Abstract
Interleukin-2 (IL-2) associated cholecystopathy is a rare manifestation of IL-2 drug toxicity in the setting of cancer immunotherapy. While the imaging data and clinical presentation can easily mimic acute cholecystitis, the correct diagnosis can be made with the particular clinical history, thus avoiding inappropriate surgical management. As more cancer immunotherapies become standard oncologic treatments, specific immunotherapy-associated side effects are also expected to be encountered more frequently in the future and should be recognized as such. We present a case of IL-2-associated cholecystopathy in the setting of renal cell carcinoma immunotherapy.
Introduction
Cancer is one of the leading causes of death in Western countries, whose incidence is expected to concomitantly rise with the predicted increase in life expectancy around the world. Patients receiving immune-based cancer therapies have recently shown significant responses to certain types of cancer, and these “immunotherapies” are thus predicted to be gradually embraced by the oncology community worldwide. As such, the unique morbidities associated with these treatments are also expected to be more frequently encountered by both oncologists and non-oncologic physicians. Greater familiarity with these potential complications can allow radiologists and other non-oncologic physicians involved with such patients to appropriate identify and manage these issues as they arise.
The cytokine interleukin-2 (IL-2) is a cancer immunotherapy agent that has previously been shown to have some efficacy in the treatment of certain metastatic cancers, and is currently utilized as a first-line treatment for both metastatic renal cell carcinoma and melanoma. High dose IL-2 therapy has several known, potentially morbid toxicities, the most recognized being capillary leak syndrome with hypotension and peripheral edema. Here we present a renal cell carcinoma patient diagnosed with IL-2-associated cholecystopathy, an unusual toxicity that can closely mimic acute cholecystitis. We also briefly discuss other gallbladder pathologies to be considered in the differential diagnosis.
The following case was reviewed and evaluated according to the University of South Florida’s Institutional Review Board Policy #311.
Case report
A 32-year-old man presented to the hospital with complaints of acute right-sided abdominal pain. His past medical history was remarkable for renal cell carcinoma initially diagnosed at the age of 26 years. At that time, there was no evidence of metastatic disease, so he underwent surgical resection of the mass. On a recent visit, a left supraclavicular lymph node was palpated on physical exam, prompting evaluation with a computed tomography (CT) study. This demonstrated multiple, pathologically enlarged cervical and supraclavicular lymph nodes. These nodes were resected with pathologic confirmation of metastasis. Further assessment with an 18 F-fluorodeoxyglucose (FDG) PET-CT study showed several other enlarged hypermetabolic abdominal lymph nodes (Fig. 1). The patient was then started on high-dose IL-2 chemotherapy. His planned treatment schedule called for daily IL-2 infusions (600,000 IU/kg IV/dose) for 10–14 days for four cycles. After completing eight of the planned 14 doses of the initial cycle of IL-2, the patient experienced acute onset of severe right-sided abdominal pain for which he presented to the hospital. Laboratory tests obtained shortly after the onset of symptoms revealed abnormally elevated findings: WBC count = 15.6 K/µL (normal range, 4.0–10.9 K/µL), total bilirubin was also within normal limits, measuring 0.90 mg/dL (normal range, 0.00–1.20 mg/dL), and alkaline phosphatase = 234 U/L (normal range, 40–130 U/L). Liver function tests were within normal limits, with AST = 28 U/L (normal range, 10–50 U/L) and ALT = 39 U/L (normal range, 0–41 U/L).
