Abstract
We report a case of combined hepatocellular-cholangiocarcinoma with stem cell features, cholangiolocellular subtype arising about 15 years after placement of an inferior vena cava stent for primary Budd–Chiari syndrome. Pre-surgical differentiation of the tumor from hepatocellular carcinoma was difficult because of elevated levels of alpha-fetoprotein and hypervascularity in the arterial phase. Histopathological examination revealed atypical cells forming ductal and alveolar structures showing a vague border with the surrounding liver. Immunostaining showed positive results for epithelial membrane antigen, mainly localized to the apical surface of the tubules, representing a characteristic finding for combined hepatocellular-cholangiocarcinoma with stem cell features, cholangiolocellular subtype. Combined hepatocellular-cholangiocarcinoma with stem cell features arising in the liver with hepatic damage from Budd–Chiari syndrome is not common, but diagnosis is important to manage the malignancy, which shows different clinical behaviors from hepatocellular carcinoma.
Keywords
Introduction
Budd–Chiari syndrome (BCS) is an uncommon condition caused by hepatic venous outflow tract obstruction and showing various symptoms through hepatic vein congestion. BCS can be subdivided into two different types: primary BCS is caused by hepatic vein stenosis due to thrombotic obstruction or phlebitis of a hepatic vein, and the secondary syndrome is due to compression of the hepatic vein by outside structures such as tumors, cysts, or abscesses (1). The occurrence of primary hepatic nodules other than hepatocellular carcinoma (HCC) and nodular regenerative hyperplasia (NRH) related to BCS is very rare, with only a few cases reported in the literature. Most such cases involved secondary BCS caused by intrahepatic cholangiocarcinoma (CC) invading the hepatic veins. However, the differentiation of hepatic nodules is so important that they drastically change the prognosis of the patient from the point of clinical management. Here, we report that combined hepatocellular-cholangiocarcinoma (C-HCC-CC), cholangiolocellular subtype developed in a patient after inferior vena cava stent (IVC) placement for BCS syndrome.
Case report
A 56-year-old woman came to our hospital because of epigastralgia and hepatomegaly about 15 years before developing a hepatic tumor. Enhanced computed tomography (CT) and angiography revealed hepatic vein and inferior vena cava (IVC) stenosis at the level of confluence, so she was diagnosed as BSC from congenital angiodysplasia or previous phlebitis and received IVC stent placement in our institution. Acetylsalicylic acid (100 mg/day) was prescribed after stent placement. Enhanced or unenhanced CT was performed once a year during her routine visit to our hospital for follow-up. The result of the hepatitis B and C virus test remained negative. There were no past history of drinking alcohol or chronic liver disease and no family history of hepatobiliary malignancy. Contrast-enhanced CT 13 years after stent placement showed a spot-like enhancement about 5 mm in diameter in the peripheral region of segment 8 of the liver (Fig. 1). The alpha-fetoprotein (AFP) level was below the reference value at that time. This enhancement was suspected to represent a small portal venous shunt or hepatic tumor such as HCC. At the time of CT, she showed an allergic reaction to the contrast material and developed facial edema. Intramuscular injection of epinephrine and an antihistamine drug resolved the symptom within hours. After the allergic episode, unenhanced CT and ultrasonography were performed for annual follow-ups of the IVC stent and nodular enhancement in the liver.
Contrast-enhanced CT findings 13 years after stent placement. The stent was inserted in IVC at the level of hepatic-vein confluence, and there was a dot-like enhancement in the peripheral parenchyma of liver (arrow).
