Abstract
Tuberous sclerosis (TS) is a relatively rare multi-organ disorder generally diagnosed in infancy and described as a clinical triad of sebaceous adenoma, mental retardation, and seizures. Angiomyolipoma (AML) is the most common benign renal tumor usually found incidentally as a solitary small echogenic lesion on grayscale ultrasound. Less commonly, it is part of the TS complex and is seen as multiple lesions in both kidneys. We describe an unusual case of TS incidentally diagnosed in a 37-year-old female patient with several and bilateral renal AMLs and a single cortical–subcortical tuber in the right parieto-occipital cerebral lobe.
Introduction
Tuberous sclerosis (TS) or Bourneville-Pringle syndrome is a rare neurocutaneous disease with autosomal dominant inheritance and a prevalence in the general population of 1/10,000 (1). Clinically, TS is characterized by the following triad: epilepsy; low IQ; and cutaneous angiofibromas; however, these are usually observed only 40% of cases (2–4). Angiomyolipoma (AML) is a rare benign neoplasm found in < 1% of surgically removed tumors (5). It is the most common mesenchymal tumor of the kidney, characterized by a variable mixture of three kinds of tissue in a variety of percentages: mature fat; smooth muscle; and abnormal blood vessels with thick walls (6). Sporadic AML has an incidence of 0.3–3%. Rarely, it occurs in 50–80% of patients with TS. We present an unusual case of TS.
Case report
A 37-year-old female patient came to our department to perform a routine abdominal ultrasound (US). US examination showed multiple and bilateral nodular renal lesions which were mainly hyperechoic with regular margins and vascular signal on color Doppler. These findings were suggestive of multiple AMLs (Fig. 1). The largest lesion showed a maximum diameter of 60 mm and was located at the upper pole of the right kidney. No abnormalities of the renal veins were observed on Doppler evaluation. The patient was otherwise in good health and did not report any neurological symptoms. Clinical examination did not show any palpable masses in the abdomen and blood pressure values were within the normal range. At physical examination, about six rounded and reddish papules were detected at the right nasal wing, with a maximum diameter < 1 cm. These papules corresponded to angiofibromas relapsed after surgical removal carried out three years earlier. Personal and familiar history were negative for kidney disease. Blood examinations were unremarkable, except for an initial mild proteinuria (18 mg/dL). Magnetic resonance imaging (MRI) of the abdomen was subsequently performed using a 3-T scanner (Siemens, Trio), before and after intravenous administration of Gd-DTPA at a dose of 0.1 mmol/kg. Several (up to 60) nodular lesions were detected in both kidneys with predominantly adipose content in sizes of 2–60 mm; these lesions were moderately hyperintense on T1-weighted (T1W) images, hyperintense on T2-weighted (T2W), showing loss of signal on fat saturation sequences and mild enhancement on post-contrast T1W sequences (Fig. 2). These MRI findings were consistent with multiple and bilateral renal AMLs. The largest mass (60 × 52 × 50 mm), located at the upper pole of the right kidney, showed hemorrhagic content. The excretory function was preserved on both kidneys. Subsequently, the patient underwent a contrast-enhanced brain MRI that showed in the cortical–subcortical side of the right parieto-occipital cerebral cortex, the presence of a single lesion characterized by low signal intensity on T1W images and high signal on T2W images without evidence of contrast enhancement on T1W sequences; these findings were consistent with a cortical–subcortical tuber (Fig. 3).

US of the right kidney shows several hyperechoic nodular lesions (arrows) suspicious for angiomyolipomas.

(a) Unenhanced T1W image and (b) contrast-enhanced T1W fat-saturated MR image on the axial plane showing multiple bilateral renal lesions. Intralesional fat components demonstrate loss of signal. The largest lesion in the upper pole of the right kidney (arrow) exhibits moderate and inhomogeneous enhancement. These findings are consistent with multiple renal angiomyolipomas.
