Abstract
Background
Glioblastoma with oligodendroglioma component (GBMO) is a subtype of conventional glioblastoma (cGBM), which is categorized as WHO grade IV. GBMO can be histopathologically distinguished from cGBM and the prognosis of GBMO is better than that of cGBM. However, no systematic review of GBMO imaging findings has been published to date.
Purpose
To clarify the radiological imaging features of GBMO compared with those of cGBM.
Material and Methods
The participants were 15 patients with GBMO and 32 patients with cGBM as a control group, all of whom were histopathologically diagnosed. A radiologist retrospectively reviewed the imaging findings of both computed tomography (CT) and magnetic resonance imaging (MRI) for density, signal intensity, contrast medium enhancement (CE), cortical swelling, and cortical swelling without CE. We statistically analyzed the imaging findings by Chi-squared test.
Results
Cortical swelling without CE in GBMO was significantly greater than that in cGBM (P = 0.004). Non-CE and heterogeneous solid enhancement were observed significantly more often in GBMO (P = 0.004). No other findings were significant.
Conclusion
There was significant difference in the findings of the CE, which exhibited solid heterogeneous enhancement in GBMO. Cortical swelling without CE can be considered significantly characteristic of GBMO.
Keywords
Introduction
Glioblastoma (GBM) is categorized as World Health Organization (WHO) grade IV and is one of the most malignant of all brain tumors. GBM is the most common brain tumor, accounting for approximately 12–15% of all intracranial tumors and 60–75% of astrocytic tumors (1). As GBM often exhibits broad infiltrative spread in the brain parenchyma and various fibers, it is very difficult to resect and treat entirely, even when irradiation and chemotherapy are also included. GBM also frequently recurs and causes cerebrospinal disseminations in the early phase after initial therapy (2,3). Generally speaking, the prognosis is extremely poor, with a one-year survival rate of less than 20% (4).
Glioblastoma with oligodendroglioma component (GBMO) is a subtype of conventional glioblastoma (cGBM), which is categorized as grade IV in the WHO classification 2007 (1). GBMO is histopathologically diagnosed as anaplastic oligoastrocytoma with necrosis and is presumed to develop from mixed low-grade glioma, which may result in the malignant transformation (5). GBMO has the pathological features of both glioblastoma and oligodendroglioma and also contains foci that resemble oligodendroglioma (1). These areas vary in size and frequency (1). GBMO can be histopathologically distinguished from cGBM. GBMO comprises approximately 4–20% of cGBM (5,6). GBMO frequently involves the loss of heterozygosity (LOH) 1p/19q, which is a good prognostic factor in oligodendroglioma, IDH1 mutation, etc. (7–9). Therefore, the prognosis of GBMO is better than that of cGBM (5,9,10). Although GBMO is discouraged in WHO classification 2016, this classification does state the existence of the GBM including the oligodendroglioma component (11). Some of these cases may be characteristic of IDH1 mutation (11). In terms of image analysis, it has been reported that quantitative susceptibility weighted imaging (SWI) is able to depict minute calcification in tumors. Thus, quantitative SWI may be useful in distinguishing GBMO from cGBM (12).
Oligodendroglioma (WHO grade IV) exhibits more characteristic calcification and cortical swelling compared with diffuse astrocytoma (13). We hypothesize that computed tomography (CT) and magnetic resonance imaging (MRI) will reveal that GBMO has features of both cGBM and oligodendroglioma. We also found cortical swelling without contrast medium enhancement (CE) in GBMO. No systematic review of GBMO imaging findings using conventional imaging techniques has been published to date. The purpose of this study was to clarify the radiological imaging features of GBMO compared with those of cGBM.
Material and Methods
Participants
The protocol was approved by each institution’s ethical committee. All human and animal studies were approved by the ethical committee and performed in accordance with the ethical standards of the 1964 Declaration of Helsinki and its later amendments.