The hepatic lesion had increased to 1.6 cm in diameter on ultrasonography in the subsequent 2 years. In addition, laboratory data showed elevated AFP (70 ng/mL) and lens culinaris agglutinin-reactive fraction of AFP (AFP-L3, 85.5%), although data had been normal until then. Because growth of the hepatic tumor was strongly suspected from those findings, the primary physician decided on further imaging. The IVC stent was a handmade stainless-steel device and significant artifacts were anticipated on magnetic resonance imaging (MRI), so dynamic CT with contrast material under steroid preparation was performed for diagnosis. Dynamic CT revealed a well-circumscribed, low-density area (mean CT value, 42 Hounsfield unit [HU]) without pseudocapsule in the peripheral liver (Fig. 2a). The nodule showed strong homogeneous enhancement (mean CT value, 145 HU) in the arterial phase (Fig. 2b). Enhancement was prolonged to the portal phase and decreased to almost the same level as surrounding hepatic parenchyma in the equilibrium phase (mean CT values, 169 and 131 HU; Fig. 2c and d, respectively). Based on these laboratory data, imaging findings, and the prevalence rate of HCC for BCS, the preoperative diagnosis of the lesion was HCC, clinical stage T1N0M0 in the union for international cancer control (UICC) staging system. Partial hepatic resection was performed for the tumor.
Dynamic CT findings 2 years later from the time of CT shown in Fig. 1. (a) Unenhanced CT shows low-density nodule in the peripheral region of the liver (arrow). (b) The nodule showed strong enhancement in the arterial phase. (c, d) Enhancement was prolonged to the portal phase and decreased to almost the same level as surrounding hepatic parenchyma in the equilibrium phase.
Intraoperative ultrasonography was performed to define the cutting line of hepatic parenchyma, and resection was carried out under intermittent interruption of blood flow to liver with a cavitron ultrasonic surgical aspirator and bipolar electrosurgical unit. Intraoperative findings suggested slight fibrotic change to the background hepatic parenchyma. The tumor was completely resected with sufficient surgical margins. No complications were encountered intra- and perioperatively and the patient was discharged 10 days postoperatively. No sign of recurrence was seen during the follow-up and AFP was returned to normal level.
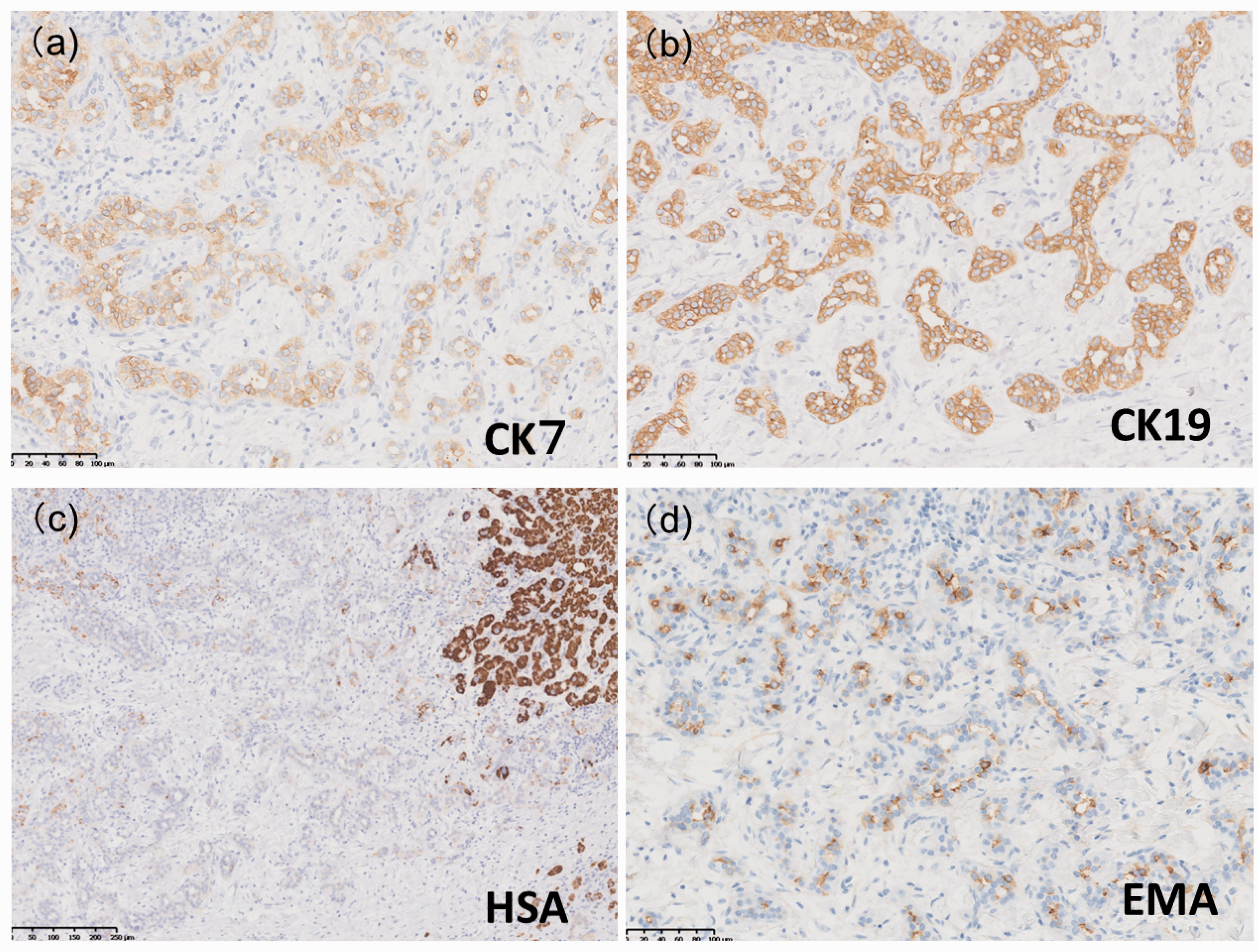
