100] vs. 66 [2–100], p = 0.72). Increased diagnostic confidence. A case example of an increase of more than 10% in readers’ confidence in the presence of contrast extravasation from the mixed kV arterial (a: white circle) to portal venous (b: white circle) phase in a 39 year-old male patient with active bleeding at the ileoanal anastomosis.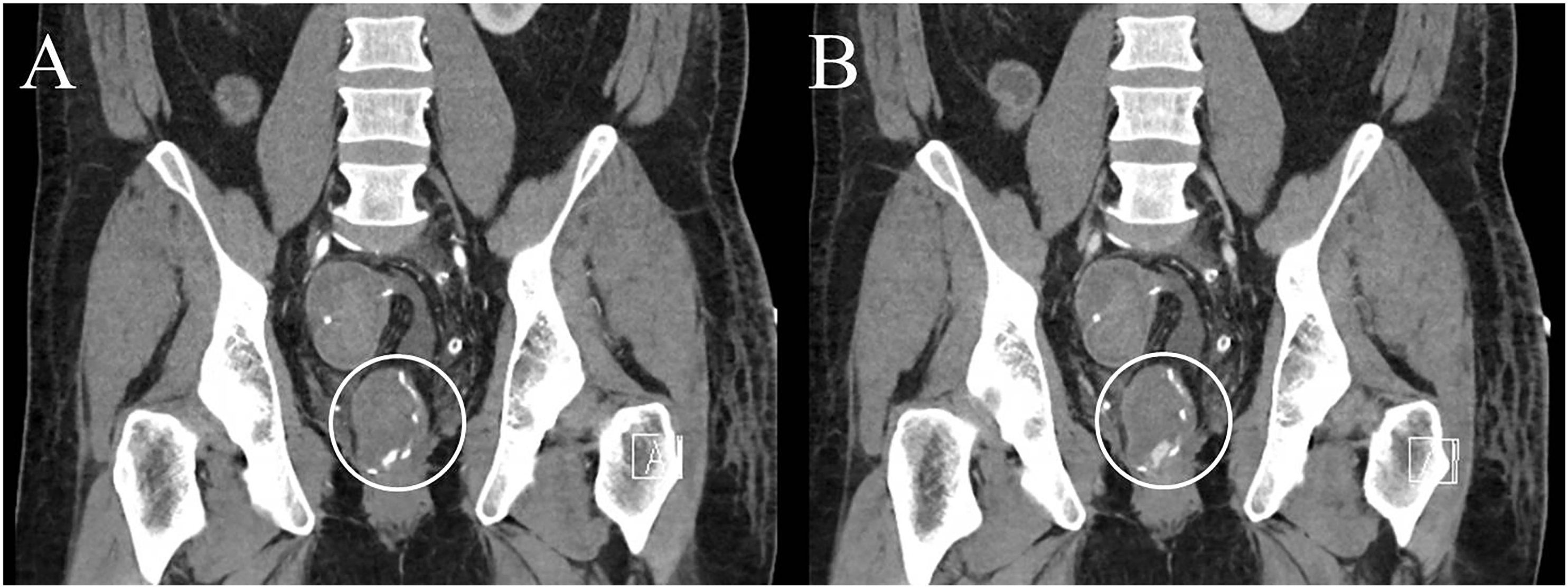
In patients without GI bleeding on the reference standard, there was no significant decrease in reader confidence in reviewing portal phase or dual energy images when considering all of the imaging findings that can be seen with recent GI bleeding (e.g., intraluminal blood or contrast extravasation; p = 0.175 and p = 0.236, respectively). The median (range) reader confidence in this group after reviewing arterial, portal, and dual energy images were 41 (2–95), 39 (1–100), and 38 (1–100), respectively. However, different findings were observed when readers considered their confidence in the assessment of luminal extravasation: the median (range) for average of confidence ratings for the presence of luminal contrast material extravasation was 23 (2–97) after review of mixed kV arterial phase images, which decreased to 12 (1–100) after adding mixed kV portal phase images (p = 0.006). Importantly, after further review of dual energy images, confidence in the presence of luminal extravasation appropriately and significantly further decreased to 8 (1–100; p = 0.018). Furthermore, in their free text comments, readers expressed that they elect to continue using the dual energy for reasons such as increased confidence in identifying hyperattenuating luminal debris.
Discussion
When using a clinical reference standard, a dedicated dual phase, dual energy acute GI bleed CT protocol in our clinical practice had a 65% sensitivity for detecting contrast extravasation or signs of recent bleeding, which is lower than previously reported in the literature.7,10,11 The specificity (90%) is similar to that of prior studies. In patients with GI bleeding on the reference standard, the addition of portal venous phase images increased the radiologists’ confidence in the presence of GI bleeding. Furthermore, in patients without GI bleeding on the reference standard, the addition of both portal venous and DE images improved confidence in the absence of active bleeding.
The dual phase, dual energy GI bleeding CT exam was the first exam investigating GI bleeding in 152/176 patients (86.4%). The GI bleed CT protocol was positive for findings indicative of GI bleeding in 47/176 (19.3%) patients compared to the reference standard, which was positive in 52/176 (29.5%) patients. CT demonstrated GI bleeding in 13 patients in whom the reference standard did not detect GI bleeding. It is likely that many of these cases are true positives (especially the large majority that demonstrated intraluminal contrast extravasation on CT) that were not detected on the reference standard due to delay in performing endoscopy, spontaneous cessation of bleeding, or mild bleeding.