We collected CT/MRI images and medical records of GBMO cases from four institutes (including our own) because of the rarity of this condition. Nineteen patients with GBMO were included. Four patients were excluded since the images had been discarded because the medical records and images of the patients who had not been admitted to hospitals for five years are not preserved in our country. The participants were thus 15 patients with GBMO (male:female ratio = 12:3; mean age, 58.7 ± 18.1 years; period, December 2006 to July 2014) in three institutes and 32 patients with cGBM as a control group (male:female ratio = 13:19; mean age, 63.6 ± 9.4 years; period, January 2010 to July 2014) in one institute. The number of normal controls was approximately twice the number of GBMO cases. All tumors were resected or biopsied by a neurosurgeon and were histopathologically diagnosed by each institute’s pathologist.
Image analysis
A diagnostic radiologist (KK) with 10 years of experience retrospectively and blindly reviewed the imaging findings of both CT and MRI for GBMO and cGBM. KK evaluated density (high/low), hemorrhage, calcification on CT, signal intensity (high/iso/low) on each MRI sequence, and CE in MRI. If the tumor had mixed density/intensity, KK recorded the dominant findings. KK evaluated both cortical swelling and cortical swelling without CE, which is characteristic of oligodendroglial tumors, in the image analysis. However, CT was not performed in one patient with GBMO. “Cortical swelling” was defined as swelling of the cortex due to tumor involvement with/without CE and “cortical swelling without CE” as the existence of a portion without CE in the swollen cortex due to tumor involvement in the T2-weighted (T2W) image and/or the T1-weighted (T1W) image and/or the FLAIR image (Fig. 1).
Finding of cortical swelling without CE. FLAIR image shows cortical swelling (a, →). T1W image with contrast medium shows faint enhancement in the white matter and the portion without CE in the swollen cortex area (b, ∇).
A neuroradiologist (MK) with 13 years of experience retrospectively examined the occurrence site of the tumor, survival period, the existence of IDH1 mutation, and MIB-1 index using the available medical records.