Histopathological findings revealed a tumor comprising atypical cells with small, round nuclei and eosinophilic cytoplasm, forming ductal and alveolar structures (Fig. 3). No mucin production was observed. The tumor showed a replacing growth pattern without a hyalinized capsule and the border with the surrounding liver was vague. On immunostaining, the atypical cells were partially positive for hepatocyte specific antigen (HSA) and positive for cytokeratin (CK) 7 and CK19 (Fig. 4). Epithelial membrane antigen (EMA) was strongly positive in the tumor, mainly localized to the apical surface of the tubules. The tumor also has the component partially positive for AFP. The final diagnosis was C-HCC-CC with stem cell features, cholangiolocellular subtype. Inflammatory changes and slight fibrosis without distortion were observed in the surrounding hepatic parenchyma around the portal area. The liver damage was classified as A2F2 according to the new Inuyama classification, which was compatible with BCS-related hepatic damage. The patient had no other chronic hepatic disease, so the damage was supposed to be caused by congestion of hepatic vein even after IVC stent placement at the level of hepatic vein.
Histological findings of resected tumor with H&E staining. (a) The tumor comprising atypical cells with small, round nuclei and eosinophilic cytoplasm, forming ductal and alveolar structures. (b) The tumor showed a replacing growth pattern without a hyalinized capsule on the border with the surrounding liver (arrow). The result of immunostaining examination.
Discussion
In this case report, we described a case of C-HCC-CC with stem cell features, cholangiolocellular subtype during follow-up for BCS. The tumor showed early enhancement in the arterial phase and no wash-out in the equilibrium phase. The finding of “wash-out” is very important to decrease the false positive by early enhancement in the diagnosis of HCC (2). We made a diagnosis of HCC depending on the finding of arterial phase and the tumor prevalence rate; although the possibility of CC component with fibrous stroma might be listed in the differential diagnosis for a small tumor without wash-out. Several reports have indicated that hypervascular nodules like HCC and NRH could develop during the clinical course of BCS (3–7). We also considered the possibility of other primary hepatic tumors, such as CC and C-HCC-CC, among the differential diagnoses. The typical CT findings of CC, the second most common primary hepatic tumor, is ring enhancement in the arterial phase, although small CCs sometimes show strong homogeneous enhancement difficult to differentiate from HCC (8). However, the elevation of serum AFP and AFP-L3 levels in this case also suggested HCC. C-HCC-CC is a comparably rare hepatic tumor characterized by strong enhancement in the arterial phase due to the HCC component (9–11). We therefore considered HCC as the most probable diagnosis and performed partial resection of the liver. Histopathological examination led to a final diagnosis of C-HCC-CC with stem cell features (cholangiolocellular subtype) and radical excision of the tumor was performed.
