Abstract
Bilateral pulmonary sequestration (PS) is a very rare congenital malformation. We describe a case of bilateral intralobar pulmonary sequestration (ILS) in a newborn. Both sequestrations received arterial supply from separate branches of the descending aorta and venous drainage was into ipsilateral inferior pulmonary veins. Prenatal ultrasonography showed cystic changes in the lungs. Computed tomography angiography (CTA) with supplemental two-dimensional (2D) and three-dimensional (3D) images was performed to clearly define the pathology and revealed bilateral intralobar pulmonary sequestration with aberrant blood supply. The child underwent successful video-assisted thoracoscopic surgical (VATS) lobectomy on the left side and thoracoscopic wedge resection on the right side. There were no complications. CTA with supplemental 2D and 3D images plays a vital role in revealing the exact pathology in congenital pulmonary malformations associated with anomalous vasculature.
Keywords
Introduction
Pulmonary sequestration (PS) refers to a rare congenital anomaly characterized by dysplastic and non-functioning pulmonary tissue receiving arterial supply from the systemic vessels. There is no connection to the normal tracheobronchial tree or the pulmonary arteries (1). PS comprises about 0.15–6.45% of all congenital bronchopulmonary malformations and is classified into intralobar sequestration (ILS) and extralobar sequestration (ELS). ILS is found in the normal lung tissue