Abstract
Background
Identification of the perforator vein is important for treating lower extremity varix.
Purpose
We evaluated the ability of 40-keV advanced monoenergetic images to depict the perforator vein in patients with lower extremity varix.
Material and Methods
Thirty-three patients aged 52–86 years were examined with contrast-enhanced dual-energy computed tomography (CT) and advanced virtual monoenergetic images (40 keV) were reconstructed. For evaluating enhancement of a lower extremity vein and the difference in CT number between the vein and muscle, we set the region of interest on the popliteal vein (PV). We also evaluated the ability of 100-kVp and 40-keV volume-rendering (VR) images to depict the perforator veins.
Results
The mean CT numbers of the PV at 100 kVp and 40 keV were 113 ± 16 and 321 ± 63 HU, respectively (P < 0.01). In 40-keV transverse images of 33 patients, 84 of the perforator veins were detected. In those 84 veins, 70 (83%) were depicted and 14 (17%) were not depicted on VR images that were reconstructed from 40-keV transverse images. At 100 kVp, 10 (12%) of the perforator veins could be depicted in VR images because the muscles buried them or the PVs were blurred due to insufficient enhancement.
Conclusion
The advanced monoenergetic reconstruction technique is useful for evaluating the perforator vein in patients with lower extremity varix.
Keywords
Background
Identification of the perforator vein is important to treat lower extremity varix (1). Contrast-enhanced computed tomography (CE-CT) is useful for evaluating the perforator vein. Vascular surgeons prefer volume-rendering (VR) images, which allow them to understand overall anatomical structure with a bird’s eye view. However, in CE-CT, enhancement of the vein is sometimes insufficient, especially in VR images, to depict the perforator vein. In VR images, the muscles of the lower extremities are difficult to remove because CT values of muscles are similar to those of veins. Using dual-energy CT (DE-CT), advanced virtual monoenergetic images imitating low-keV (kilo electron volt) images can be reconstructed (2). In low-keV images, contrast enhancement can be stronger than in usual CE-CT images (3). In the present study, we evaluated the ability of 40-keV advanced monoenergetic images to depict the perforator vein in patients with lower extremity varix in VR images.
Material and Methods
The Institutional Review Board approved this retrospective study. The requirement for written informed consent was waived for the retrospective analysis. Forty-four patients with varix in their lower extremities were examined with DE-CT between April 2016 and February 2017. Eleven patients with other cardiovascular diseases were excluded, including cardiomegaly in five cases, aortic dissection in four cases, arterio-venous shunt in one, and aortoiliac occlusive disease (Leriche syndrome) in one case, since they may affect deep venous enhancement of lower extremity due to abnormal circulation. Thirty-three patients aged 52–86 years (mean age = 68 years) were included in this study. DE-CT (Somatom Definition Flash, Siemens Healthcare, Forchheim, Germany) was used for all patients in this study using 100-kVp (peak kilo volt) and 140-kVp tube voltages with 0.9 pitch. Acquisition collimation was set to 32 × 0.6 mm. Advanced virtual monoenergetic images (40 keV) were reconstructed for each patient. All imaging data were reconstructed with a slice thickness of 1.5 mm at 1-mm increments. A contrast medium (300 mgI/mL, 150 mL) was injected through an upper extremity vein at a flow rate of 2.0 mL/s. Scanning was performed 210 s after the start of contrast medium injection.
Subjective image evaluation
We evaluated how clearly the perforator veins were depicted in VR images generated from 100-kVp and 40-keV images using a 4-point scale as follows: grade 3 = clear; grade 2 = mildly blurred or slightly masked with muscle but adequately diagnostic; grade 1 = non-diagnostic due to blurring veins or masking with surrounding muscle; and grade 0 = not visualized.
Grades 2 and 3 were considered “depicted.” Two board-certified radiologists were blinded to the imaging techniques and evaluated the images individually. During image evaluation, any parameters of VR images could be changed freely to detect the perforator veins. Any discrepancies were resolved by consensus.
Objective image evaluation
For evaluating enhancement of a lower extremity vein and the difference in CT values between the vein and muscle, we set the region of interest on the popliteal vein (PV) and gastrocnemius muscle (GM) (Fig. 1). We then calculated the difference between the CT values of the PV and GM (PV-GM value). These CT values that were obtained from 100-kVp and 40-keV images were compared.

Transverse contrast-enhanced computed tomography images at the popliteal level in a 73-year-old woman: (a) 100-kVp image; (b) 40-keV image. Advanced virtual monoenergetic images were reconstructed at 40 keV (b). Regions of interest were set on the popliteal veins and gastrocnemius muscles for each image.
Diameters of the perforator veins were measured in 40-keV transverse images. We then compared diameters of perforator veins that were depicted in VR images with those that were not depicted in VR images.
