Abstract
The incidence and severity of methicillin resistant Staphylococcus aureus (MRSA) infections are increasing and cause high mortality and morbidity. We describe the first pediatric case in Scandinavia with Panton-Valentine leucocidin (PVL) positive MRSA septicemia who developed bilateral pneumonia, arthritis of the knee, and osteomyelitis of the tibia. Radiological investigation and interpretation directed the treatment, especially the surgical debridement, and combined with clinical and biochemical findings lead to close interdisciplinary treatment with frequent surgical interventions and antimicrobial combination therapy. The outcome was a healthy patient without sequelae, a favorable course unlike those previously described in the literature. This case underlines the necessity of a close interdisciplinary cooperation in children with severe MRSA infection encompassing pneumonia, septic arthritis, and osteomyelitis, using different imaging modalities to guide the surgical and antibiotic treatment.
Introduction
Osteomyelitis and septic arthritis are common in pediatrics (1) and Staphylococcus aureus is the main cause of acute hematogenous osteomyelitis in healthy children. Over the last decade the occurrence of methicillin resistant S. aureus (MRSA) has increased in the community (2,3). An increase in incidence and severity of acute hematogenous osteomyelitis and septic arthritis has been observed, especially in cases concerning MRSA (1). It is known that MRSA strains have different virulence factors that can make the infections difficult to treat, e.g. Panton-Valentine leucocidin (PVL) (3,4). PVL also increases the risk of developing necrotizing pneumonia (5,6). Only few of the published pediatric osseous infections with MRSA septicemia and pneumonia have a favorable outcome and death is often the consequence (7–9). MRSA is a common cause of bone and soft-tissue infections worldwide (2,3,5,10,11). However, osseous MRSA infections are still uncommon in Scandinavia. To our knowledge, the current patient is the first described case of PVL-positive MRSA septicemia with bilateral pneumonia, osteomyelitis, and septic arthritis in a Scandinavian child and, furthermore, the first to emphasize the significance of using different imaging modalities when directing and planning the cause of treatment.
Case report
A 13-year-old healthy and athletic boy presented with fever (39.1℃) and a 1-day history of limping and left-sided knee pain. The knee was without visible changes, but the patient had pain and tenderness at the infrapatellar ligament accentuated by passive flection. Radiography of the knee and leg was normal without signs of joint effusion. Leucocytes and C-reactive protein (CRP) were elevated, being 16.8 × 109/L (normal range, 4.40–10.5 × 109) and 170.2 mg/L (normal range of CRP < 8), respectively. The boy was sent home. The following day the patient continued to be febrile and was admitted to the pediatric department on suspicion of soft tissue infection surrounding the knee. An ultrasound of the knee was described as normal. On day 3, staphylococci were cultured from blood and intravenous (IV) vancomycin treatment was initiated, as a sibling was a carrier of MRSA. At this time, his knee was red and swollen and cutaneous micro-ulcerations had supervened. Magnetic resonance imaging (MRI) showed synovitis with knee joint effusion and signs of medullary osteomyelitis in the proximal tibia accompanied by a subperiostal fluid accumulation (Fig. 1a–c). CRP was 430 mg/L and sedimentation rate (SR) 49 mm/h (normal range, 2–15 mm/h). An acute arthroscopy with synovectomy and lavage of the knee joint was performed in the orthopedic ward. Cultures from the medullary canal, as well as cultures from blood, nasal swaps, and biopsy materials from the knee all grew MRSA. The strain was positive for PVL.