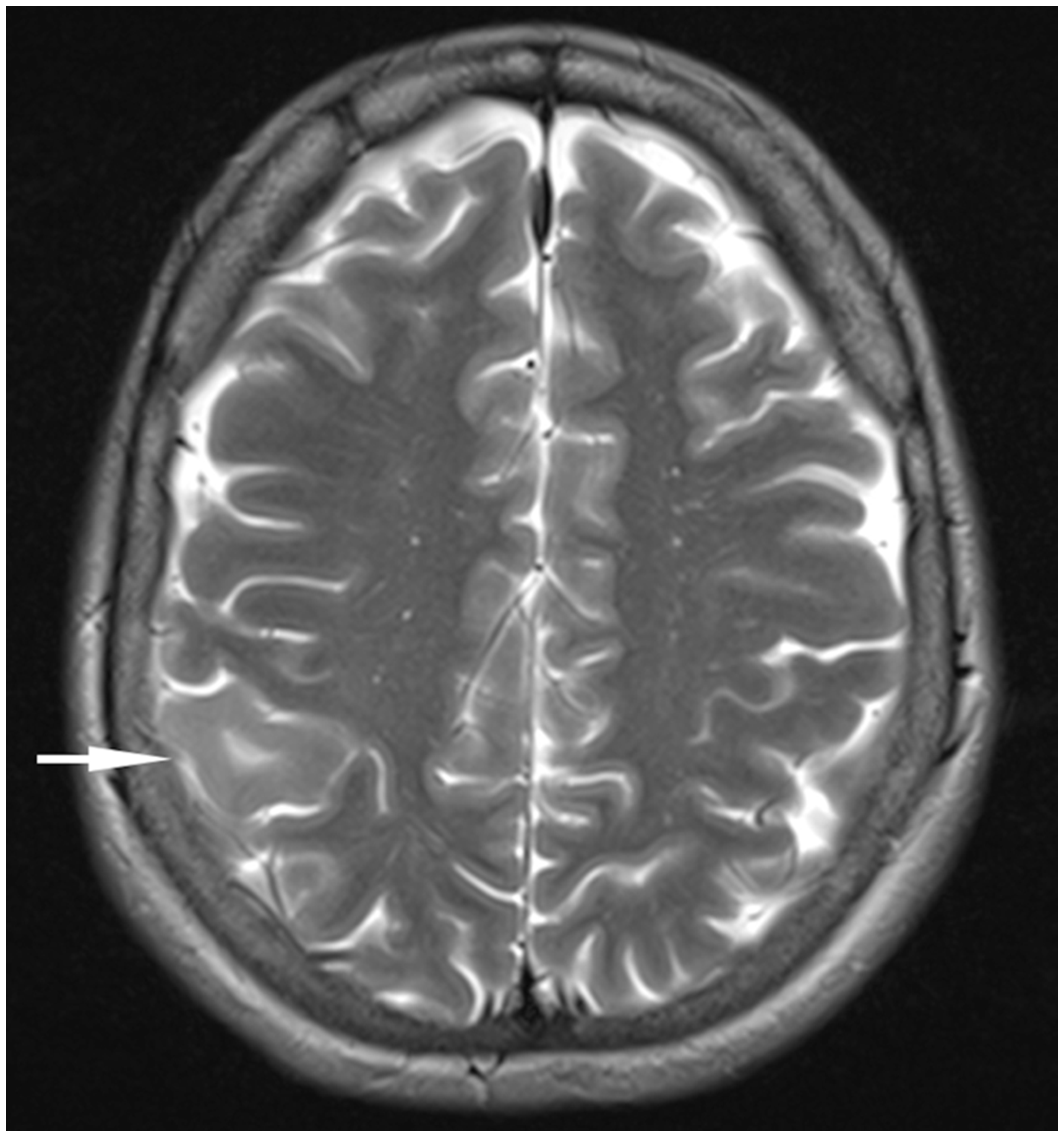
T2W MR image (axial plane) showing a right parieto-occipital cortical–subcortical lesion, with high signal intensity (arrow), consistent with a tuber.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Discussion
TS is a dominantly inherited disorder characterized by the growth of hamartomas in multiple organs. Renal AMLs are the most common urinary system manifestation of TS. Classically the disease is described as a clinical triad of sebaceous adenoma, mental retardation, and seizures. About 85% of patients with TS have central nervous system involvement: radial migration lines; cortical and subcortical tubers; sub-ependymal nodules; giant sub-ependymal astrocytomas; and retinal hamartomas.
A single cortical–subcortical tuber was identified in our patient in the right parieto-occipital side. Cortical–subcortical tubers are hamartomas, generally characterized by high signal intensity on T2W and low signal on T1W images and only in 10% of cases show contrast enhancement on T1W sequences. Gallagher et al. (7) classified these lesions in three classes according to the signal intensity of their subcortical white substance: type A = isointense on T1W images and hyperintense on T2W and fluid attenuated inversion recovery (FLAIR) sequences; type B = hypointense on T1W images and homogeneously hyperintense on T2 and FLAIR; type C = hypointense on T1W images, hyperintense on T2 but heterogeneous on FLAIR, showing a hypointense central region surrounded by a hyperintense rim. The tuber detected in our patient would therefore belong to type B. Cutaneous involvement is also very frequent: pigmentation defects of the skin and mucous membranes and nail fibrosis are often observed. Other manifestations include pulmonary lymphangioleiomyomatosis, associated with heart rhabdomyomas and hamartomatous tumors of the thyroid, liver, pancreas, lung, kidney, adrenals, and ovaries (8).
In patients with TS, AMLs are mainly found in women in the third and fourth decade of life and are usually very large, asymptomatic, multifocal, and bilateral. In sporadic cases, AMLs are encountered in women aged 40–70 years and are often symptomatic, single, and unilateral (9). AMLs in TS patients tend to have high female prevalence suggesting the possibility of hormonal influence (10). The diagnostic criteria of TS were finalized in 1998 by the conference promoted by the Tuberous Sclerosis Association and National Institutes of Health, followed by Consensus Conferences, the last in 2012 (11,12). The definitive diagnosis is based on the presentation of at least two major criteria or on one major criterion followed by two (or more than two) minor criteria. The possible diagnosis is granted if one major criterion or two or more minor criteria are present. A mutation in the TSC1 or TSC2 gene is sufficient for a definitive diagnosis, even if major and minor are totally absent.
