Abstract
Background
Xanthogranulomatous cholecystitis (XGC) is an uncommon variant of chronic cholecystitis. The differential diagnoses of XGC include gallbladder cancer (GBC), adenomyomatosis, and actinomycosis of the gallbladder.
Purpose
To assess the usefulness of computed tomography (CT) findings in the diagnosis of XGC and differentiation from GBC.
Material and Methods
We retrospectively assessed the pathological and radiological records of 13 patients with pathologically proven XGC and 33 patients with GBC.
Results
Significant differences were observed for the following five CT findings: diffuse wall thickening (XGC = 85%, GBC = 15%, P < 0.01); absence of polypoid lesions (XGC = 100%, GBC = 48%, P < 0.01); intramural nodules or bands (XGC = 54%, GBC = 9%, P < 0.01); pericholecystic infiltration (XGC = 69%, GBC = 9%, P < 0.01); and pericholecystic abscess (XGC = 23%, GBC = 0%, P = 0.018). We defined the scoring system based on how many of the five CT findings were observed. Our scoring system, which included these findings, revealed that patients with three or more findings had sensitivity of 77% (95% confidence interval [CI] = 57–87) and specificity of 94% (95% CI = 86–98).
Conclusion
Our scoring system can assist in the differentiation of XGC from GBC.
Keywords
Introduction
Xanthogranulomatous cholecystitis (XGC) is an uncommon variant of chronic cholecystitis, characterized by a destructive inflammatory process and the formation of multiple yellow-brown intramural nodules and foamy histiocytes (1,2). The incidence of XGC among cholecystitis is 1.46% and it is treated via cholecystectomy (3).
The differential diagnoses of XGC include gallbladder cancer (GBC), adenomyomatosis, and actinomycosis of the gallbladder (2). Clinical symptoms of XGC include pain, positive Murphy sign, vomiting, leukocytosis, obstructive jaundice, cholangitis, and palpable mass (4). However, these clinical features are not specific to XGC, and there are often no clinical differences between patients with XGC and GBC, as both XGC and GBC are associated with destructive inflammation (5).
Treatment of XGC is done by simple cholecystectomy, which is also used for normal cholecystitis (6). There has been no consensus for the preference for open cholecystectomy or laparoscopic cholecystectomy. Prolonged operating times and technical difficulties have been observed, along with high conversion rates (laparoscopic to open cholecystectomy) of up to 80% (2). A diagnosis of GBC requires wider excision for tumor-free resection margins and regional lymph node dissection (7). However, these treatment options may be considered excessive for XGC. Therefore, the similarity between XGC and GBC can lead to unnecessary or excessive surgical interventions for XGC.
Computed tomography (CT) findings have revealed useful signs that can aid in differentiating XGC from GBC (4,5). However, the preoperative diagnosis has remained as a challenge. Thus, the aim of the present study was to assess the usefulness of CT findings in the diagnosis of XGC and differentiation from GBC.
Material and Methods
Patients
Our institutional review board approved this retrospective study. The requirement for informed consent from patients was waived because of the retrospective nature of the study. We retrospectively searched the pathological and radiological records of patients with pathologically proven XGC or GBC at our hospital over a period of 10 years from November 2007 to November 2017. The data of all patients who had undergone gallbladder resection during this time period were reviewed (n = 946). Forty-six patients were identified with pathologically proven XGC or GBC (13 XGC and 33 GBC). All of them were included in the present study.
Imaging protocol
CT was performed with a 64-MDCT scanner (Aquilion ONE, Canon, Tochigi, Japan). The protocol was: collimation = 0.5 mm; thickness = 5.0 mm; pitch = 0.813; and X-ray tube potential = 120 kVp. A tube current of 290–550 mA with auto tube current modulation technique was used. After a non-contrast scan, a total of 100 mL of non-ionic iodinated contrast agent was administered to the patients at a rate of 3.0 mL/s. The contrast-enhanced CT scans were initiated 60 s after the injection. The liver dynamic contrast-enhanced CT scans were performed after the injection as follows: arterial phase = 35 s; portal phase = 55 s; delayed phase = 90 s.
