Abstract
Background
Amniotic fluid (AF) is one of the major determinants of fetal biophysical profile and can predict pregnancy outcome. Abnormal value of amniotic fluid index (AFI) may indicate fetal and maternal disorders.
Purpose
To establish reference standards for AFI for this population after 28 weeks of pregnancy and evaluate the relationship between AFI, the estimated fetal weight (EFWT), and fetal gender.
Material and Methods
A prospective cross-sectional estimation of AFI and FWT was done in 385 apparently healthy pregnant women in the gestational age range of 28–42 weeks. General scanning in longitudinal, transverse, and oblique directions of the abdomen was done to assess the fetal wellbeing and determine the fetal gender for each participant. AF was measured in each of the quadrants of the abdomen. The four values of the AF were summed to get the AFI. Measurement of the head circumference, biparietal diameter, abdominal circumference, and femoral length of the fetus were obtained. The EFWT was calculated using Hadlock’s formula. The data were divided into five groups: 28–30 weeks 6 days; 31–33 weeks 6 days; 34–36 weeks 6 days; 37–39 weeks 6 days; and 40–42 weeks. AFI for the different gestational age group studied and their percentiles were obtained.
Results
AFI normogram for the local population was established. Positive and significant correlations of AF and EFWT were seen in all the gestational age groups of the women. No significant correlations between AF and EFWT was seen in male fetuses. Regression equations of EFWT and AFI were obtained for female fetuses.
Conclusion
The normal range for AFI values for the third trimester was established and could be used to evaluate AFI in the studied population. Established fetal weight should be taken into account when interpreting AFI for female fetuses but this need not be applied for male fetuses.
Keywords
Introduction
Amniotic fluid (AF) is the protective liquid contained in the amniotic sac of a pregnant uterus. It develops as a fluid-filled extracelomic cavity identified near the time of implantation even before the embryo is recognized (1). Its functions include nutritive, protective, and diagnostic functions (1–3). In some abnormal cases, the AF volume may be more or less than expected. There can be a case of virtually no fluid in the amniotic sac. This is known as anhydramnios (1). If the AF volume is less than expected, it is referred to as oligohydramnios. Polyhydramnios occurs as a result of either increased production or decreased removal of AF. A study (1) has reported that abnormally high or low AF volume was associated with a significant increase in risk of fetal death.
AF volume is a function of the fetal gestational age, the amount of water transferred to the gestation across the placental membrane, the flux of water across the amnion, the patency of the AF membrane, presence or absence of fetal anomalies (1). Water in the AF originally comes from the maternal plasma and passes through the fetal membranes based on hydrostatic and osmotic forces. When placenta and fetal vessels develop, water and solute from maternal plasma pass across the placenta to the fetus and then to the AF. AF exchange is through a bi-directional diffusion between the fetus and the AF across the fetal skin, the surface of the amnion, placenta, and the umbilical cord before keratinization. Swallowing and urination through the urethra dominate after keratinization of the fetal skin and patency of the urethra (1).
The amount of AF in the amniotic sac can be assessed non-invasively by use of ultrasound (4,5). Works done on establishing a normogram for AF volume showed that AF volume varies from place to place probably due to racial or environmental factors (6–12). Research have suggested links between AF volume, estimated fetal birth weight, and fetal gender (11,13–15). This study is aimed at establishing a normogram of AF in this population and assessing the possible contribution of fetal weight (FWT) and gender to AF volume in the third trimester of apparently normal pregnancy.
Material and Methods
A prospective cross-sectional estimation of amniotic fluid index (AFI) and FWT was done in 385 apparently healthy pregnant women with a gestation age range of 28–42 weeks. Ethical approval was obtained from Faculty of Health Sciences and Technology, Nnamdi Azikiwe University, Nnewi Campus, Nigeria. The participants lay supine on the ultrasound couch. Adequate quantity of ultrasound gel was applied on the abdomen. General scanning in longitudinal, transverse, and oblique directions of the abdomen were done to assess the fetal wellbeing and determine the fetal gender. AF was measured in each of the quadrants of the abdomen. The four values of the AF were summed to get the AFI. Measurement of the head circumference, biparietal diameter, abdominal circumference, and the femoral length of the fetus were obtained. The EFWT was calculated using Hadlock’s formula (16). The data were collected by two sonographers. Specific training on collection of data for the study was done. Reliability test was done using interclass correlation coefficient (ICC) and a value of 0.7 was obtained on a scale of 1 to 0. This show significant reliability. The average of the two measurements by the two sonographers were used as the final value for the analysis. The data were grouped into five periods: week 28–30; week 31–33; week 34–36; week 37–39; and week 40– 42, since it might be misleading when all pairs of measurements from 28 weeks onwards were analyzed together. This is because there is an inverse relationship between AFI and gestational age and direct relationship between EFWT and gestational age (16). Descriptive statistics such as mean, standard deviation, range, and percentiles of the measured AFI were calculated and the normal range of AFI in the third trimester was established. The Shapiro–Wilk test was used to test for normality of the data (P = 0.10 for AFI, P = 0.08 for FWT). Using Pearson’s correlation method, the coefficients of correlation of AFI with EFWT was derived for each of the group studied. Regression equations as well as coefficient of correlation and determination of AFI against EFWT were found for the groups that show significant correlation. The above test statistics were done at P value < 0.05.