without its own pleura, while ELS has its own pleural lining. Most of the cases of PS are usually unilateral and bilateral PS are very uncommon (2,3). In a review of bilateral PS, nine out of a total 17 cases were bilateral ILS (3).
We describe a case of bilateral ILS. A newborn male infant was admitted to our hospital to reassess cystic changes in the lungs seen on the prenatal ultrasonography. Further studies confirmed the diagnosis of bilateral intralobar PS. Video-assisted thoracoscopic surgical (VATS) lobectomy using a standardized anterior approach (4) was performed initially on the left side when the child was aged 18 months. At 23 months, resection of right-sided sequestered lung tissue was performed using the VATS technique.
Case report
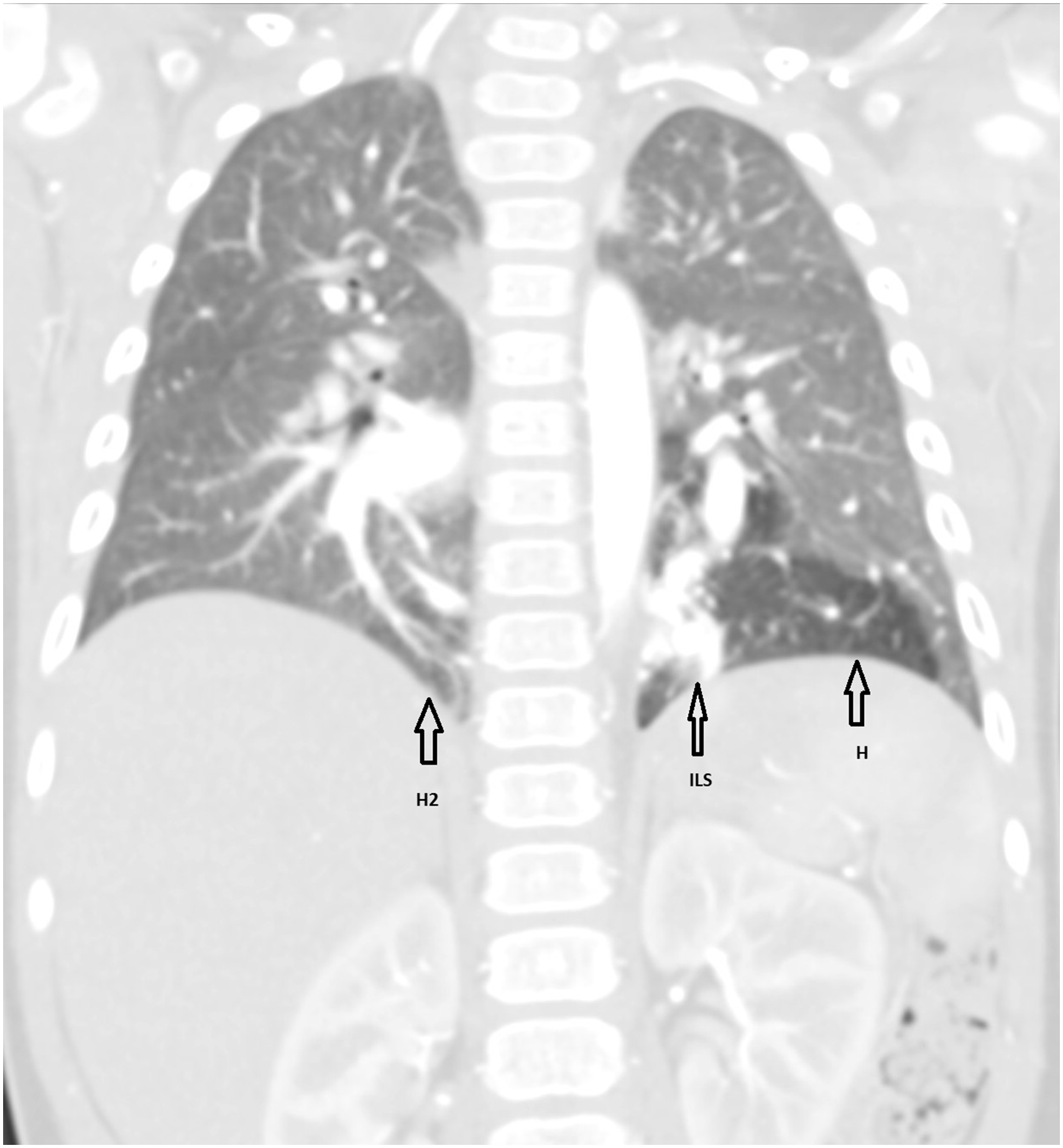
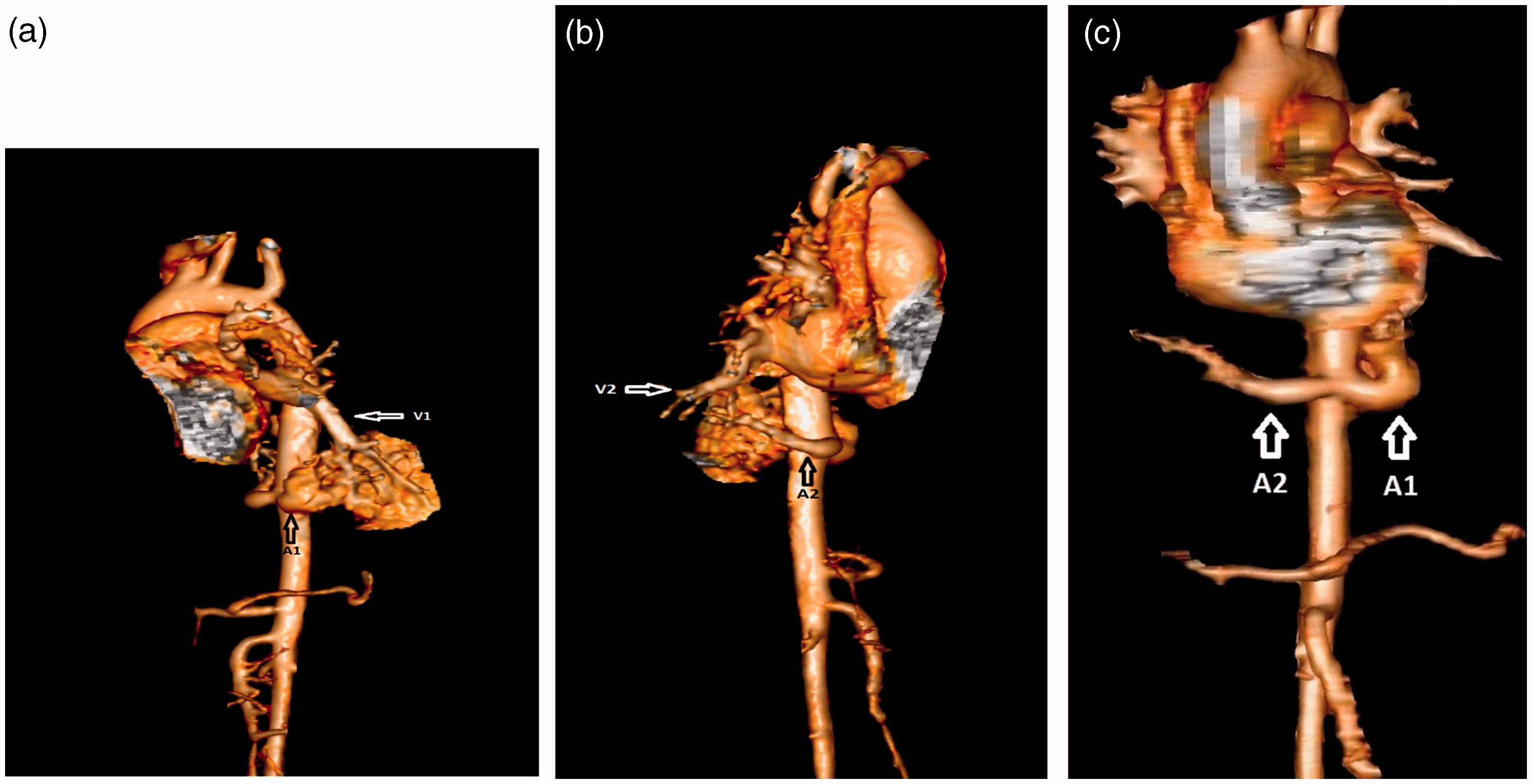
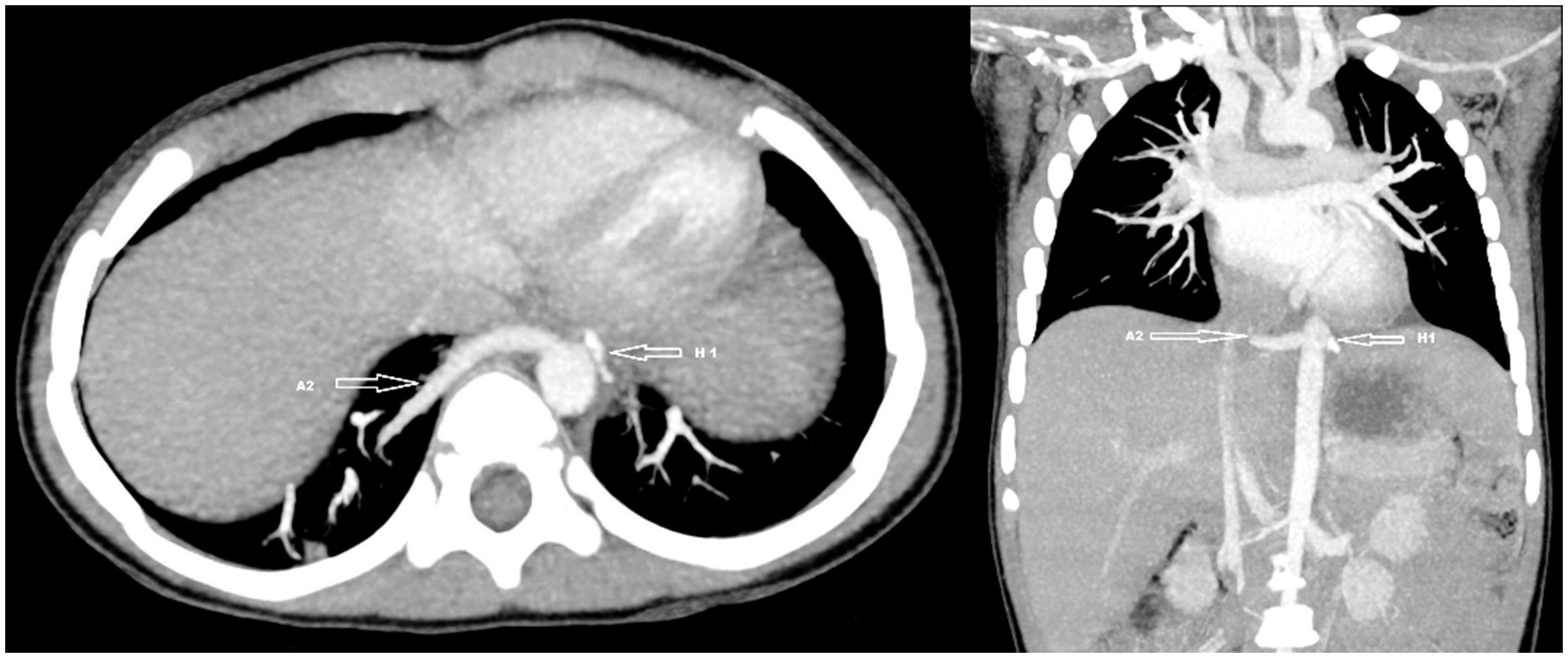
A newborn male infant was referred to our hospital with prenatal diagnosis of cystic lesions in the lungs. A chest X-ray (Fig. 1) taken immediately after birth showed increased density behind the heart but the finding was non-specific. To accurately define the pathology, computed tomography angiography (CTA) of the thorax and upper abdomen was performed at the age of 11 months. Because of increased risk of conversion of VATS lobectomy to thoracotomy in low-weight children (5), the CTA was performed when the child gained a weight of 12 kg. A multidetector Toshiba Aquilion one vision edition CT scanner was