Image analysis
Enhanced CT images were retrospectively reviewed by a radiologist who had 13 years of experience as a radiologist and was certified by the Japanese Society of Radiology. In case of dynamic CT, we adopted delayed phase. We reviewed CT findings that were reported in previous studies: existence of gall stones; patterns of wall thickening–diffuse or not; existence of polypoid lesions; intramural nodules or bands; mucosal line enhancement; continuous mucosal line; existence of pericholecystic infiltration; existence of a pericholecystic abscess; loss of interface between the gallbladder and liver; early enhancement of the liver bed; common bile duct dilation (>10 mm); and lymphadenopathy > 10 mm. In these findings, we picked up the statically different findings and made a scoring system.
To assess the objectivity of this scoring system, we additionally performed a blind sub-analysis on the reproducibility of five CT findings by two reviewers (with 24 and 17 years of experience in abdominal radiology, respectively). Each of the two reviewers also blindly analyzed all cases.
Statistical analysis
Wilcoxon’s test and Fisher’s exact test were used to verify the numerical and proportional differences, respectively, between the two groups. A P value < 0.05 was considered statistically significant. We used the receiver operating characteristic (ROC) curve for our preoperative scoring system with CT findings. Kappa test was used for assessment of inter-observer variability. A K value of up to 0.20 was interpreted as slight agreement, 0.21–0.40 as fair agreement, 0.41–0.60 as moderate agreement, 0.61–0.80 as substantial agreement, and 0.81 or greater as almost perfect agreement. These statistical analyses were performed using SPSS software (version 21, IBM SPSS Statistics, Tokyo, Japan).
Results
Patient characteristics
Patient characteristics are summarized in Table 1. In GBC, 23 (72%) patients were stage IIB or lower.
Patient characteristics.
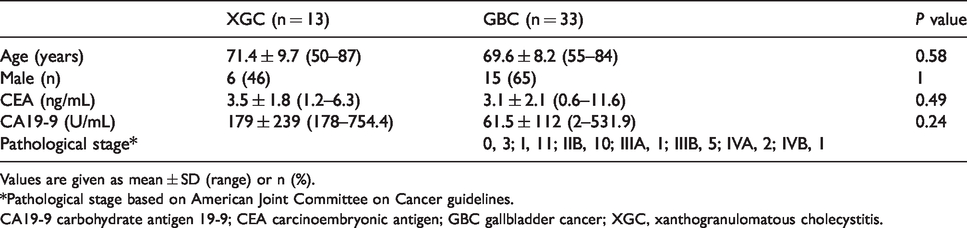
Values are given as mean ± SD (range) or n (%).
*Pathological stage based on American Joint Committee on Cancer guidelines.
CA19-9 carbohydrate antigen 19-9; CEA carcinoembryonic antigen; GBC gallbladder cancer; XGC, xanthogranulomatous cholecystitis.
Analysis of CT findings
Patients with XGC included 11 contrasted CT, one liver dynamic CT, and one non-contrasted CT. Patients with GBC included 16 contrasted CT, 14 liver dynamic CT, and three non-contrasted CT. Examples of CT findings from patients with XGC and GBC in the present study are shown in Fig. 1. The incidence, sensitivity, specificity, and accuracy of CT findings are summarized in Table 2. Significant differences in the incidences were observed for the following five CT findings: diffuse wall thickening; absence of polypoid lesions; intramural nodules or bands; pericholecystic infiltration; and pericholecystic abscess. Diffuse wall thickening was observed in 11 (85%) patients with XGC but in only 5 (15%) patients with GBC (P = 0.00002). Diffuse wall thickening had a high sensitivity (85%), high specificity (85%), and the highest accuracy (85%). No polypoid lesions were present in patients with XGC; however, they were present in 17 (52%) patients with GBC (P = 0.0007). The absence of polypoid lesions had the highest sensitivity (100%), specificity of 52%, and accuracy of 65%. Intramural nodules or bands were observed in 7 (54%) patients with XGC, but they were observed in only 3 (9%) patients with GBC (P = 0.002). Their sensitivity, specificity, and accuracy were 54%, 91%, and 80%, respectively. Pericholecystic infiltration was present in 9 (69%) patients with XGC, but it was present only in 3 (9%) patients with GBC (P = 0.0001); it had high accuracy (85%), specificity (91%), and a sensitivity of 69%, similar to the values for diffuse wall thickening. Pericholecystic abscesses were present in 3 (23%) patients with XGC, but they were not present in patients with GBC (P = 0.018); this finding had the lowest sensitivity (23%), highest specificity (100%), and moderate accuracy (78%).