(a) Initial MRI, coronal T1 (left image) and axial proton fat-saturated images of the knee show irregular bone marrow signal intensity in the tibial metaphysis consistent with edematous changes (arrow on left-sided image) in addition to intraarticular fluid collection (black arrow on right-sided image) and fluid collection in the surrounding soft tissue (white arrows). (b, c) Supplementary STIR (left images) and post-contrast T1 fat-saturated images, coronal slice of the whole leg (b) and axial slices (c) corresponding to the metaphysis show a not enhancing fluid collection subperiostally (white arrows) compatible with abscess formation in addition to pronounced soft tissue edema and a small not enhancing soft tissue collection (black arrow on (b)).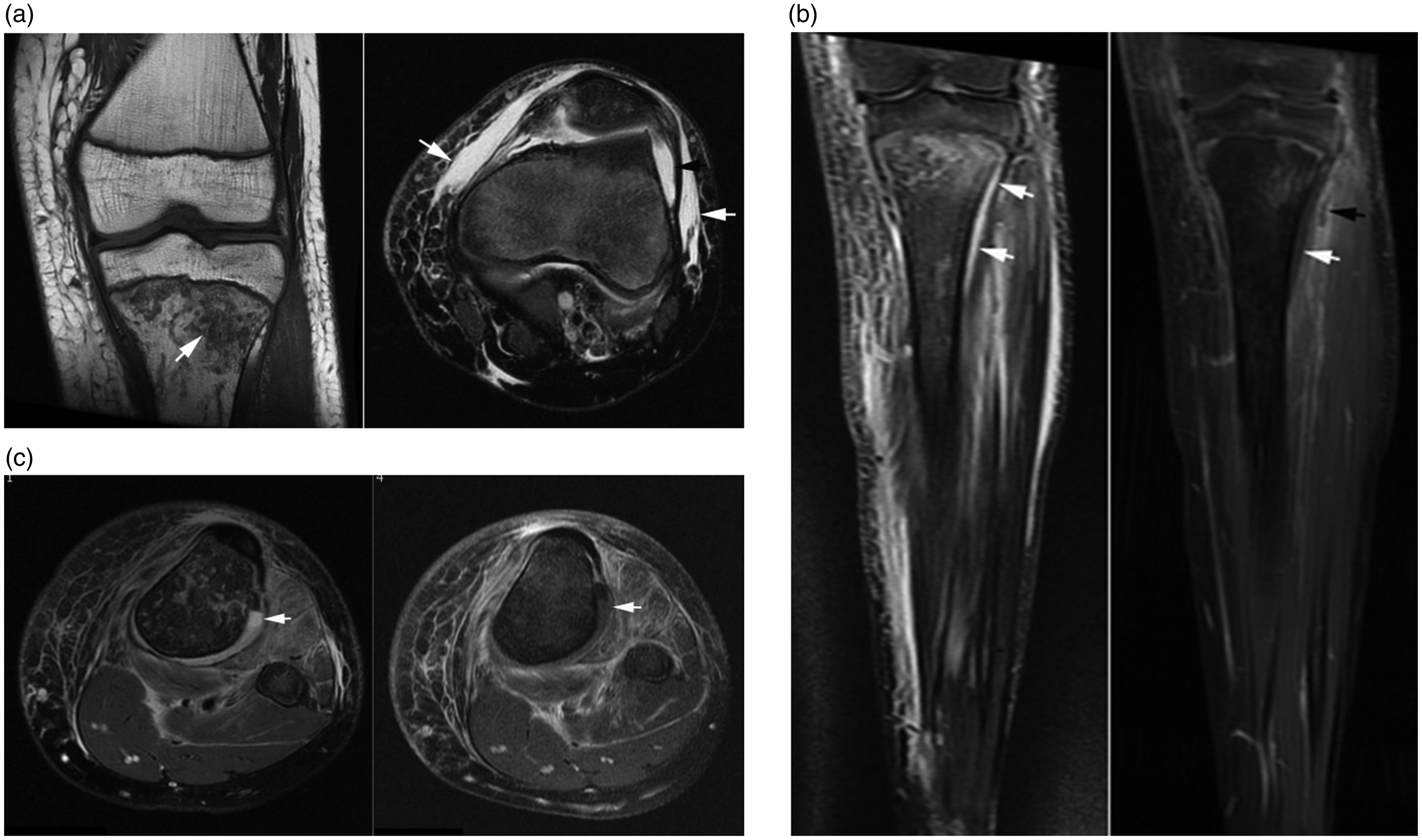
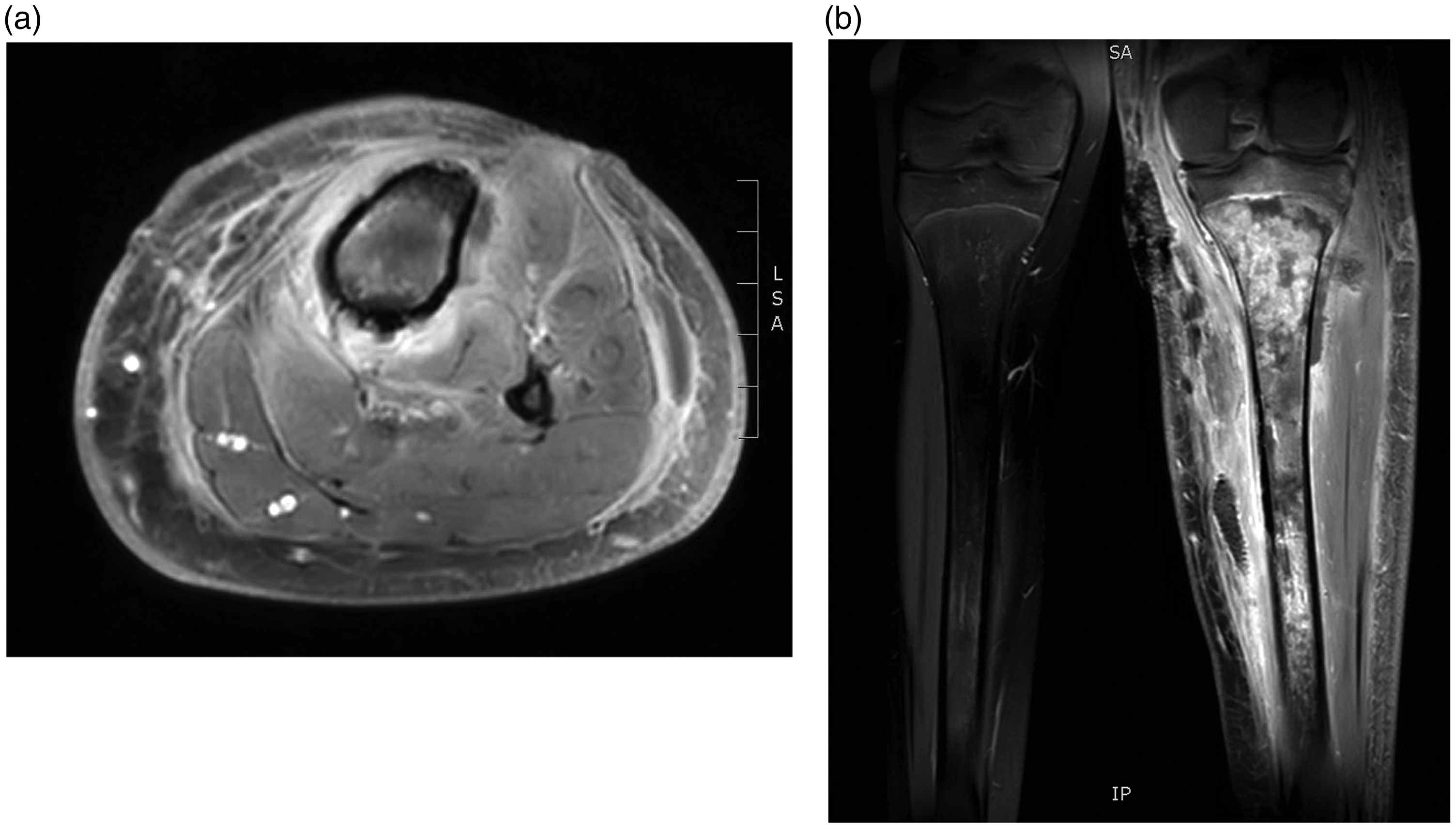
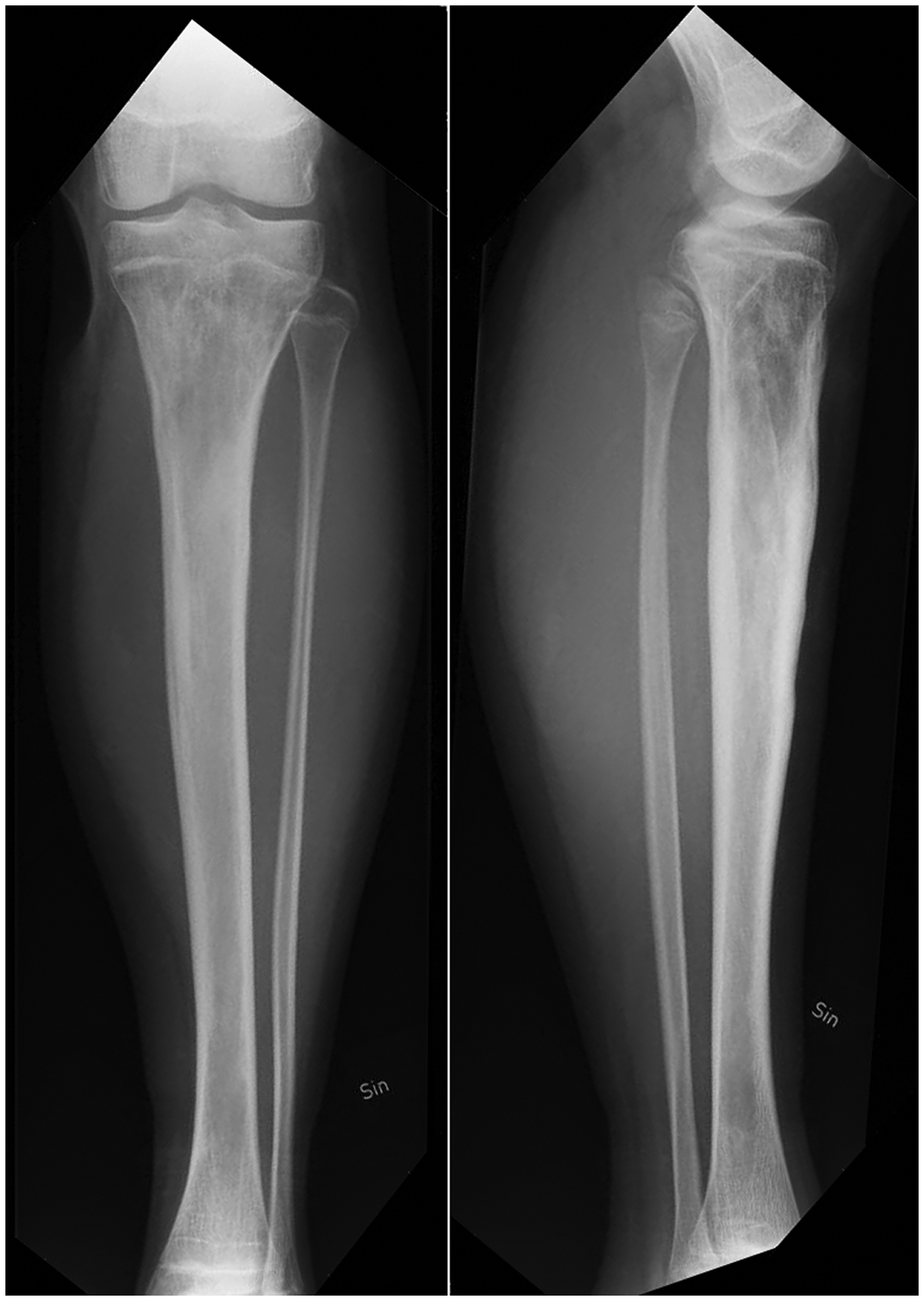
Although wounds were debrided every other day and the patient was treated with IV vancomycin, his pain increased, and CRP and leucocytes continued to rise. On day 7, MRI showed progression of the infection in the medullary canal leading to renewed surgical debridement. Despite surgery and IV treatment, the patient’s condition deteriorated. He became septic and developed multiple bilateral pulmonary infiltrates (Fig. 2). Due to respiratory distress, he