The diagnostic accuracy of our GI bleed CT protocol was lower than in previously published studies. One meta-analysis of a total of 198 patients from nine institutions estimated CT to have a sensitivity and specificity of 89% and 85%, respectively. 11 In a more recent prospective study, Sun et al. also demonstrated improved performance but included only patients with signs of acute GI bleeding within 6 h of CT. In our study, we could not determine the exact time of the last episode of GI bleeding before GI bleed CT in some patients, which may reflect general medical practice at most institutions. A longer delay in GI bleed CT acquisition could result in a greater proportion of negative CT examinations, as bleeding is episodic. This theory is supported by evidence provided from an experimental study on the utility of CTA for detecting active GI bleeding at different bleeding rates. 10 Dorbitz et al. showed high sensitivity and specificity for CTA in a pig model of acute GI bleeding when the bleeding rate was 0.5–1.0 mL/min. 10
In the current study, the sensitivity of CT for detecting and localizing active GI bleeding was higher for bleeding from the lower compared to the upper GI tract. Furthermore, CT seems to be sensitive in detecting GI bleeding due to diverticulosis, one of the most common causes of lower GI bleeding. This implies a practical role for CT in patients with suspected lower GI bleeding, particularly since it can be performed without bowel preparation. Because colonoscopy without bowel preparation is not recommended due to lower diagnostic yield, 3 a dedicated GI bleed CT protocol may be an efficient initial triage which can then guide decision-making about which patients should undergo cleansing and diagnostic or therapeutic colonoscopy. One meta-analysis of 549 patients with lower GI bleeding found a combined sensitivity of 90% and a combined specificity of 92%. 12 However, the range of the diagnostic value of GI bleed CT in the included studies varied significantly, from 38% to 100% for sensitivity and 17% to 100% for specificity.
In the current study, portal venous phase images increased the diagnostic confidence of GI bleeding, similar to some prior studies assessing the utility of a portal venous phase in detecting acute GI bleeding.13–16 In one study, extravasation of contrast material was identified on portal (but not arterial) phase images in 40% of patients. 13 In another study, Liu et al. reported significantly higher rates for detection of GI bleeding on portal venous phase images compared to arterial phase images. 14 In contrast, Kim et al. reported a similar diagnostic accuracy for arterial, portal, and a combination of arterial and portal phase images in 46 patients with acute GI bleeding. 17 but similarly observed an increase in readers’ confidence in the majority of cases after using a combined set of arterial and portal phase images. Dobritz et al. investigated the utility of arterial and portal venous phase images in the detection of acute small bowel bleeding in a pig model. 10 They observed that low velocity bleeding usually was detectable only on portal venous phase images.
To the best of our knowledge, there has been no clinical evaluation of the added value (in terms of performance or radiologist confidence) of dual energy images in the diagnosis of acute GI bleeding. In the current study, we found dual energy images useful in improving diagnostic confidence in patients without GI bleeding on the reference standard. In patients with GI bleeding, the lack of improvement in readers’ confidence with the addition of dual energy images may result from the improvement in diagnostic confidence already afforded by portal venous phase imaging (i.e., where imaging findings are obvious). Geffroy et al. showed that true unenhanced images are helpful in identifying intraluminal debris, which may be confused with intraluminal contrast extravasation. 18 Additionally, Sun et al. showed that true unenhanced images can be replaced by VNC images in a DE GI bleed protocol without a change in observer performance, eliminating the need for unenhanced images. 7 It is likely that the improvement in diagnostic confidence in cases without GI bleeding can be attributed to the role of VNC images: in the absence of active bleeding, VNC images allow differentiation of intraluminal debris or calcification from active bleeding. Intraluminal debris may decrease confidence in negative exams without true unenhanced or VNC images as a radiologist may not be confident that the high attenuation material is not iodine.
The first and most notable limitation of this study is its retrospective nature. Second, the exact onset of GI bleeding and the period between the last bleeding episode and CT acquisition could not be determined in most cases. Consequently, we could not use the time interval between the acute GI bleed presentation and the CT examination as an inclusion criterion. Therefore, we decided to include consecutive cases using an intent-to-treat paradigm. It is likely that our study underestimates the sensitivity of GI bleed CT due to inclusion of patients with delayed presentation following GI bleeding. Our study design was created to prioritize evaluation of portal phase over dual energy images first so that results could be extrapolated to single energy CT.
In conclusion, the sensitivity of a dual-phase, dual energy GI bleed protocol had a high specificity and negative predictive value. Our findings support the use of a dedicated dual phase, dual energy CT protocol for triage in hemodynamically stable patients with GI bleeding, as portal phase image improved reader confidence in presence or absence of GI bleeding, and dual energy images further improved confidence in the absence of GI bleeding. Further studies are needed to determine which of these factors (signs, symptoms, time to imaging, etc.) lead to increased diagnostic yield.
Footnotes
Acknowledgments
The authors wish to thank Desiree Lanzino, PT, PhD, for her assistance in editing the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs. Fletcher and McCollough receive research funding from Siemens Healthineers, with funds provided to institution. D.H. Bruining performs consulting and receives research support from Medtronics. A.F. Halaweish, PhD, is an employee of Siemens AG.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research support for this work was provided, in part, to Mayo Clinic from Siemens Healthineers.
Consent to participate
IRB statement (materials and methods): After approval by our institutional review board, we examined the clinical records of patients with GI bleeding that underwent a dedicated dual energy GI bleeding protocol CT employing abdominopelvic CTA and portal phase imaging.