Statistical analysis
The overall survival rate for GBMO and cGBM was calculated and evaluated by log-rank test using the available data. The MIB-1 index between GBMO and cGBM was also evaluated by Student’s t-test using the available data. CT and MRI findings of GBMO and cGBM were analyzed by Chi-squared test. These data were statistically calculated using the Statistical Package for the Social Sciences, Version 22.0 (SPSS Inc., Chicago, IL, USA). A P value < 0.05 was considered significant.
Results
Features of each patient group.

Student’s t-test, P < 0.05 is considered significant.
Chi-squared test, P < 0.05 is considered significant.
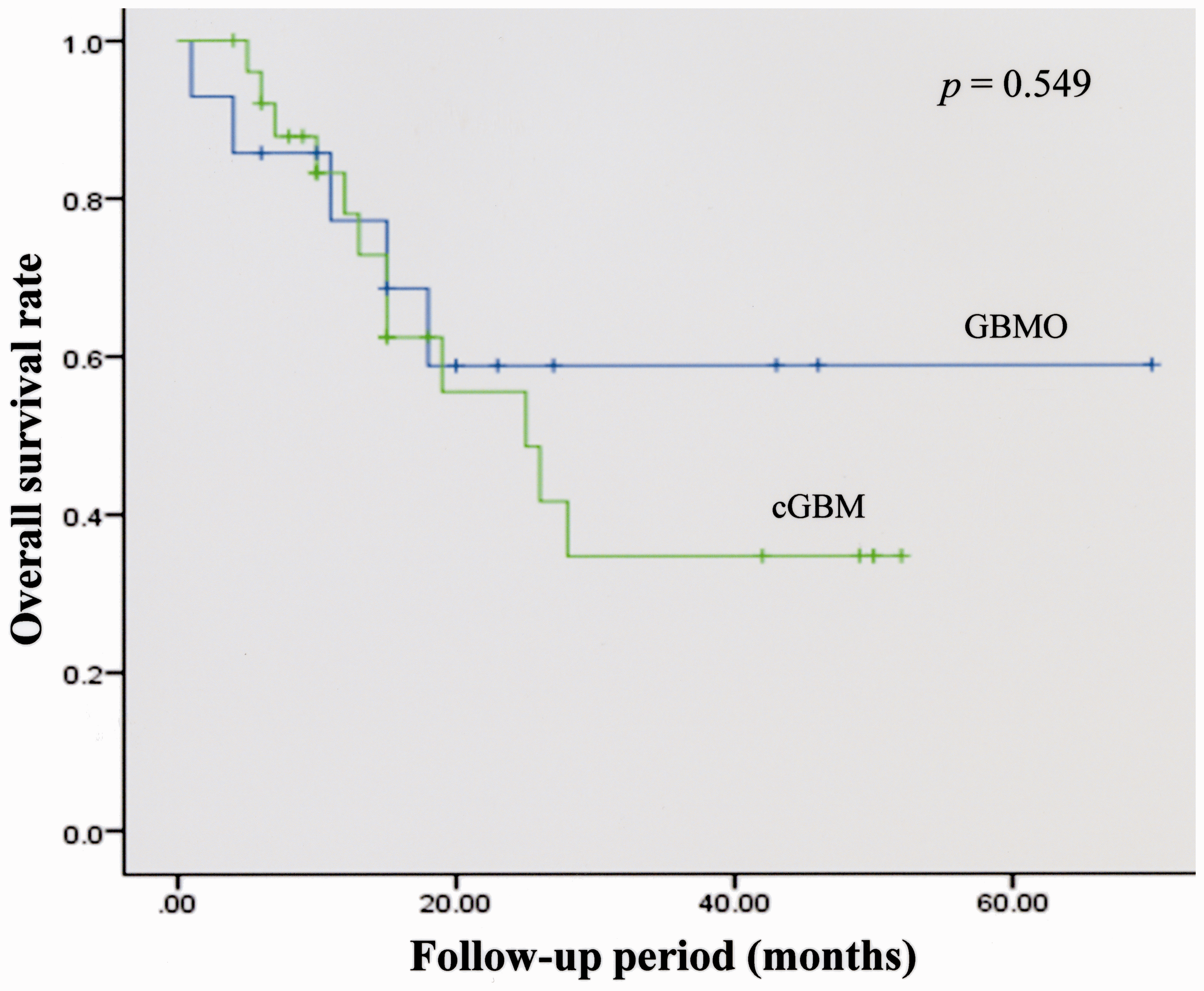
Overall survival rate in the two kinds of tumors. The overall survival rate of GBMO is superior to that of cGBMO. However, there is no statistical significance between the two tumors (by log-rank test).
Image findings of GBMOs compared with cGBM.

P value cannot be calculated.
Chi-square test, P < 0.05 is considered significant.
Discussion
We revealed that GBMO has significant difference in the findings of CE. Solid heterogeneous enhancement and cortical swelling without CE, in particular, is significantly characteristic of GBMO compared to the findings of cGBM. There is no significant difference in the other findings between the two kinds of tumors.
Oligodendroglial tumor is likely to involve the cerebral cortex histopathologically (14). Thus, cortical swelling was frequently seen in our image analysis as well. However, cortical swelling alone is not a significant finding because it is frequently seen in cGBM. In particular, cortical swelling without CE is significantly seen in 80% of GBMOs and in 34% of cGBMs (Table 2). Cortical swelling without CE was defined as the part of the cortical swelling area on T2W images and/or T1W images and/or FLAIR images without CE area in the swollen cortex. Cortical swelling without CE resembles cortical involvement in oligogendroglioma and oligoastrocytoma. It is a specific finding of GBMO and it may reflect the oligodendroglioma component in glioblastoma in this tumor (Table 2, Figs. 3–5). It is difficult to distinguish between the two kinds of tumors using other findings besides CE and cortical swelling without CE.