used to perform the CTA using 18 mL i.v. contrast and a scan delay of 12 s. The CTA was performed in Rigshospitalet, Copenhagen, Denmark. To increase the diagnostic value, the CTA was supplemented by two-dimensional (2D) and three-dimensional (3D) images as suggested by Lee et al (6). The CTA established the final diagnosis of bilateral ILS. A 3.5 × 1.1 cm ILS was found located paravertebrally in the left inferior lobe receiving its arterial supply from a large branch of the descending aorta (Figs. 2, 4a, and 4c). Surrounding the sequestration was a hyperinflated area of approximately 4.8 cm (Fig. 3). In the right inferior lobe there was a relatively smaller ILS with the blood supply from a separate branch of the descending aorta (Figs. 2, 4b, and 4c). The right-sided ILS included an area of hyperinflation but no definite opacity was visible on right side (Fig. 3). Venous drainage from both sequestrations was via ipsilateral inferior pulmonary veins (Fig. 4a and 4b). No further pathology was found. CTA with supplemental 2D and 3D images clearly revealed these anomalies and helped to plan further treatment. The patient was asymptomatic; however, due to increased risk of future infections and malignant transformation related to bronchopulmonary foregut anomalies (2), it was decided to excise the bilateral PS prophylactically. At first, left-sided inferior lobe VATS lobectomy was performed at the age of 18 months because it was the most affected side. There were no