The distinction of BCS-related HCC from NRH is clinically important in the setting of BCS follow-up, because HCC drastically changes the management and prognosis of the patient. The influence of BCS on the carcinogenesis of HCC remains controversial. The prevalence of HCC in BCS patients has been reported as 25–48%, with a moderate range of differences between reports and regions (4). The rate of hepatitis virus infection complicating BCS is presumed to be responsible for these differences. Shin et al. extrapolated different carcinogenic processes between BCS and hepatitis-related HCC from the evidence of clinical differences such as tumor invasiveness and prognosis, leading to a positive acknowledgment that BCS-related HCC does exist clinically (3). On the other hand, the etiology of NRH is considered to involve an abnormality of blood flow; more specifically, obstruction of the venous drainage leading to atrophy of the hepatic parenchyma under the influence of venous congestion and hypertrophy of the region less influenced by the obstruction. The emergence of BCS-related HCC is mainly indicated by imaging findings and the level of tumor markers. Increases in AFP and AFP-L3 levels appear reliable for screening. Typical imaging findings of HCC with BSC are the same as for HCC of other etiologies. HCC tends to be heterogeneous, with a pseudocapsule and a larger lesion than NRH. HCC has an enhancing pattern showing early arterial enhancement with delayed wash-out on dynamic CT and MRI. On MRI, HCC appears hypointense on T1-weighted (T1W) imaging and slightly hyperintense on T2-weighted (T2W) imaging, with high intensity on diffusion-weighted imaging and shows no uptake of gadolinium-ethoxybenzyl-pentetic acid (4,5). NRH tends to show multiple homogeneous lesions in peripheral areas of the liver, sometimes accompanied by central scarring (6).
Secondary BSC was reported to occur as a complication of primary hepatic tumor containing a CC component, with only five cases reported in the English and Japanese literature to date (12–16). Four of the five cases were about CC and the remaining case involved double cancer with multiple HCCs and CC, where the CC component displayed invasion into the IVC. Of note, all those reports showed that the tumor invaded into the hepatic vein, with eventual obstruction of the IVC progressing to tumor-related secondary BCS. No previous reports have described hepatic tumor containing a CC or C-HCC-CC component during follow-up of a patient with BCS. Our case represents the first report of C-HCC-CC with stem cell features in a patient with BCS, which has the possibility of complications due to BCS-related hepatic damage.
C-HCC-CC with stem cell features, cholangiolocellular subtype is a hepatic tumor composed of malignant cells differentiating to cholangioles and duct of Hering with a multilineage potential (17,18). This entity was previously classified as a kind of intrahepatic cholangiocellular carcinoma, although it was defined as one variant of C-HCC-CC in the World Health Organization classification 2010 (19). C-HCC-CC is divided into two types: the classical type and the type with stem cell features. The latter has the component of small cells with high nucleus to cytoplasma ratio, which have the morphological immunostaining profile of stem/progenitor cell. Furthermore, C-HCC-CC with stem cell features is divided into three subtypes: typical subtype; intermediate-cell subtype; and cholangiolocellular subtype. C-HCC-CC with stem cell features, cholangiolocellular subtype is an extremely rare hepatic tumor (1%) arising in patients with hepatic damage (20). Because of this rarity, the causal relationship between the tumor and BCS has not been sufficiently researched. The carcinogenic process of C-HCC-CC with stem cell features under a BCS setting remains unclear and warrants further study. However, under the existing conditions, histological background of the malignancy may affect the treatment process and chemotherapy regimen. Transcatheter arterial chemoembolization and radiofrequency ablation can be applied as non-surgical treatment options for HCC but are not a standard treatment for tumors with a CC component. From this perspective, pathological diagnosis with biopsy or surgery is essential for clinical management of BCS-related hepatic tumor when the laboratory and imaging findings of the tumor are atypical for HCC and NRH.
In conclusion, we encountered a case of C-HCC-CC with stem cell features, cholangiolocellular subtype, during follow-up. Although HCC and NRH commonly appear in patients with BCS, the diagnosis of primary hepatic tumor related to BCS should be made with care including the possibility of CC and C-HCC-CC. Furthermore, pathological examinations are recommended for glowing tumor with atypical radiological finding because hepatic tumor with CC component leads drastic change to patient treatment and follow-up.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