Statistical analysis
The Shapiro–Wilk W test was used to determine whether sizes of the perforator veins were normally distributed. Parametric data were compared using the paired t-test. The non-parametric subjective data of the perforator veins were compared using the Wilcoxon signed rank test. For this subjective evaluation, inter-observer agreement was calculated by using kappa statistics. Kappa scores of 0.41–0.60, 0.61–0.80, and >0.80 were considered to indicate moderate, good, and excellent agreement, respectively.
Results
In 40-keV transverse images of 33 patients, 84 of the perforator veins were detected on both of transverse 100 kVp and 40 keV images (Fig. 2). In those 84 veins, 10 (12%) and 70 (83%) were depicted on VR images reconstructed from 100 kVp and 40-keV transverse images, respectively (Fig. 3). The grades of image quality for the perforator veins are shown in Table 1. Average scores of the 100-kVp and 40-keV images were 0.4 ± 0.9 and 2.4 ± 1.1, respectively (P < 0.001). Inter-observer agreement was excellent (Kappa = 0.83 and 0.92 for 100-kVp and 40-keV VR images, respectively).
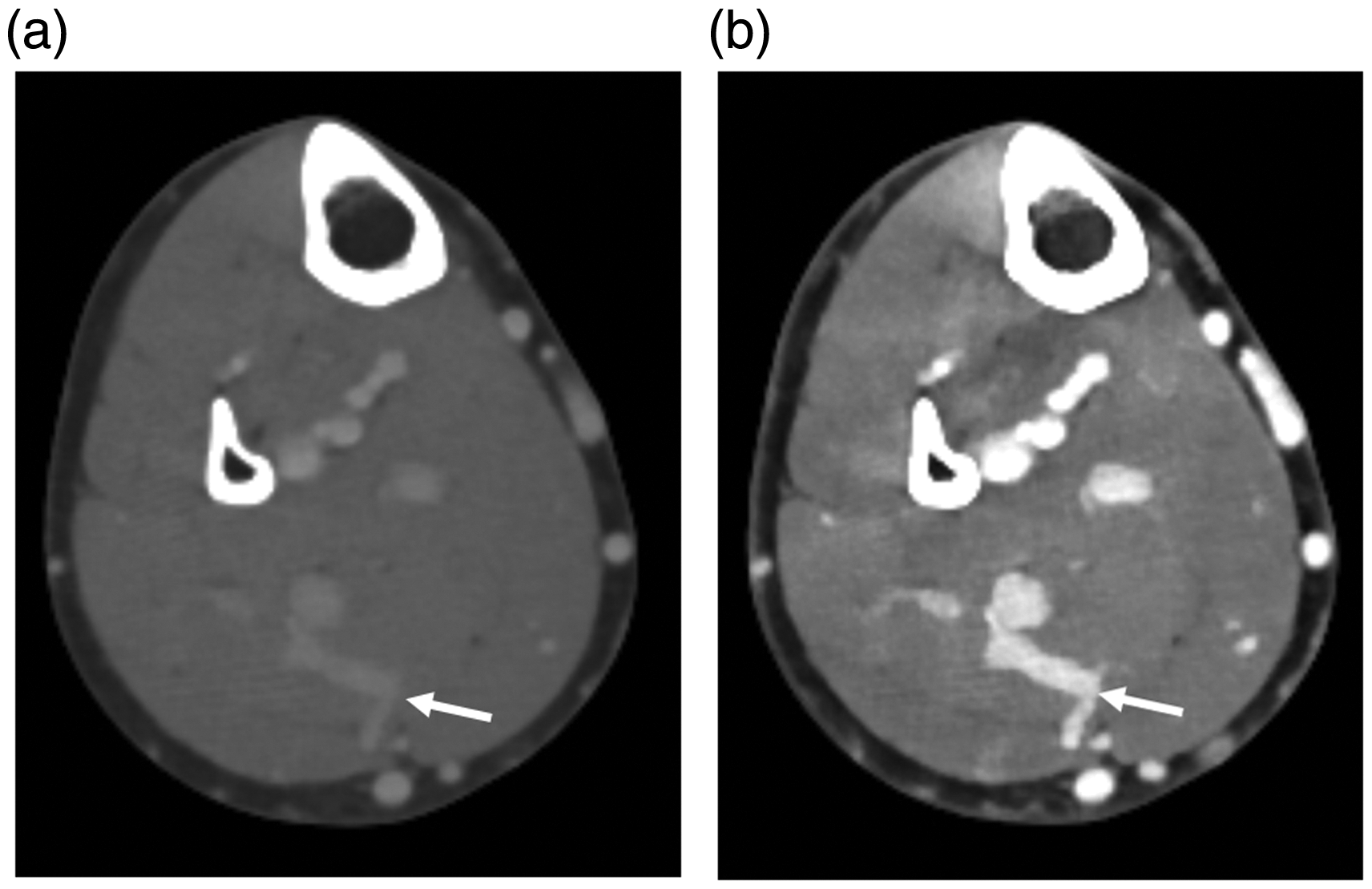
Transverse contrast-enhanced computed tomography images of the perforator vein (arrow) in a 73-year-old woman: (a) 100-kVp image; (b) 40-keV image. Window level and width of both images were 100 and 600, respectively.