complications after the surgery. A follow-up CT scan (Fig. 5) was performed three months postoperatively which showed no complications in the left side while right-sided ILS was unchanged. The follow-up CT scan was performed to ensure that minimal lung resection was still possible on the right lower lobe. About five months after the left sided VATS lobectomy, VATS wedge resection of the right-sided sequestered lung tissue was performed without complications.
Chest X-ray shows increase in density behind the heart. CTA showing two aberrant arteries A1 and A2 arising from aorta and supplying ILS on the left and right sides, respectively. CTA showing left-sided ILS surrounded by an area of hyperinflation (H). There is a hyperinflated area (H2) in the right lower lung lobe. There is no visible opacity on the right side. (a) 3D image showing venous drainage of left-sided ILS into ipsilateral inferior pulmonary vein (V1). A1 represents the aberrant arterial supply of left-sided ILS. (b) 3D image showing venous drainage of right-sided ILS into ipsilateral inferior pulmonary vein V2. A2 represents the aberrant arterial supply of right-sided ILS. (c) Frontal view of 3D image showing two aberrant arteries, A1 and A2, arising from the aorta and supplying ILS on the left and right sides, respectively. Follow-up CT scan showing insertion of hemolock (H1) after left-sided VATS lobectomy. There were no complications on the left side. A2 represents the feeding artery to the right-sided ILS.