Our patient presented two major criteria, multiple and bilateral renal AMLs and at least six cutaneous angiofibromas, associated with a single cortical–subcortical tuber. Multiple, bilateral renal AMLs are highly diagnostic of TS, according to the Gomez criteria on multifocality (11). Clinical diagnosis of TS is much easier when neurocutaneous disorders are present in early childhood, usually with childhood spasms. In a minority of cases, diagnosis appears in late childhood or adolescence, generally when the manifestations are predominantly dermatologic. The diagnosis in the adult life, as in our patient, is extremely unusual (13). The majority of AMLs appears as hyperechoic lesions on US due to fat content; in patients with TS, AMLs can be numerous and expand throughout the entire kidney with loss of normal corticomedullary differentiation (14).
Occasionally, AML have a low fat content and therefore they may appear relatively hypoechoic. In this case, they are very similar to renal cell carcinoma (RCC); therefore, differential diagnosis can be challenging. Furthermore, AMLs characterized by an irregular echogenicity due to the presence of intra-tumoral hemorrhage areas or necrosis, usually lead to misdiagnosis (15). A careful comparison of MR images with and without fat suppression is mandatory to formulate a more accurate diagnosis of low-fat AMLs. The loss of signal intensity in fat suppression sequences is diagnostic for AML. Alternatively, AMLs can also be strongly suspected using phase-opposed images that create a chemical shift of the second kind, localized at the interface between fat and non-fat components (16). The fat content can also be identified on unenhanced computed tomography as hypodense areas with Hounsfield Unit values below –10 (17). While clear cell RCCs frequently show both fat content and calcifications, the absence of the latter’s supports the diagnosis of AML, since calcifications within an AML are uncommon. In our patient, AMLs were intra-renal except for the largest one on the right, at the posterior upper pole, showing exophytic growth.
The most common complication of renal AML is Wunderlich’s syndrome, a rare urological emergency due to a spontaneous, non-traumatic bleeding in the sub-capsular or peri-renal space, which occurs in 10% of the cases. This condition must be timely suspected, diagnosed, and treated. It is characterized by the classic triad of symptoms: acute abdominal pain; palpable mass; and hypovolemic shock (18). The most common cause of non-traumatic renal bleeding is the AML (19). Yamakado et al. (20) claimed that there are significant relations between tumor size, formation and size of aneurysms, and their break in AML. Lesions > 4 cm, particularly in TS patients, are associated with a higher risk of bleeding and greater probability of requiring surgical or endovascular intervention (6). In our patient, the largest AML, located at the right, in the upper pole, measured 60 × 52 × 50 mm for a total volume of 68.4 mL, larger than the average renal AML previously described in literature. In addition, it contained a large hemorrhagic component within it, suggesting previous asymptomatic bleeding.
Finally, in our case there was no evidence of aneurysmatic dilation of the renal vessels on both color Doppler images and contrast-enhanced MRI. Invasion of the renal vein is a rare but well-known complication for benign AML, not associated with malignancy or metastasis development. Factors contributing to the development of renal vein invasion are the large size and central position of AML (21). The malignant transformation of AML, although extremely rare, has been documented. AML with many perivascular epithelial cells could be classified as an epithelioid AML. Malignant AML is considered the transformation of epithelioid AML (22) and in one-third of cases it has been associated with the development of metastasis (23) and death in 35–50% of the cases reported in literature (24). This variant has a surprising resemblance to RCCs.
Numerous criteria can be used to predict malignancy in renal AML including tumor size (25), necrosis (26), and prevalence of atypical mitotic figures (27). The final diagnosis of malignancy can only be confirmed on the basis of presence of metastasis (23,25). Asymptomatic AMLs with a diameter < 4 cm can be followed over time. For larger nodules, with uncontrolled bleeding or extended necrosis, or when a RCC has been diagnosed in the same kidney, total nephrectomy (28) is recommended. A moderate approach with angio-embolization or partial nephrectomy is preferable, especially in patients with small, bilateral, and multiple AMLs. Recently, cryotherapy and radiofrequency ablation have been suggested as alternative treatments.
In conclusion, we described an unusual case of TS, incidentally discovered in adulthood, in a patient with only mild abdominal pain and general good health conditions, despite the massive renal involvement by countless AMLs and the extent of the disease at the parieto-occipital cerebral level. To our knowledge, the present case is the first in literature characterized by such a large number of renal AMLs, with at least 20 on the right kidney and 40 on the left. In addition, this case provides an example of the extraordinary expression heterogeneity of this disorder, both for the age of presentation and severity of the disease.
Footnotes
Declarations of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