Examples of CT findings from patients with XGC and GBC in this study. (a) XGC with gall stone (white arrow); (b) XGC with diffuse wall thickening (white arrows); (c) GBC T2bN0M0 with gallbladder mass (white arrowhead); (d) XGC with intramural hypo attenuating nodules (white arrows); (e) XGC with more enhancement of the mucosal line than of the liver (white arrow); (f) XGC with a continuous mucosal line (white arrow); (g) XGC with pericholecystic infiltration (white arrow); (h) XGC with pericholecystic abscess (white arrow); (i) XGC with loss of interface between the gallbladder and liver (white arrow); (j) XGC with early enhancement of the liver (white arrow); (k) GBC T3N0M0 with common bile duct dilation: 12 mm in size (white arrowhead); (l) GBC T3N0M0 with lymphadenopathy: 11 mm in size (white arrowhead). CT, computed tomography; GBC, gallbladder cancer; XGC, xanthogranulomatous cholecystitis.
Incidence, sensitivity, specificity, and accuracy of CT findings for patients with XGC and GBC.

Values are given as n (%) or % (95% CI).
*Four patients for whom only plane CT findings were available (1 XGC, 3 GBC) were excluded.
CI, confidence interval; CT, computed tomography; GBC, gallbladder cancer; XGC, xanthogranulomatous cholecystitis.
During the assessment of mucosal findings, four patients (one XGC and three GBC) were excluded because only non-enhanced CT findings were available. There was no significant difference in mucosal line enhancement. Continuous mucosal lines were observed more frequently in patients with GBC, but this difference was not significant.
No significant differences between XGC and GBC were observed in the occurrence of gallstones, loss of interface between the gallbladder and liver, early enhancement of the liver, common bile duct dilation, or lymphadenopathy > 10 mm in diameter.
We defined the scoring system by considering how many out of five CT findings were observed. If three out of five CT findings were observed, the score was 3. The ROC curves for this scoring system are shown in Fig. 2; the area under the curve was 0.941. If the score included 3 or more findings, then sensitivity was 77% (95% confidence interval [CI] = 57–87) and specificity was 94% (95% CI = 86–98). This score had the highest accuracy (89%, 95% CI = 78–95) compared to that of each CT finding. Additional analysis by two inter-observers revealed sensitivity, specificities, and accuracy were 77%, 88%, and 85%, respectively, for reviewer 1, and 77%, 82%, and 80%, respectively, for reviewer 2. Each of the K values were 0.786 and 0.695. They showed substantial agreement among for three reviewers.

Receiver operating characteristic curves used to score the computed tomography findings. The AUC was 0.941. If the score included three or more findings, then sensitivity was 77% (95% CI = 57–87) and specificity was 94% (95% CI = 86–98). AUC, area under the curve; CI, confidence interval.
Discussion
Preoperative diagnosis of XGC and GBC continues to be a challenge. The scoring system in the present study had a high diagnostic performance, based on five findings on CT: diffuse wall thickening; absence of polypoid lesions; intramural nodules or bands; pericholecystic infiltration; and pericholecystic abscess.