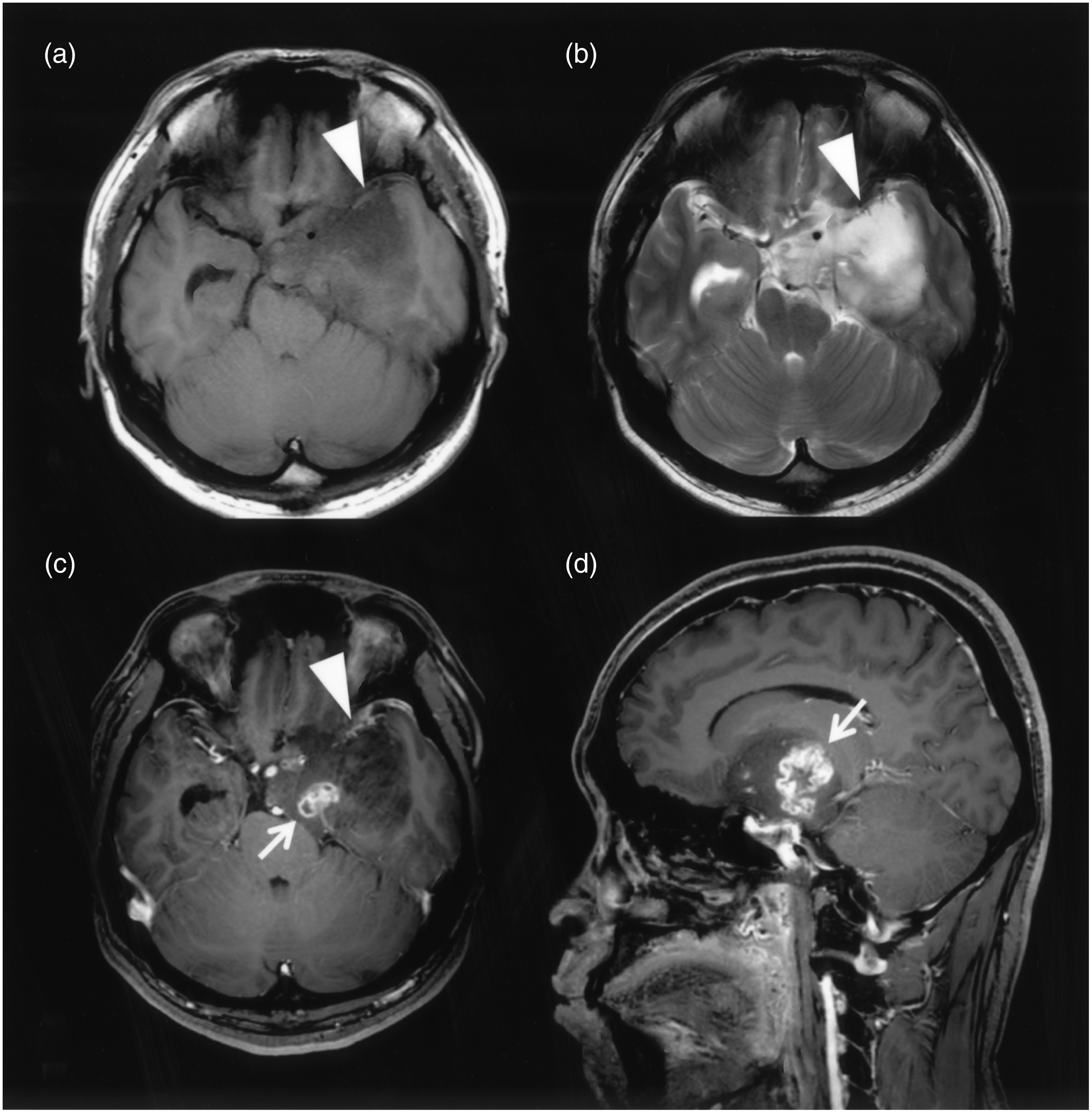
A 70-year-old man with GBMO. CT and FLAIR shows a high-density tumor-like cortex with edema in the right temporal lobe (a, b). T1W images with CE reveal solid heterogeneous enhancement (→) and cortical swelling without CE (∇) (c).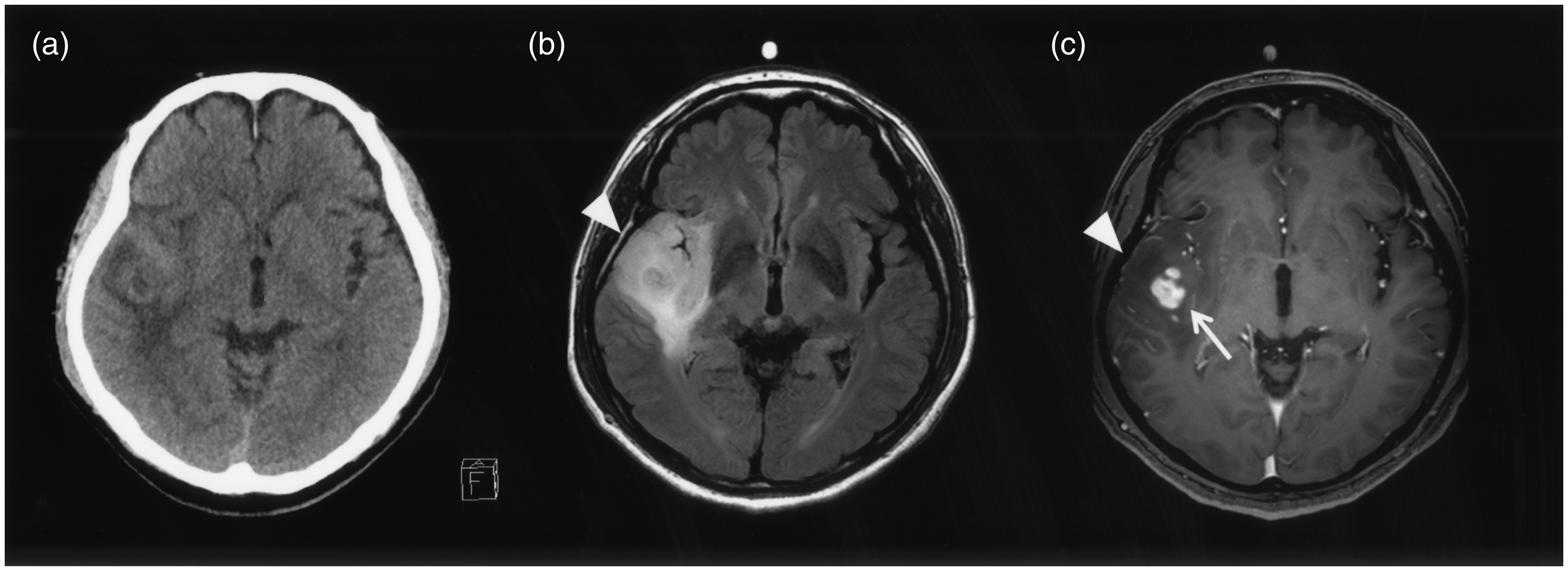
It is important to distinguish GBMO from cGBM in pretherapeutical image analysis because of the differing prognoses (5,9,10). We hypothesize that GBMO has features of both cGBM and oligodendroglioma in CT and MRI. Calcification, which is characteristic of oligodendroglioma, does not enable us to distinguish GBMO from cGBM. GBMO could not be separated from cGBM using CT. SWI may enable us to observe small calcifications or microcalcifications (12). However, SWI was unavailable in many of our patients with GBMO and cGBM. Oligodendrogliomas may have mild and/or marked solid enhancement or no enhancement at all (15,16). On the other hand, cGBM has characteristic marked heterogeneous ring-like enhancement. All of our patients with cGBMs had heterogeneous ring-like enhancement. Nine patients with GBMO showed heterogeneous solid enhancement, which differed from both heterogeneous ring-like enhancement and no enhancement (Table 2, Figs. 3 and 4). One report states that the microvascular density of GBMO is lower than that of cGBM and anaplastic astrocytoma, categorized as WHO grade III (17). This may result in the significant difference between the two kinds of tumors in terms of CE.