Results
A total of 385 fetuses consisting of 206 (53.5%) male fetuses and 179 (46.5%) female fetuses, were studied. The AF decreases gradually as the gestational age advanced (Table 1). The EFWTs were shown to increase as the fetal age advanced. Fetuses in group 1 (week 28–30) had a mean AFI of 15.3 ± 1.2 SD and a mean weight of 1537 g ± 49.7 g SD. Group 2 fetuses (week 31–33) had a mean AFI of 12.8 ± 1.0 SD and a mean weight of 2053 g ± 31.0 g SD. Group 3 fetuses (week 34–36) had a mean AFI of 12.6 ± 1.1 cm SD and a mean weight of 3891 ± 31.1 g SD.
Significant positive correlations were found in female fetuses in all the gestational age groups studied Table 2. Predictive equations of AFI using values of EFWT were derived for the female fetuses Table 2.
Discussion
The range of AF across the gestational age categories is 8.05–19 cm, which is similar to results of some previous studies (6–12,17,18). This study and some of the previous studies (6–12,17,18,) showed that AFI decreases gradually as the gestational age advances in the third trimester to 42 weeks and that the range of the AFI in the third trimester (28–42 weeks) are within 8–25 cm. According to Shripad et al. (11), this range is the worldwide acceptable values. The range of AFI values obtained in this study for the different gestational age group in the third trimester were, however, not identical with previous studies (6–12,17,18) (Fig. 1).
Comparison of the current study with previous studies done in other locations. Distribution of AF across the fetal gestational age studied. Correlation and regression of AF with FWT in the male and female fetuses of the gestational age groups studied. AFI represented by “Y” (cm) and the estimated fetal weight was represented by “X” (g) in the regression equations.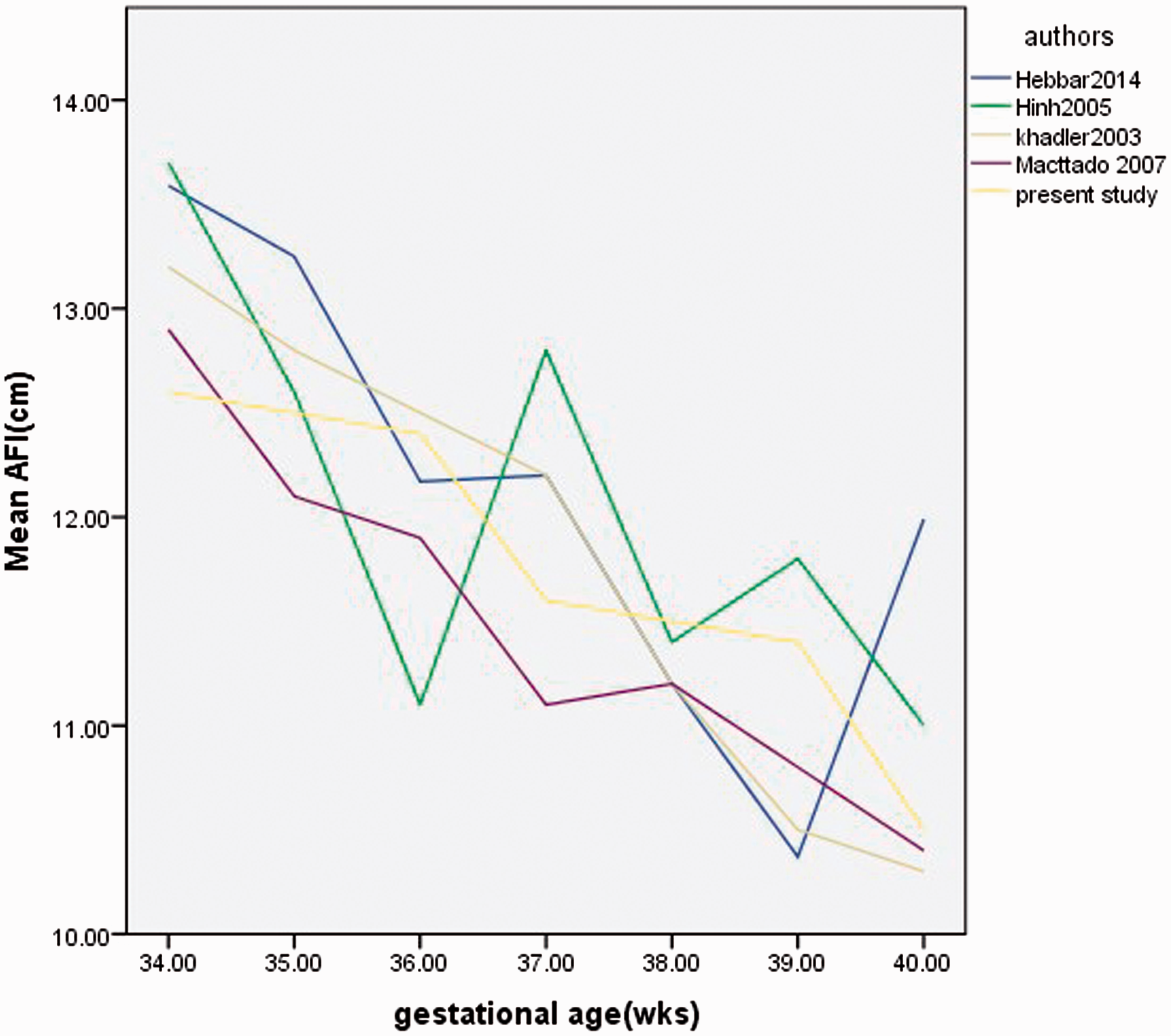


The variability could have arisen due to geographical location, race, and socioeconomic factors. Studies by Charma and Bobson (7) carried out in the northern part of Nigeria and Agwu et al. (6) carried out in southeast Nigeria were in agreement with this study. They reported that the AF decreases as the gestational age increases. Alao et al. (19), in the western part of Nigeria, reported a different pattern as they noted that the AFI increases as the gestational age advances till term (42 weeks). The study conducted by Igbinidu et al. (20) in a similar environment (southwest Nigeria) found a pattern, which is partly similar to the present study and also to that of Alao et al. (19). Igbinidu et al. (20) reported that the AFI decreases from a gestational age of 28 weeks to 36 weeks and then increases to term (≥40 weeks). Igbinidu et al. (20) reported that this might be because of the relative heterogeneity of the Nigerian population. If the minimum (5th percentile) and the maximum (95th percentile) of the AFI were considered as the normal range, it was noted that the corresponding values too were different at different gestational age; the more advanced the gestational age, the lesser the values Table 1. This is in agreement with most of the previous studies (7–12).