The results of the present study and previous reports are summarized in Table 3 (4,5,8–11). Diffuse wall thickening was observed in 37 (91%) patients with XGC. Four previous studies support our results: XGC patients showed significant differences from GBC patients (4,5,8–11). In the study by Rajaguru et al. (5), the absence of polypoid lesions was observed in 74% of patients with XGC. Intramural nodules or bands were consistent with those in xanthogranulomatous lesions, necrosis, or abscesses (9). Similar to our results, this finding was observed in 54%–100% of patients with XGC (4,5,8–11). Pericholecystic infiltration was observed in 61%–69% of patients with XGC (4,9,10). Goshima et al. (4) reported this finding in 53% of patients with GBC, which was not significantly different from the results for patients with XGC. However, pericholecystic infiltration was observed only in 9% of patients with GBC in our study. The low incidence of the presence of pericholecystic infiltration may have resulted from the higher number of patients with early stages of disease (up to IIB) in the GBC group in our study. Pericholecystic abscesses were rarely documented. We distinguished pericholecystic abscesses from intramural nodules by determining whether they were present in the extramural or intramural hypoattenuated area.
Incidence of CT findings in our study and previous reports.

Values are given as percentages of patients.
*Differences in percentages are statistically significant.
CT, computed tomography; GBC, gallbladder cancer; XCG, xanthogranulomatous cholecystitis.
Mucosal line enhancement and continuous mucosal lines were reported as useful findings for the diagnosis of XGC (4,5,8,11). However, in the present study, these findings did not show significant differences between XGC and GBC; therefore, they were excluded from our scoring system. In contrast to our results, continuous mucosal lines were observed in 10%–35% of patients with GBC in other studies (4,5,8,11). A possible explanation for our results is the higher prevalence of patients with GBC of an early stage.
The mucosal findings documented in previous reports were based on enhanced CT conditions and a GBC group. The five CT findings reported in this study did not require enhanced CT. Therefore, our scoring system was considered to be widely available regardless of contrast agent in CT scanning. In addition, our scoring system allows differential diagnosis of GBC at an early stage, which is clinically difficult to distinguish from XGC.
The present study had some limitations. First, all CT images were evaluated by a single radiologist without blinding. Second, it included a limited number of patients. Thus, this study had selection bias and observer bias. Based on our scoring system, two additional reviewers’ accuracy was 80% (reviewer 1) and 85% (reviewer 2) (data not shown). Therefore, our scoring system would have a high accuracy (>80%) regardless of the reviewer.
As diagnostic CT findings of acute cholecystitis, pericholecystic fat stranding, mural stratification, pericholecystic hypervascularity, spontaneous hyper-attenuation of gallbladder wall, short (≥32 mm) and long (≥74 mm) gallbladder axis enlargement, and gallbladder wall thickening (≥3.6 mm) were reported (12). Three of them are common to the findings of the present study. This reflects the similarity of XGC and acute cholecystitis.
The effects of preoperative percutaneous drainage of acute cholecystitis were reported (13). However, we must avoid this for GBC in the points of dissemination of carcinoma. Preoperative image diagnosis of XGC and GBC is important; further prospective studies are required to verify the usefulness of this scoring system. As another modality of image diagnosis, magnetic resonance imaging (MRI) findings were reported. Using in-phase and opposed-phase chemical shift imaging, XGC nodules showed reduced signal intensity on out-of-phase. Diffusion destruction is seen more often in GBC than in XGC (2). A combination of our scoring system and MRI findings would be able to obtain a correct diagnosis of XGC.
In conclusion, significant differences in the incidence of diffuse wall thickening, absence of polypoid lesions, intramural nodules or bands, pericholecystic infiltration, and pericholecystic abscess were observed between patients with XGC and those with GBC. Our scoring system (with three or more of five findings) could assist in the differential diagnosis of XGC from GBC.
Footnotes
Acknowledgments
The authors thank Dr. Toda Joe and Dr. Kimura Tomo for their helpful additional analysis as inter-observers.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