was admitted to the intensive care unit (ICU), and on suspicion of necrotizing pneumonia, oral linezolid was added to the antimicrobial treatment. After 4 days in the ICU, the patient’s general condition improved and he returned to the orthopedic ward. Clinical and biochemical improvement continued during the next weeks on relevant antimicrobial therapy, but after approximately 3 weeks the CRP rose again. MRI showed a new subperiostal abscess and progression of the osteomyelitis with edema and enhancement in the entire medullary canal and with cortical destruction of the tibia, despite prolonged and intensive treatment (Fig. 3a and b). After renewed surgery, the patient’s condition improved both clinically and paraclinically and he was discharged after 49 days of hospitalization, having undergone several different imaging modalities (Table 1) leading to multiple surgical interventions and 7 weeks of IV antibiotics. After discharge, the patient was followed in the outpatient clinic with frequent clinical, biochemical, and radiological controls. Although the patient was in good condition and free of pain, the osteomyelitis progressed radiologically for several months (Fig. 4) leading to a follow-up MRI supplemented by computer tomography (CT) and finally both a bone and leukocyte scintigraphy (Fig. 5) concluding with no active infection. Radiological signs eventually subsided and after 22 months, plain radiography showed a normal tibia (Fig. 6). In total, the patient’s fever persisted for 51 days and CRP and SR were elevated for 23 weeks.
Chest X-ray shows bilateral pronounced pulmonary infiltrates with accompanying left-sided pleural effusion. (a, b) Osteomyelitis of the tibial diaphysis with abscesses and destruction of the diaphysis. Overview of the imaging examinations, their diagnostic purpose, and the change of treatment. Radiography showing irregular osseous structure in the proximal tibia with a mixture of lytic and sclerotic areas in addition to periosteal new bone formation. Sequestration cannot be excluded. Bone (upper image) and leucocyte scintigraphy show tracer uptake in the entire tibia and no sign of pathological leucocyte accumulation (lower image). X-ray of the left leg 22 months after the initial therapy showing nearly normal conditions.



Discussion
Osteomyelitis and septic arthritis are common in pediatrics (1). However, cases concerning MRSA are still uncommon in Scandinavia and worldwide only few cases with septicemia and severe pneumonia have been described with a favorable outcome (7,8). The significance of imaging modalities in the diagnosis and planning of the treatment has never been emphasized.