An 81-year-old man with GBMO. DWI shows high intensity with low ADC value (a). Both cortical swelling without CE (∇) and slight heterogeneous CE (→) is seen (b–d). A 27-year-old man with GBMO. The tumor is in the left temporal lobe. Both cortical swelling without CE (a–c, ∇) and heterogeneous ring-like enhancement (d, →) is seen.
Oligodendroglial tumors, including oligodendroglioma and oligoastrocytoma, are defined as benign tumors, categorized as WHO grade II (13,18). Oligodendroglial tumors have diffuse high-intensity areas on T2W and FLAIR images similar to diffuse astrocytomas. The coarse calcifications are characteristics of these types of tumors (15,16). It is important to note that oligodendroglial tumors are typically located in the cortex and white matter, and infiltration of the overlying leptomeninges may be seen. Oligodendroglial tumors tend to proliferate in the subcortical white matter with extension to the cortex. On the other hand, in diffuse astrocytoma, there is no destruction of the anatomical structures, such as the cortex and compact myelinated pathway (14). Therefore, cortical involvement is frequently seen in oligodendroglial tumors.
Conventional glioblastoma is the most malignant brain tumor of all astrocytic tumors, categorized as WHO grade IV. GBMO is a subtype of cGBM, which is also categorized as WHO grade IV (1,5,19). GBMO has the pathological features of both glioblastoma and oligodendroglioma and also contains foci that resemble oligodendroglioma. These areas vary in size and frequency (1). Thus, GBMO can be histopathologically distinguished from cGBM and GBMO may differ from cGBM in our image analysis. It is of clinical importance that the prognosis of GBMO is better than that of cGBM because GBMO frequently involves the loss of heterozygosity (LOH) 1p/19q, which is a good prognostic factor in oligodendroglioma, IDH1 mutation, etc. (7–9,20,21). Several studies report that GBMO has a significantly better prognosis than cGBM (5,9,10,22). Our patients with GBMO had a better prognosis (approximately 60%) compared to those with cGBM, although there was no statistical significance (Fig. 2). The prognosis of GBMO may be influenced by the oligodendroglioma component in the tumor. A high MIB-1 index indicates high proliferation and a bad prognostic factor (23). However, the MIB-1 of GBMO was higher than that of cGBM. This discrepancy needs further study through the evaluation of more GBMO cases.
This study contains several limitations. GBMO in the WHO classification 2007 is discouraged in the WHO classification 2016 (11). However, the WHO classification 2016 suggests that GBMO may consist of IDH-wild type glioblastoma, including, in particular, the small cell variant, given the morphological overlap with oligodendroglial cells, IDH-mutant glioblastoma, or IDH-mutant and 1p/19q codeleted anaplastic oligodendroglioma (11). IDH mutation was not checked in all cases because many of the patients were treated before the importance of genomic study had become widespread. We dealt with a small number of patients with GBMO because it is relatively rare. In image analysis, it may be difficult to find “cortical swelling without CE” if a radiologist is not familiar with the findings of oligodendroglial tumors, which may introduce a bias. Advanced image techniques such as SWI and texture analysis are becoming standard in tumor imaging (12,24). However, these techniques were not performed because of the retrospective nature of this study. In future studies, we need to review more patients with GBMO and to examine the findings of other modalities such as FDG-PET.
In conclusion, there is a significant difference in CE in MRI. Specifically, there is a significant difference in the findings of CE, which exhibit solid heterogeneous enhancement in GBMO instead of ring-like enhancement, compared with the findings of cGBM. In particular, cortical swelling without CE is significantly characteristic of GBMO. The finding of solid heterogeneous enhancement and cortical swelling without CE may indicate the existence of oligodendroglioma foci in the GBM.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Grant-in-Aid from the Global COE program of the Japan Society for the Promotion of Science