Volume-rendering images of the lower extremities in a 73-year-old woman: (a) 100-kVp image; (b) 40-keV image. (a) In a 100-kVp volume-rendering (VR) image, the distal perforator vein (arrow) was slightly blurred but diagnostic (grade 2). The proximal perforator vein (dashed arrow) was non-diagnostic due to blurring and masking with muscle (grade 1). (b) In a 40-keV VR image, the perforator veins were clearly depicted in 40-keV images (arrows) (grade 3).
Image quality of two types of volume-rendering images.

In the 14 perforator veins that could not be detected, the reason for this lack of detection for 12 veins was that the sizes were too small to be depicted in VR images. Although the remaining perforator vein was not small, this vein could not be sufficiently enhanced because the volume of contrast medium was probably insufficient owing to a large space of the varix.
The mean (± SD) CT value of the PV at 100 kVp was significantly lower than that at 40 keV (113 ± 16 vs. 321 ± 63 HU, P < 0.01). The mean CT value of the GM at 100 kVp was significantly lower than that at 40 keV (67.1 ± 5.9 vs. 113 ± 15.9 HU, P < 0.01). The mean PV-GM value at 100 kVp was significantly lower than that at 40 keV (48.9 ± 17.1 vs. 208 ± 57.7 HU, P < 0.01).
The mean (± SD) diameter of perforator veins that could not be depicted in VR images was smaller than that of perforator veins that could be depicted (2.2 ± 0.40 vs. 4.1 ± 0.95 mm, P < 0.01).
Discussion
The perforator veins of patients with a varix in the lower extremities are usually examined with ultrasonography or CE-CT (4). Ultrasonography is a useful modality because it is non-invasive and does not use ionizing radiation. However, the quality of this modality depends on skills of the examiner and the condition of the patient’s leg, including the extent of subcutaneous edema and volume of fat. Moreover, detecting all of the perforator veins in the lower extremities can sometimes be difficult. Because CT can scan the whole lower extremities, CE-CT has high utility for screening veins in the lower extremities (5). The VR technique is used for viewing the whole structure of vessels in CT angiography. However, in usual delayed scans of CE-CT (100 kVp), surrounding tissues, such as the muscle, can prevent observation of veins because enhancement is sometimes insufficient. In this study, we demonstrated the utility of 40-keV images for depicting the perforator vein in VR images (Fig. 3 and Table 1).
Of the 84 perforator veins, 14 (17%) could not be depicted in VR images that were reconstructed from 40-keV images. These undepicted veins were smaller than the perforator veins that were depicted in VR images. According to the guideline of the Society for Vascular Surgery and the American Venous Forum (1), “pathological” perforating veins are defined as those with an outward flow with a duration ≥500 ms, with a diameter ≥3.5 mm, and a location of beneath healed or open venous ulcers. The Japanese guideline for management of lower leg ulcers/varicose veins (4) defined the normal diameter of the communicating branches as ≥3 mm. Therefore, VR images that are reconstructed from 40-keV images are considered useful because pathologically dilated perforator veins can be depicted, but small perforator veins cannot be depicted.
In low-keV images that are reconstructed using the virtual monoenergetic technique, contrast enhancement can be strengthened compared with usual CE-CT images, as shown in our study. Low-keV images of the conventional virtual monoenergetic technique have a large amount of noise. The surface of VR images that are reconstructed from transverse images with a higher amount of noise tend to be rougher (6). In the advanced monoenergetic technique, noise of low-keV images can be reduced by fusion with 70-keV images, which have a lower amount of noise (7). Moreover, iterative reconstruction technique also helps to reduce image noise. It was reported that radiation doses of DE-CT scan for monoenergetic imaging were as high as those of CT one generation ago (8, 9). However, radiation doses of DE-CT can be higher than those of low kVp scans (e.g. 70 or 80 kVp) with a latest CT scanner.
The perforator vein of one case could not be depicted because of insufficient contrast enhancement for a large varix. When the varix is large, increasing the contrast medium or pumping by massaging the patient’s lower extremity might be required.
The present study had several limitations. First, not all of the perforator veins of this study were evaluated with ultrasonography or pathologically. However, we considered CE-CT could evaluate vascular structures objectively. Second, contrast enhancement was not evaluated at the perforator veins but at the popliteal vein. Since many perforator veins are small, partial volume effect may not be avoidable. Therefore, we considered the larger veins (the popliteal veins) were better to objectively measure enhancement of veins. Third, slice thickness of CT images was 1.5 mm. It might be so thick that detectability by vein size and visibility of the perforator veins of VR images could be affected.
In conclusion, the advanced monoenergetic reconstruction technique with 40-keV images is useful for evaluating the perforator vein in patients with lower extremity varix.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by JSPS KAKENHI Grant Number 18K07724.