Axial CT image from a recent FDG PET-CT study 1 month prior to IL-2 chemotherapy initiation demonstrated bulky retroperitoneal lymphadenopathy (white arrows). No associated gallbladder hypermetabolism or significant gallbladder wall thickening was seen (black arrows). Gallbladder wall thickness = 2 mm.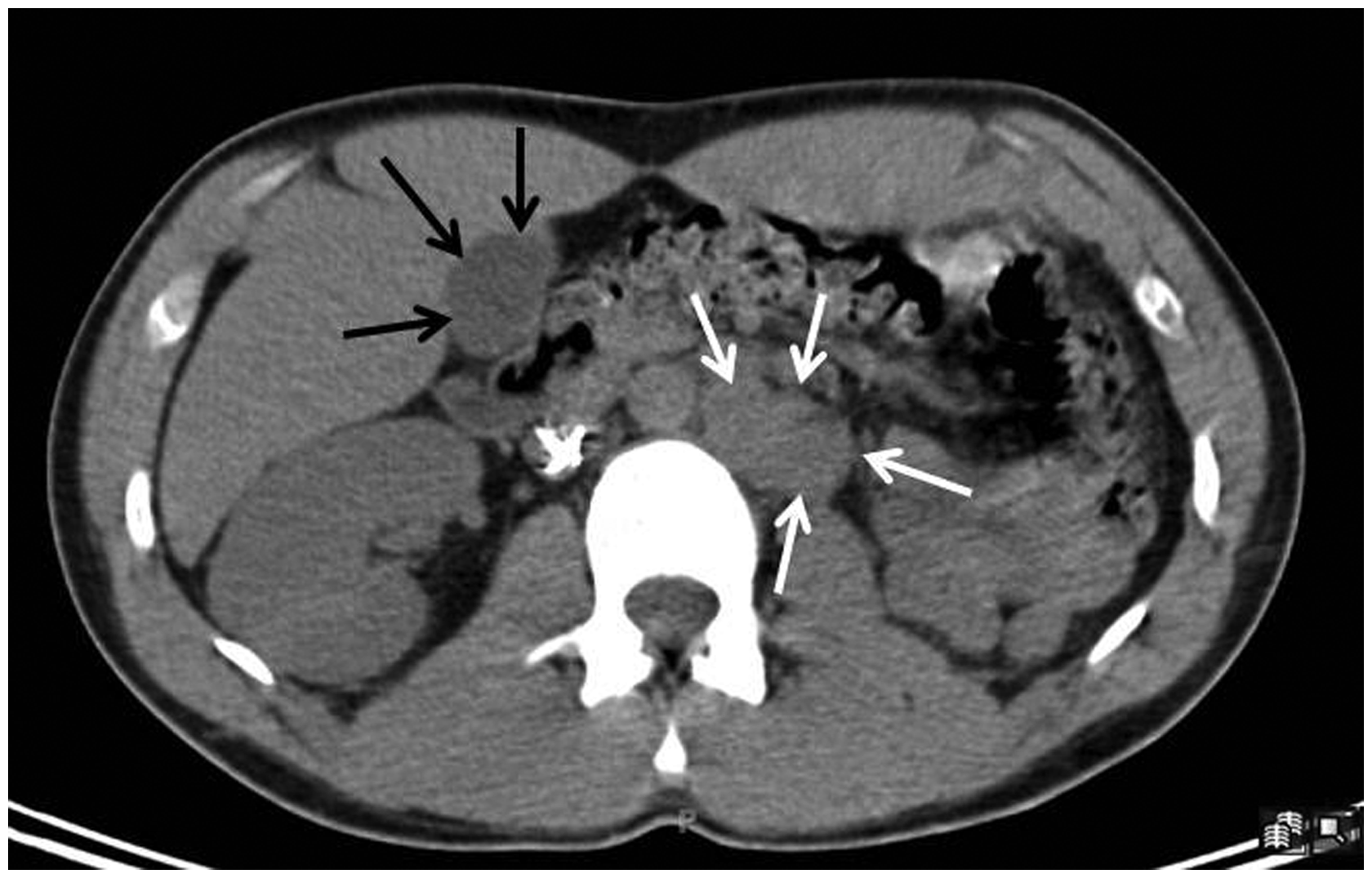
A right upper quadrant abdominal ultrasound was subsequently ordered and revealed heterogeneous echotexture and marked uniform thickening of the gallbladder wall (up to 16 mm), with trace pericholecystic fluid present. No echogenic gallstones were definitively identified. The common bile duct was not abnormally dilated, measuring less than 6 mm in maximum diameter (Fig. 2). The gallbladder wall thickening and pericholecystic fluid were not present on the most recent available prior imaging study, the aforementioned PET-CT obtained 1 month prior.
Axial sonographic image through the gallbladder demonstrates gross circumferential gallbladder wall thickening ( = 16 mm, white measurement markers) and heterogeneous echotexture.
The sonographic findings in combination with the particular clinical history were thought to be consistent with IL-2-associated cholecystopathy. The remaining IL-2 doses were thus discontinued, and the patient was discharged. By the patient’s next clinic appointment 3 weeks later, his abdominal pain had completely resolved. A follow-up abdominal CT demonstrated complete resolution of the previously identified gallbladder wall thickening (Fig. 3).
An axial CT image obtained 2 months after cessation of IL-2 therapy shows that the previously identified gallbladder wall thickening had resolved (white arrow; gallbladder wall thickness ≤2 mm).