Our patient had a prolonged infectious period with persistent fever for 51 days and elevated CRP and SR for 23 weeks. He was infected with a PVL-positive strain of MRSA. PVL is a virulence factor believed to make soft tissue and osseous infections, as well as pneumonia, more difficult to treat (3,4). Higher values of CRP and SR in PVL-positive MRSA infections compared to PVL-negative infections have been reported in several studies (6,11) as well as more febrile days (4,6) and higher complication rates (4). Our observations are compatible with this.
The present patient’s osteomyelitis was complicated by septicemia and severe bilateral pneumonia. This is consistent with studies (5,6) showing higher rates of abscess formation and of necrotizing pneumonia in patients infected with PVL-positive strains, as well as higher rates of septicemia and occasionally septic shock (5).
Patients infected with PVL-positive MRSA often need repeated surgery and have longer recovery times than those infected with PVL-negative strains (5,6). This was true for our patient being hospitalized for 49 days and having surgical debridement every other day often guided by imaging (Table 1). We consider the osteomyelitis in our case to be severe based on the prolonged infectious period and the anatomic location. According to Cierny-Mader classification (12), which includes prognostic factors, our case could be classified as type IV, diffuse osteomyelitis, which is the most complex type to treat and a type that often become chronic (12).
We used MRI, along with clinical and paraclinical parameters, to follow the development of the osteo-articular infection and to plan ongoing treatment, whereas radiography was used to follow pulmonary involvement. MRI is eminent in detecting soft tissue and osseous edema and abscess formation (13) and is therefore a useful tool when monitoring bone infection. Especially in cases that do not respond to therapy, MRI can provide useful information (14). Ultrasonography can also be used to detect soft tissue changes, including subperiostal abscess formation in osteomyelitis, but can be misguiding in the early stage of infection (15), as experienced in our case.
In addition to MRI, we used a CT when the osteomyelitis progressed radiologically during the follow-up period. CT is superior to MRI for diagnosing sequesters and osseous destruction (16). If an ongoing osteomyelitis cannot be excluded by MRI and CT, a supplementary bone and leucocyte scintigraphy can give additional information. In the present case, the leucocyte scintigraphy was without increased uptake indicating no active infection and therefore it did not result in further surgical debridement’s or changes of antibiotics. Within months the radiological signs gradually subsided with osseous healing. Another choice of scan could have been a positron emission tomography (PET) scan (16) but this often results in false positive findings in recently operated patients.
Twenty-two months after onset of symptoms, our patient had fully recovered, with the exception of surgical scar tissue at the knee region, which was diminished by plastic surgery. According to Kefala-Agoropoulou et al. (8), only few of the published pediatric bone infections with MRSA have a favorable outcome. In their review of the literature, they found 23 cases during 1997–2007, of which 16 children died and only one had a favorable outcome. Most deaths were associated with severe MRSA pneumonia, a complication also observed in the present patient. It is commonly accepted that necrotizing pneumonia increases the risk of death and severe complications (9,17) and in an adult population, Gillet et al. found a mortality rate of 56% (18). They also found that a primary focal infection decreased the risk of death. However necrotizing pneumonia in children is still uncommon (19). The clinical features are similar to those of uncomplicated pneumonia, but the patients often fail to respond to antibiotic treatment and the disease runs a more severe course. The need for intensive care support has been reported to be greater in patients with PVL-positive staphylococcus infections (19). This was also necessary in the present patient where Linezolid was added as a toxin suppressing agents (17,19).
Despite a PVL-positive MRSA strain with initial treatment failure, development of pneumonia, and continued destruction of bone, our outcome was a healthy boy without symptoms or sign of relapse 17 months after completion of treatment. Compared to patients described in the literature with the same type of infection and clinical complications, our patient had a very favorable outcome.
This case underlines the necessity of increased focus and close interdisciplinary cooperation on children with septic arthritis and osteomyelitis, especially in cases with initial treatment failure, complicated by septicemia and pneumonia. Besides, it illustrates the paramount importance of radiological imaging and interpretation to help direct the treatment, especially the surgical debridement.
In conclusion, in the case of orthopedic infections caused by multidrug resistant bacteria and with limited effect of antibiotic therapy, the surgical treatment must be optimized in accordance with the old principles of infectious surgery; meticulous debridement, followed by lavage and dead space management, repeated until the infection is eliminated. We attribute the favorable outcome of this case to our close interdisciplinary communication resulting in sufficient radiological investigations and interpretation and repeated infectious surgery, combined with relevant antimicrobial therapy.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