The fetal kidneys are the major source of AF production during the third trimester and this is done by means of fetal urine production while fetal swallowing accounts for about half of the total amount of fluid removed during third trimester (11). There is probably a greater increase in the rate and amount of fluid swallowing compared to micturition in the third trimester; this may account for the decrease in the AF volume as the gestational age advances in the third trimester. But because of the variations noted, this present study is in concordance with the previous studies (5,7,8,10–12), which showed that AFI standards have to be defined for specific populations in order to eliminate variations resulting from geographical locations, race, and socioeconomic groups.
Significant correlations were found between AFIs and EFWT in all the gestational age groups of the female fetuses Table 2. This was in agreement with most of the previous studies (13–15,21,22), which reported that AFI correlated positively with EFWT in female fetuses in the third trimester. No significant correlations were found between AFIs and EFWT in the male fetuses of all the gestational age groups (Table 2). Parveen et al. (14) and Sriram et al. (15), however, noticed a significant positive correlation between AFI and the EFWT in male fetuses in the gestational age group above 38 weeks. It was suggested that the differences in hormonal environment between female and male fetuses may be the reason for the correlation of AF and EFWT in female fetuses (14,23). Carber et al. (23) found that estrogen-treated female rats had higher water intake and greater amount of dilute urine output compared to controls. This may be due to abnormalities in synthesis and release of antidiuretic hormone. Bigger female fetuses are expected to have higher estrogen levels due to estrogen production in their adipose tissue and this could affect the synthesis, release, or action of anti-diuretic hormones (15). The finding by Ademola et al. (24) and Nitin et al. (25) are not in agreement with this study. Ademola et al. (24) found an overall correlation coefficient between AFI and the EFWT to be 0.241 (P > 0.05). He neither categorized the data into male and female nor grouped the data into gestational age groups. This could be why the difference exists from Ademola et al.’s study. Nittin et al. (25) found the same correlation coefficient in all the gestational age subdivisions (P > 0.05; r = 0.413) and did not group the data into male and female. This may be the cause for the difference in the study compared to this present study.
Fetal size should therefore be considered in evaluation of AFI in female fetuses of all the gestational age groups in the third trimester. In male fetuses, however, it might not be needed to take into consideration the EFWT in the evaluation of AFI. Regression equations for the prediction of AFI using the estimated weight of the fetus were therefore derived for the female fetuses.
In conclusion, the normal range for AFI values for the third trimester was established and could be used to evaluate AFI in the studied population. EFWT should be taken into account when interpreting AFI for female fetuses but this need not be applied for male fetuses. Carrying out this study in a larger population per categories of the fetus would help in the validation of the correlation of AFI with EFWT in female fetuses for clinical application.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