Discussion
High-dose IL-2 therapy is currently used as a first-line treatment for both metastatic renal cell carcinoma and melanoma, for which it has been shown to improve durable responses and increase survival. This reported success has been attributed to the ability of IL-2 to promote development of activated, tumor-targeting T lymphocytes. Unfortunately, there are known, potentially severe morbidities with high dose IL-2 administration. These various side-effects are believed to be the result of immune mediated damage to the endothelium and increased release of vasoactive substances, leading to marked vascular permeability (i.e. capillary leak syndrome) (1). IL-2-associated toxicities include tachycardia, hypoxia from pulmonary edema, constitutional symptoms, renal insufficiency, and mental status changes. High dose IL-2 therapy is also associated with certain gastrointestinal side-effects such as nausea, vomiting, and diarrhea. Cholestasis and hepatic injury have also been described.
An uncommon side-effect associated with IL-2 immunotherapy is the development of cholecystopathy, as characterized by edematous thickening of the gallbladder wall. With regards to IL-2-associated cholecystopathy, there have been only a few case reports describing the development of visible gallbladder abnormalities with high-dose IL-2 administration. These reports were described in patients receiving IL-2 for underlying HIV infection (2,3). These patients presented with right upper quadrant abdominal pain and exquisite abdominal tenderness upon palpation within several days of initiating therapy, mimicking classic acute cholecystitis. Imaging of the gallbladder in these patients most often revealed marked gallbladder wall thickening without evidence of gallstones.
To our knowledge, there has been only a single published report describing IL-2-associated cholecystopathy in the setting of cancer therapy, supported by pathologic confirmation (4). In this patient, an incorrect diagnosis of acute cholecystitis had been originally made, and a cholecystectomy was performed. Subsequent histologic analysis of the resected gallbladder revealed markedly atypical findings for classic acute cholecystitis: extensive eosinophil and T cell infiltration, but no significant infiltration by polymorphonuclear leukocytes. As such, the case was described as “acalculus lymphoeosinophilic cholecystitis”. Contrary to classic acute cholecystitis, IL-2-associated cholecystopathy does not require surgical or even significant medical intervention, but has been found to resolve within several days to weeks upon the cessation of IL-2 therapy – along with identified radiologic abnormalities. The key features that allowed confident diagnosis of IL-2-associated cholecystopathy in this particular case is the absence of appreciable gallstones, in combination with the clinical history. Another more subtle sonographic imaging clue to the diagnosis is the lack of marked luminal gallbladder distention relative to the degree of gallbladder wall thickening (Fig. 2), implying no significant mechanical outlet obstruction. As the presence of gallstones may potentially complicate the diagnosis of IL-2-associated cholecystopathy vs. acute gallstone-associated cholecystitis, a nuclear hepatobiliary (DISIDA) may be helpful for determining the correct diagnosis in such situations. In this particular case report, findings of a negative hepatobiliary scan were used to support the diagnosis of IL-2-associated cholecystopathy over acute cholecystitis (5).
As IL-2-associated cholecystopathy has mostly been described in the absence of gallstones on sonographic exam, the primary considerations for the imaging differential diagnosis are congestive cholecystopathy and acute acalculous cholecystitis. Acute acalculous cholecystitis, i.e. acute inflammatory changes with edematous thickening of the gallbladder wall in the absence of gallstones, has traditionally been described in debilitated or critically ill patients undergoing severe stress, trauma, surgery, or multisystemic failure. It has been seen in up to 10% of cases of acute cholecystitis and has been described in the setting of a variety of infections, such as hepatitis, as well as opportunistic infections in AIDS patients (6–8). Congestive cholecystopathy refers to edematous gallbladder wall thickening secondary to chronic liver diseases, the etiology of which is likely to be secondary to portal hypertension (9). Portal hypertension is most frequently encountered in the background of cirrhosis. There are, however, numerous other non-cirrhotic causes of portal hypertension that may present with gallbladder wall thickening, including hepatic veno-occlusive disease after recent hematopoietic stem cell transplantation, tumor infiltration/replacement of the liver (lymphoma), granulomatous diseases (schistosomiasis, sarcoidosis), drugs/toxins (arsenic, vinyl chloride), or portal vein thrombus. Autoimmune diseases such as biliary cirrhosis, sclerosing cholangitis, and autoimmune pancreatitis may also lead to portal hypertension and subsequent gallbladder wall thickening (10). In all of these cases, clinical history is of paramount importance for diagnostic determination and differentiation from IL-2-associated cholecystopathy.
In conclusion, interpretation of abnormal gallbladder findings is dependent on the particular clinical context. In cancer patients receiving IL-2 immunotherapy, IL-2-associated cholecystopathy may present with findings that can closely mimic those of acute cholecystitis. Failure to consider this possibility may result in inappropriate surgical intervention that potentially exposes the patient to unnecessary surgical morbidities.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