Pathological examination of the left lower lobe revealed a large feeding artery with no accompanying vein (Fig. 6a). The veins were located in the interlobular septa and drained into a pulmonary vein in the hilar area (Fig. 6b). On the right side, a large feeding artery was identified. The veins were located in the septa and drained into a pulmonary vein in the hilar aspect of the specimen (Fig. 6c).
(a) Pathological examination of the left lower lobe shows a cross-section of the large systemic feeding artery. (b) Pathological examination of the left lower lobe shows a section of the parenchyma with the hyperinflated area in the bottom on right side. The veins (arrows) are seen in the septa. (c) Pathological examination of right-sided resected lung. Section of the parenchyma with the large systemic feeding artery (large arrows) and the pulmonary veins in the septa (small arrows).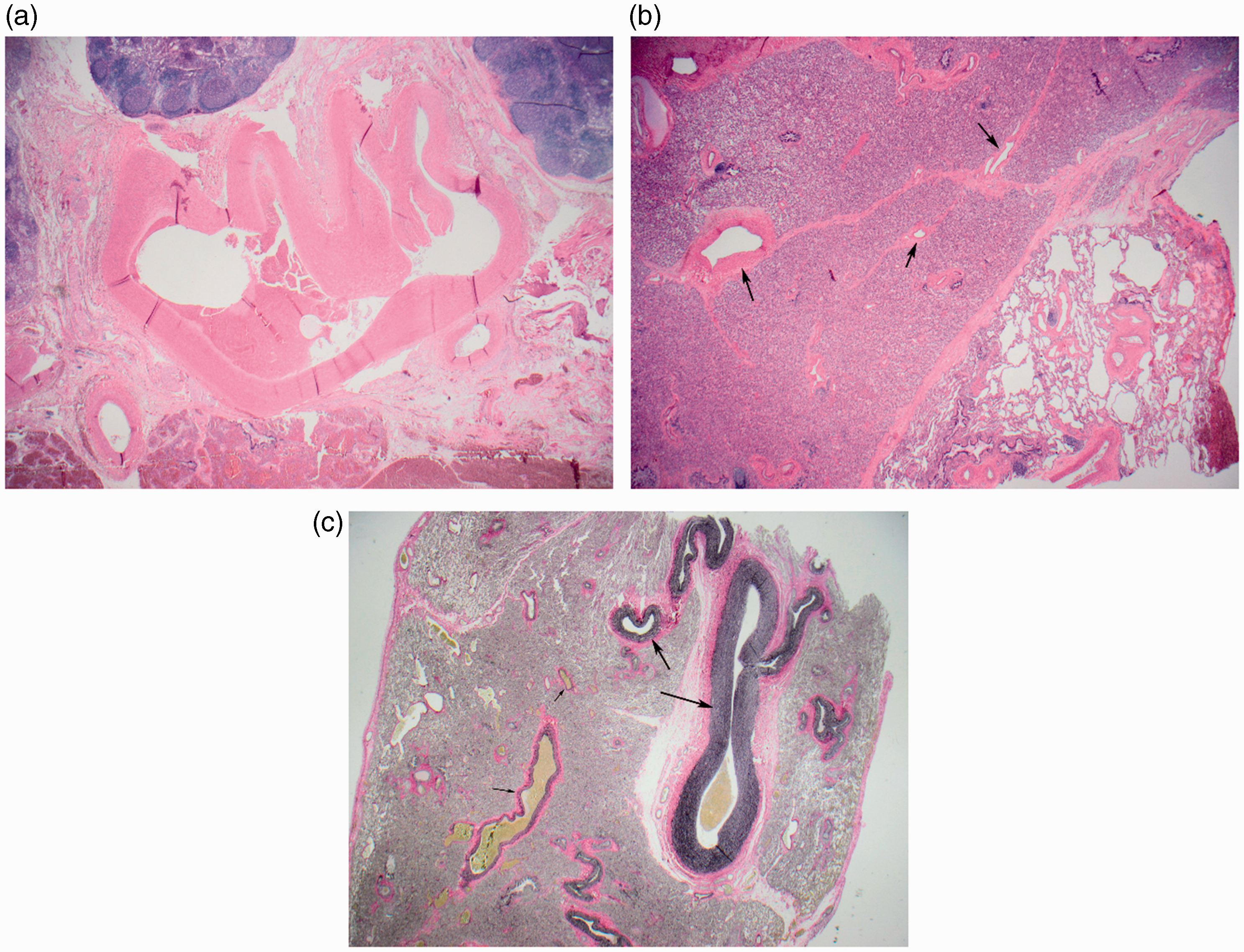
The pathology of resected tissue on both sides showed chronic inflammation, atelectasis, and bronchial obstruction. These changes were compatible with ILS.
Discussion
Incidence of ILS is about 75% of all PS (7). ILS is mostly found in the posteromedial part of the lower lung lobes and occurs mainly in the left lung (8). As in our case, bilateral PS are very uncommon (2,3). Arterial supply in both types of PS is derived from the aorta or its branches. ILS is usually drained via pulmonary veins while venous drainage of ELS is usually via systemic veins (1). Repeated lung infections, congestive heart failure, hemoptysis, and malignant transformation are complications of PS. Because of partial sequestration with a connection to bronchial ventilation or secondary ventilation from adjacent tissues, ILS has high risk of infections (2). On the other hand, ELS has a lower risk of respiratory infections (2), but there is a risk of torsion of its vascular trunk with subsequent infarction (9). Ten pediatric patients with hemorrhagic infarction of ELS have been described (9). Once complications arise, removal of large pulmonary tissue may be needed. Consequently, surgical excision is accepted as the definitive treatment even in asymptomatic patients (2). Recognition of the aberrant blood supply of the sequestered lung is vital in ultimate diagnosis and planning of surgical treatment (1,6).
Previously, arteriography was used to identify abnormal vessels associated to PS, but recent advances in CT technology offer less-invasive techniques to evaluate the pathology of PS (7,8). Other possible non-invasive technique for diagnosis of PS is magnetic resonance imaging (MRI) and magnetic resonance angiography (MRA). Long imaging time, sedation for children, and artifacts caused by respiratory movements are the disadvantages of MRI (8). Moreover, MRI is costly and is not easily available (7). Radiation exposure is a drawback of multidetector computed tomography (MDCT) (8). In our case, the radiation dose used in the first CTA was 71.6 mGycm and 77.4 mGycm in the follow-up CT scan. Thus, both values were within the European Diagnostic Reference Levels (DRLs) of 80 mGycm (10).
In our case described in this report, CTA with supplemental 2D and 3D images successfully revealed the aberrant vascular supply and changes in the bronchopulmonary anatomy. 2D and 3D images added to the diagnostic value of CTA by clearly delineating anatomical relationships especially for vessels. Lee et al (6) compared the diagnostic accuracy of axial MDCT images, multiplanar MDCT images, and 3D MDCT images in detecting the anomalous vessels associated with congenital lung malformations. The results showed that the diagnostic accuracy of identifying anomalous veins associated with congenital lung malformations was 60% with axial MDCT compared to 80% with multiplanar MDCT images and 100% with 3D MDCT images. They suggested that supplemental multiplanar and 3D MDCT images should be considered routinely in the evaluation of patients with congenital lung malformations associated with anomalous veins as they enhance the diagnostic accuracy (6). The evaluation of aberrant blood supply is also useful in planning the surgical treatment of congenital lung anomalies and can prevent massive hemorrhage which otherwise can result from accidental division of an undetected anomalous vessel (6).
In conclusion, 2D and 3D images substantially increase the diagnostic value of CTA for the assessment of congenital lung diseases associated with anomalous vasculature, e.g. PS, and is the investigation of choice for the diagnosis and preoperative assessment of these patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
